Potential membrane-tethered Metallo-β-lactamase isolated from emerging pathogen, C. indologenes.
Published in Microbiology
The discovery of antibiotics is one of the greatest breakthroughs in therapeutic medicine as they have saved and continue to save an immeasurable number of lives from deadly bacterial infections. However, the overuse of antibiotics has promoted the development of antimicrobial resistance within the bacterial populations. Clinicians have limited treatment options against antibiotic resistant bacteria as we are heading towards a ‘post-antibiotic’ era.
Due to the large amount of antibiotics persisting in healthcare environments and wastewater, they have become a significant driver in the development of antimicrobial resistance as they can select for resistance bacteria. Environmental bacteria such as Elizabethkingia, Pedobacter, and Chryseobacterium are increasingly found in healthcare environment and can cause outbreaks. Chryseobacteria are environmental bacteria that are primarily found in soil and water and are ubiquitous in nature. In clinical settings, it is frequently detected on humid surfaces of indwelling devices such catheters. These bacteria are associated with various types of infections, including pneumonia, sepsis, cystic fibrosis, and COVID. Chryseobacterium is intrinsically resistant to β-lactams including carbapenems and to the last-resort antibiotic, colistin, hence the use of last-resort antibiotics in healthcare can select for Chryseobacterium.
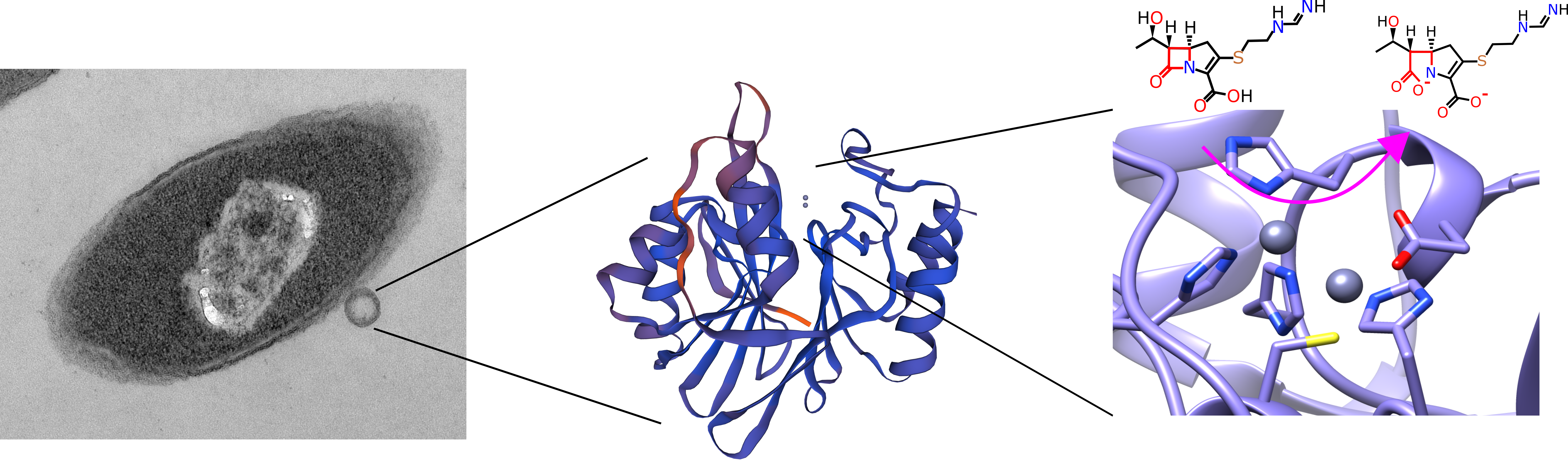
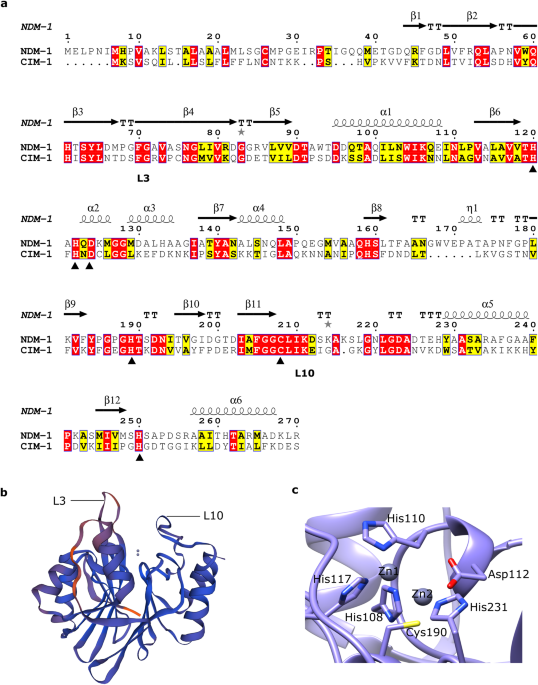
In this study, we identified and characterized a previously unknown β-lactamase from C. indologenes isolated from a healthcare environmental sample. This protein was named CIM-1 for C. indologenes metallo-β-lactamase (MBL). β-lactamases are enzymes that hydrolyse and inactivate β-lactam antibiotics. MBLs are the most concerning β-lactamases due to their broad substrate spectrum. Moreover, none of the commercially available inhibitors are active against these enzymes thus they pose a great threat to public health.
We showed that CIM-1 is most probably a membrane tethered enzyme; only the second membrane-associated MBL to be characterized after the notorious and clinically relevant, New-Delhi-metalloprotease (NDM-1). Association with the outer membrane renders proteins such as CIM-1 resilient against degradation and facilitates their incorporation into outer membrane vesicles. CIM-1 has a very high affinity for all β-lactam antibiotics including the carbapenem antibiotics so it will very effectively hydrolyse antibiotics when it is excreted in outer membrane vesicles. C. indologenes is often found as co-infections together with other, and often more virulent pathogens. Here the high-affinity CIM-1 excreted through vesicles could efficiently hydrolyse β-lactam antibiotics in the surrounding environment and so confer antibiotic resistance upon the whole community of pathogens.
Treating these infections can be a challenge as other dangerous pathogens which are originally susceptible to the treatments may be able to survive the antibiotic treatments.
In addition to CIM-1, we also identified other potentially lipidated MBLs among the homologous of CIM-1. Not only do outer membrane vesicles contain proteins, but they also mediate horizontal gene transfer. This has already been observed in the dissemination of NDM-1. Thus, we anticipated that more and more membrane tethered MBLs may be identified and eventually these MBLs will spread to other bacteria through mobile genetic elements, which highlights the necessity of further investigation of lipidated MBLs and their impact on antibiotic resistance in multi-species infections.
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
Mechanistic insights into human host and microbiome interactions
Publishing Model: Open Access
Deadline: May 31, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in