Real-time signal processing via chemical reactions for a microfluidic molecular communication system
Published in Electrical & Electronic Engineering and Protocols & Methods
Molecular communication (MC) is the process of using chemical molecules to exchange information, where MC systems rely on molecules to carry information between a transmitter and a receiver. More common wireless communication systems, such as mobile phones and internet-connected devices, rely on electromagnetic waves to convey information.
MC systems are typically used when communication is needed at the microscale, like between two cells, and are used in situations where traditional wireless communication would be unsafe, such as in the body. Often, the materials in such electronic devices can cause irritation or inflammation when in direct contact with living tissues, and the presence of strong electromagnetic fields in Wi-Fi can interfere with biological processes like heartbeat regulation – leading to unintended consequences.
In both molecular communication systems and traditional systems, several techniques are needed to enhance the quality of the connection, and this is referred to as signal processing. While it is often thought of as solely being used for telecommunication, signal processing is also used in manufacturing, sensor technology, and beyond.
Traditionally, when scientists have created and deployed MC systems in the real world, they have focused on demonstrating the feasibility of exchanging information via different molecules, e.g., acids, DNA molecules, and colour pigments, while leaving all signal processing tasks to electronic devices. These electronic devices would effectively encode an electrical signal of 0s and 1s into molecules, using them to carry information from a transmitter to a receiver, where they would then have those signals translated back to binary.
However, the utilisation of such devices limits the extent to which MC systems can be used for biomedical applications, such as the restoration of damaged tissue and smart drug delivery. The electrical elements of historic MC systems are not biologically compatible, and the complexity involved in reducing their size means they are often unsuitable to function at the necessary scale.
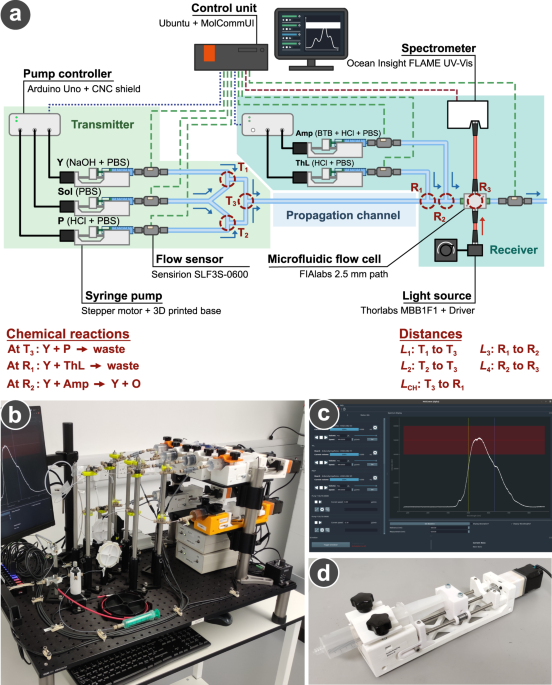
To solve this problem, a team led by Dr. Yansha Deng with postdoctoral researchers Dr. Vivien Walter and Dr. Dadi Bi from King's College London proposed to use chemical reactions in a microfluidic system for signal processing, replacing the biologically incompatible electronic components.
A microfluidic system manipulates very small amounts of fluid using many channels roughly 0.001 mm in size. Accordingly, these systems can carry out a number of separate chemical reactions simultaneously. By designing three separate chemical reactions to achieve three different signal processing functions, the MIMIC platform can effectively replicate the function of electronic signal processing components, without their dangers. Furthermore, the small size of these systems means they are cheap to produce, and as they carry information in relatively few molecules, that information can be analysed rapidly.
The team designed a reaction at the transmitter to adjust the width and optimise a transmitted signal, and two at the receiver to remove interference from the signal and then amplify it to a level where it could be detected. The result was a signal with minimal losses traveling lengths of up to 25m.
Dr. Deng said, “For the first time, our work has shown that man-made molecular communication devices can process chemical signals without electronic devices and thus be biologically compatible. We’ve used acid-base reactions as a proof of concept, but the information carriers can be swapped around, whether that be cells of glucose or man-made nanoparticles. By allowing for this modification, this approach has opened the door to a bevy of biological applications where there previously were none.
“For example, with a biologically compatible system, you could integrate sensor technology directly into someone with diabetes bloodstream. This could sense the concentration of glucose in the blood and automatically release insulin to bring that level down if it exceeds a dangerous threshold.
“The next step for us is to downsize the system we’ve built to a compact, light, and portable microscale device, and to adapt our platform with application-specific chemical reactions. Now the framework has been established, the torch has now been passed to future researchers to re-tool our prototype to their area of research and application, which pushes the dial not just on effective healthcare, but also in manufacturing, environmental regulation, and beyond.”
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in