Surface Alanine Braille Guides the Discovery of Electron Mediator Binding Sites in CO Dehydrogenase
Published in Chemistry
In our latest research, we tackled a challenge in the field of gas-converting enzymes1: pinpointing the mediator binding sites crucial for electron transfer. While it might seem straightforward, identifying these sites in complex enzyme systems is challenging. This complexity arises partly because it was traditionally believed that electron mediators facilitate electron transfer without directly binding to the enzymes. Yet, the presence of KM values for these enzymes with electron mediators2 suggests a more specific interaction, indicating direct binding sites, a theory our study sought to confirm and expand on.
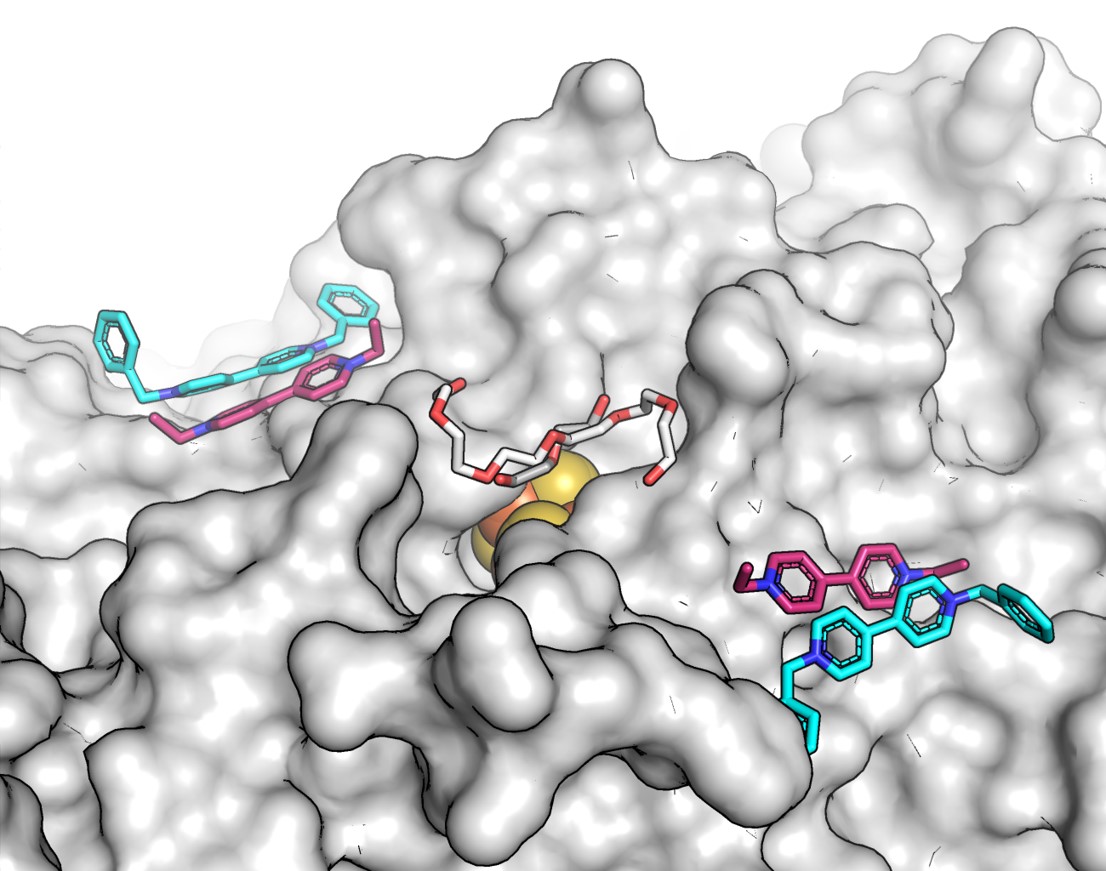
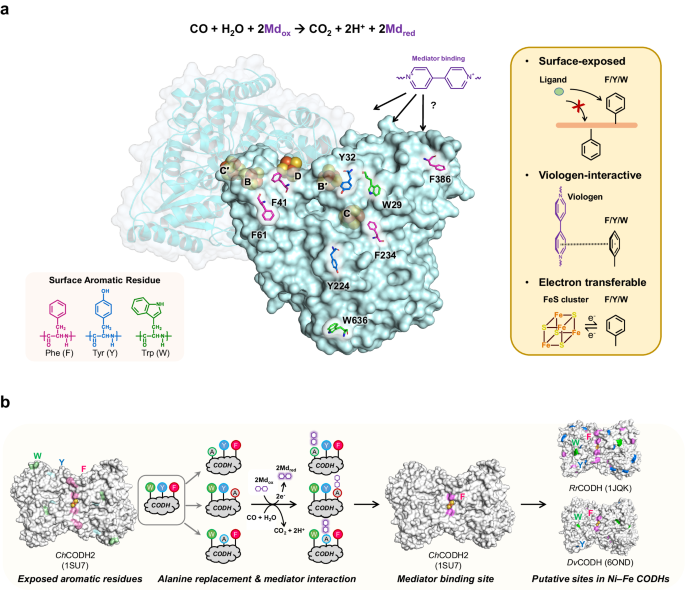
We focused on carbon monoxide dehydrogenases (CODH) and their interaction with electron mediators like viologens, which are key to improving the cleanup of industrial off-gases2,3. By employing surface alanine scanning (akin to alanine braille) to identify interaction sites, we've discovered where these mediators bind with the ChCODH enzyme from Carboxydothermus hydrogenoformans, advancing the optimization of CODH for industrial gas purification (Figure 1).
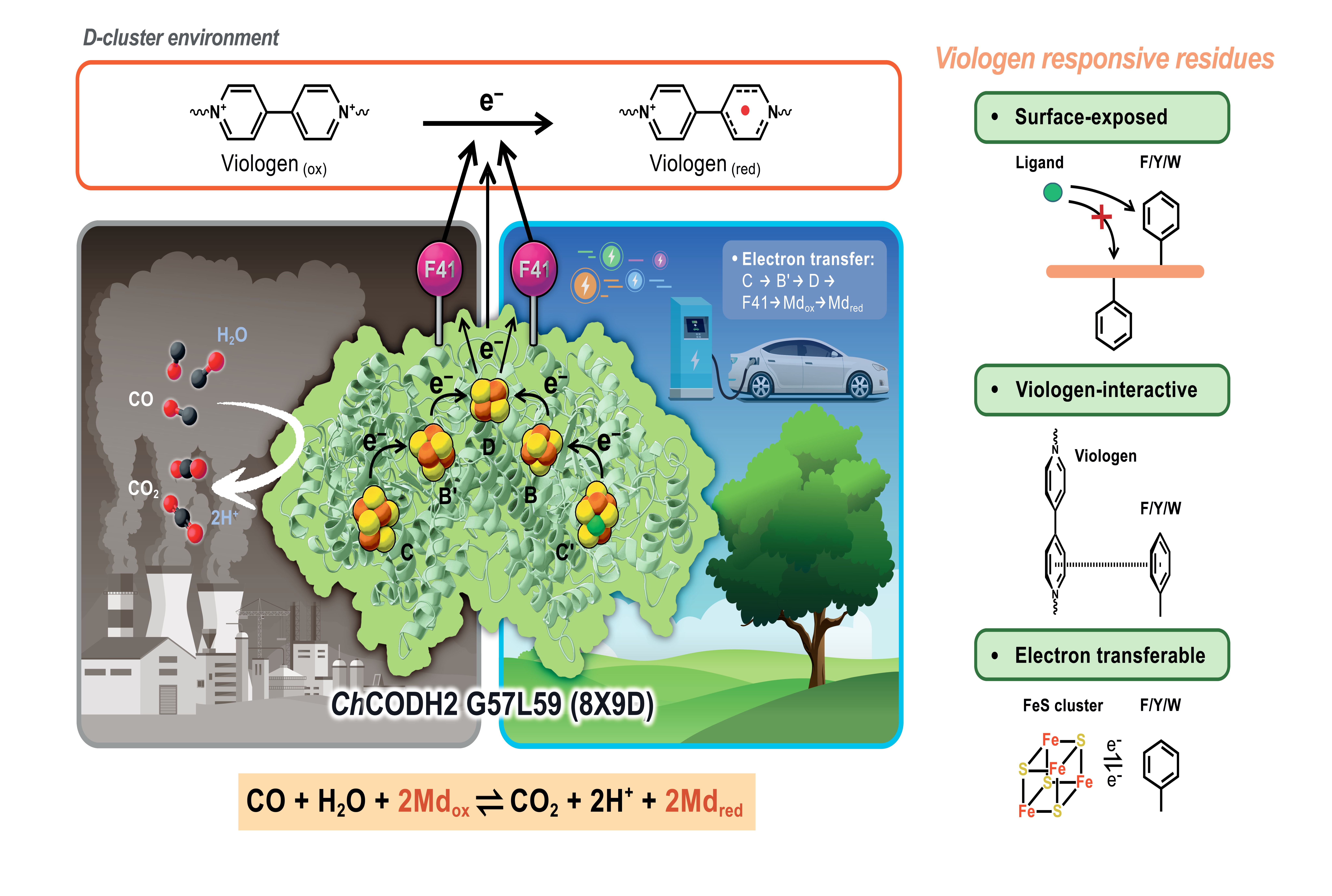
Our findings reveal that modifying the D-cluster interaction site of the enzyme, particularly through R57G/N59L mutations, significantly increases its affinity for ethyl viologen. This adjustment not only improves the enzyme's performance but also demonstrates its application potential across various waste gas treatments, underlining its broad industrial relevance.
This research offers a greener approach for repurposing waste gases from industries like steel production, converting them into valuable compounds4,5. It presents an eco-friendly alternative to conventional gas treatment methods, which are often polluting and resource-intensive.
Understanding enzyme-mediator interactions is crucial for designing efficient biocatalysts. Our study clarifies how specific amino acids on the enzyme's surface facilitate these interactions, providing a blueprint for developing more effective biocatalysts. This knowledge is useful for advancing industrial biotechnology, positioning enzymes as feasible tools for sustainable practices.
Overall, our work not only deepens the understanding of enzyme-mediated gas conversion but also demonstrates its industrial application potential. We're setting the stage for further enzyme engineering innovations, contributing to more sustainable industrial practices that align with global environmental objectives.
References:
- Kim SM, Kang SH, Jeon BW, Kim YH. Tunnel engineering of gas-converting enzymes for inhibitor retardation and substrate acceleration. Bioresour. Technol. 394, 130248 (2024). https://doi.org/10.1016/j.biortech.2023.130248
- Kim SM, et al. O2-tolerant CO dehydrogenase via tunnel redesign for the removal of CO from industrial flue gas. Nat. Catal. 5, 807–817 (2022). https://doi.org/10.1038/s41929-022-00834-y
- An enzyme's new 'tunnels' boost its waste-grabbing prowess. Nature 609, 657 (2022). https://doi.org/10.1038/d41586-022-02906-2
- Hwang HW, et al. Two-stage bioconversion of carbon monoxide to biopolymers via formate as an intermediate. Chem. Eng. J. 389, 124394 (2020). https://doi.org/10.1016/j.cej.2020.124394
- Lee J. et al. Molar-scale formate production via enzymatic hydration of industrial off-gases. Nat. Chem. Eng. https://doi.org/10.1038/s44286-024-00063-z (2024).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in