Spatio-temporal dynamics enhance cellular diversity, neuronal function and further maturation of human cerebral organoids
Published in Bioengineering & Biotechnology
The bioengineerined and whole matured human brain organoids stand as highly valuable three-dimensional in vitro brain-mimetic models to recapitulate in vivo brain development, neurodevelopmental and neurodegenerative diseases. Various instructive signals affecting multiple biological processes including morphogenesis, developmental stages, cell fate transitions, cell migration, stem cell function and immune responses have been employed for generation of physiologically functional cerebral organoids. However, the current approaches for maturation require improvement for highly harvestable and functional cerebral organoids with reduced batch-to-batch variabilities.
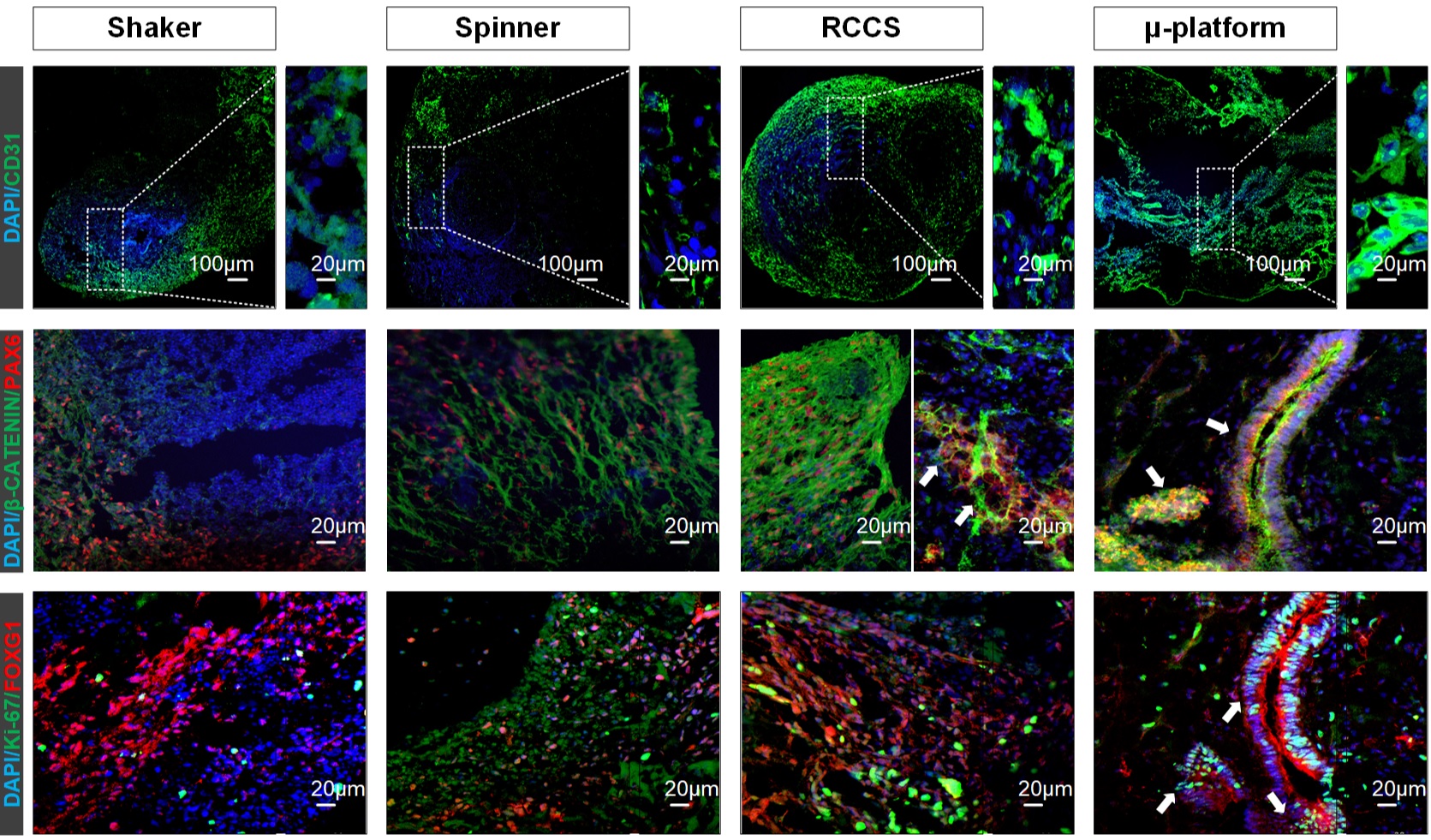
In this study, we demonstrate two different engineering approaches, the rotating cell culture system (RCCS) microgravity bioreactor and a newly designed microfluidic platform (µ-platform) to improve harvestability, reproducibility and the survival of high-quality cerebral organoids and compare with those of traditional spinner and shaker systems. RCCS and µ-platform organoids have reached ideal sizes, approximately 95% harvestability, prolonged culture time with Ki-67+/CD31+/β-catenin+ proliferative, adhesive and endothelial-like cells and exhibited enriched cellular diversity (abundant neural/glial/ endothelial cell population), structural brain morphogenesis, further functional neuronal identities (glutamate secreting glutamatergic, GABAergic and hippocampal neurons) and synaptogenesis (presynaptic-postsynaptic interaction) during whole human brain development. Both organoids expressed CD11b+/IBA1+ microglia and MBP+/OLIG2+ oligodendrocytes at high levels as of day 60. RCCS and µ-platform organoids showing a high level of physiological fidelity can serve as functional preclinical models to test new therapeutic regimens for neurological diseases and benefit from multiplexing.
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Dec 31, 2026
Cell death and inflammatory signalling
Publishing Model: Hybrid
Deadline: Oct 28, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in