Our group is interested in controlling crystallization, and if we can challenge established thinking in the process, so much the better! Previously, we showed how thermodynamic control of crystallization could be achieved in droplet microemulsions1 – nm-sized droplets stabilised by surfactants of a liquid (or solution) dispersed in another immiscible liquid. Thermodynamic control in crystallization is beneficial because it enables stable polymorphs to be obtained and identified, eliminating the danger of a new drug transforming into a more stable, and crucially less soluble, form post-marketing, as happened in the infamous Ritonavir case2. Using our microemulsion methodology, we demonstrated how stable polymorphs could be achieved for simple covalent molecules under conditions that would normally produce metastable forms1,3-5.
A key question was whether this same methodology could be applied to giant covalent structures such as quartz. Thermodynamic control requires reversibility; breaking and reforming intermolecular bonds under ambient conditions is energetically feasible, breaking and reforming covalent bonds far less so! Synthesising nanoquartz under ambient conditions would challenge the common preconception that high temperature and pressure conditions were always needed. Furthermore, it would represent a significant advance because the selective synthesis of nanoquartz had never been achieved previously under any conditions...
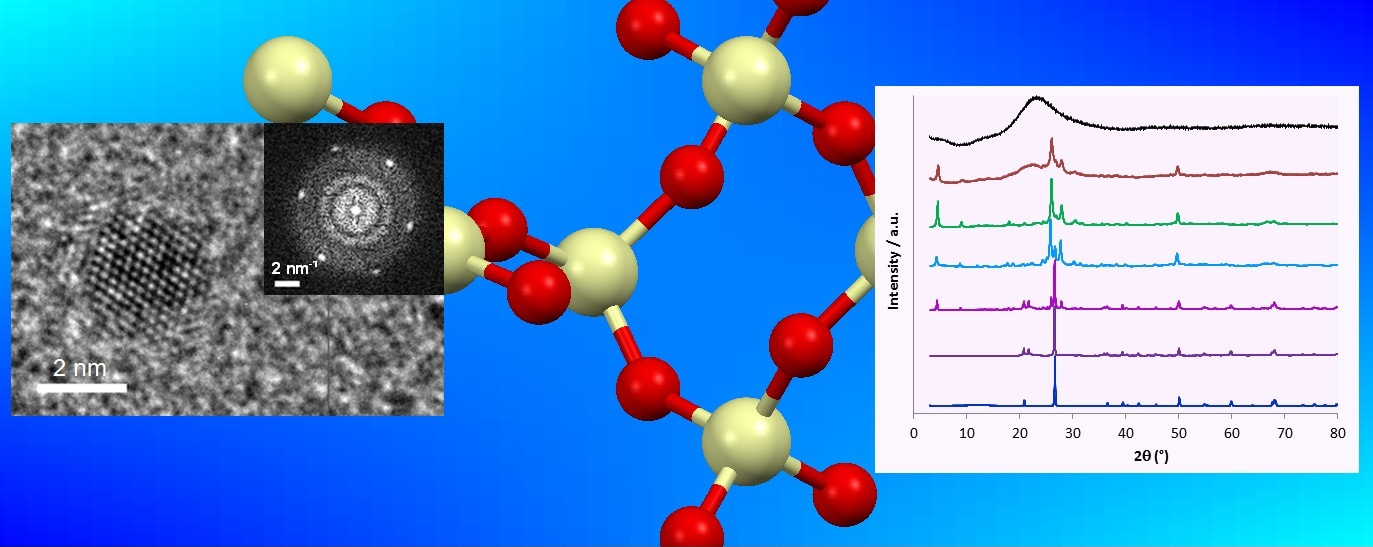
It is well-known that amorphous silica can be obtained by acidifying sodium metasilicate solutions. We believed that by confining the sodium metasilicate to the interior of microemulsion droplets, thermodynamic control would produce nanoquartz instead of amorphous silica and that this would occur even without acidification. Unusually(!), success followed rapidly with nanoquartz being readily identified using TEM. We speculate that the required Si-O bond breakage and formation is facilitated by large fluctuations in alkalinity in the droplets. This arises due to the entrapment and then exchange of hydroxyl and rarer hydronium ions during collisions that are sufficiently energetic to produce transient dimers.
This early success was delightful but quickly tempered by our inability to initially extract the nanoparticles for further characterisation. The large surfactant:silica mass ratio of >800:1 and the 2 nm nanoquartz size meant that centrifuging produced far more surfactant in the precipitate than nanoquartz. Fortunately, the nanoquartz particles aggregate in one of our chosen microemulsion systems and after 2 weeks, sufficient quantity precipitated in scaled-up experiments to obtain viable quantities for further analysis. XRD, FTIR and 29Si ssNMR analysis confirmed that nanoquartz had indeed been synthesized under ambient conditions. The 2 nm nanoquartz had a perturbed structure compared to bulk a-quartz – not unexpected given the large surface contribution in our 2 nm particles. This presented us with an ideal opportunity to study the growth and annealing of the nanoquartz under mild hydrothermal conditions. Accordingly, we were able to synthesize nanoquartz with sizes of 2-5 nm and controllable structure for the first time. We hope that this nanoquartz, free from other contaminants, will be ideal for studies on lung diseases such as silicosis, where respiratory quartz is implicated. Furthermore, our two-stage microemulsion-hydrothermal synthesis methodology enables significant control over the size and surface structure of nanocrystals. Perhaps this same methodology could provide benefits by optimizing many nanocrystal catalysts?
You can read the full story in our Communications Chemistry article here.
See Phil making the nanoquartz in the video here - nanoquartz: easy to synthesise much harder to extract!
References
- Nicholson, C. E., Chen, C., Mendis, B. & Cooper, S. J. Stable polymorphs crystallized directly under thermodynamic control in three-dimensional nanoconfinement: a generic methodology. Cryst. Growth Des. 11, 363-366 (2011).
- Chemburkar, S. R. et al. Dealing with the Impact of Ritonavir Polymorphs on the Late Stages of Bulk Drug Process Development. Org. Proc. Res. Dev. 4, 413–417 (2000).
- Chen, C., Cook, O, Nicholson, C. E. & Cooper, S. J. Leapfrogging Ostwald’s rule of stages: crystallization of stable γ-glycine directly from microemulsions. Cryst. Growth Des. 11, 2228-2237 (2011).
- Nicholson, C. E. & Cooper, S. J. Crystallization of mefenamic acid from dimethylformamide microemulsions: obtaining thermodynamic control through 3D nanoconfinement. Crystals 1, 195-205 (2011).
- Chen, C., Nicholson, C. E., Ramsey, H. E. & Cooper, S. J. Non-classical crystallisation of dipicolinic acid in microemulsions. Cryst. Growth Des. 15, 1060-1066 (2015).




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in