The oncogenic fusion protein DNAJB1-PRKACA can be specifically targeted by peptide-based immunotherapy in fibrolamellar hepatocellular carcinoma
Published in Cancer
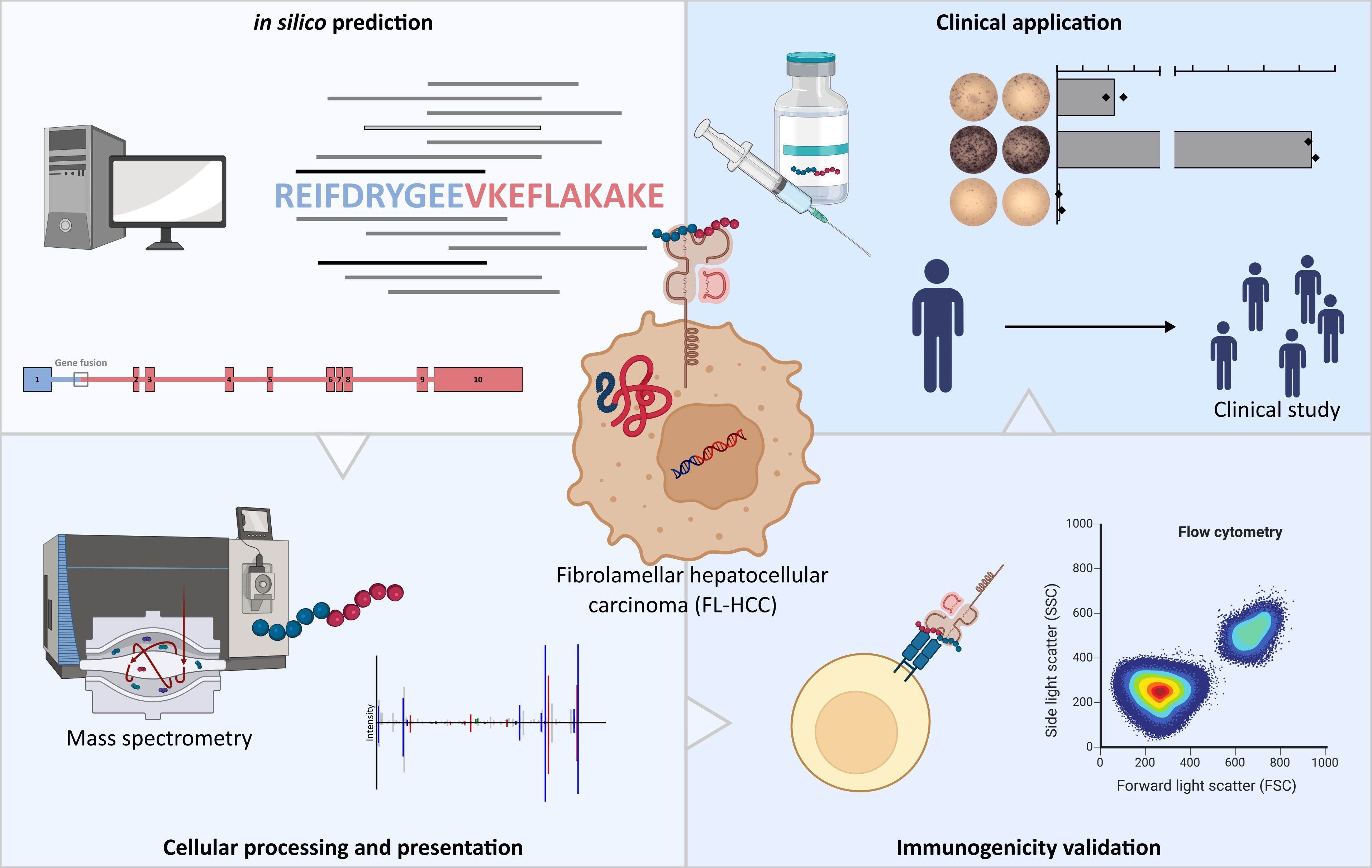
Fibrolamellar hepatocellular carcinoma (FL-HCC) is a lethal liver cancer. Until today, surgical resection is the only effective therapy if the disease is diagnosed before the occurrence of metastases, and long-term survival is jeopardized by tumor recurrence calling for the development of novel specific treatment options for FL-HCC patients. In our work we examined the DNAJB1-PRKACA fusion transcript, which is the oncogenic driver in 100% of FL-HCC patients, as target for the development of novel T-cell based immunotherapies for this devastating tumor disease.
At first we addressed the question if the DNAJB1-PRKACA fusion protein can serve as source for fusion protein specific neoantigens presented as peptides on human leukocyte antigen (HLA) molecules to the immune system. To do this we performed in a first step in silico predictions for potential HLA-ligands derived from the DNAJB1-PRKACA fusion protein. Using a prediction workflow combining the algorithms SYFPEITHI and NetMHCpan we identified nine unique binding cores for a total of 1,290 different HLA class II alleles as well as 13 HLA class I ligands spanning the fusion region of the DNAJB1-PRKACA protein.
To proof the cellular processing and presentation of predicted DNAJB1-PRKACA-derived HLA-ligands we created a DNAJB1-PRKACA fusion protein expression model in hepatocellular carcinoma (HCC) cell lines. We transduced the HCC cell lines HLE, SMMC-7721, and HepG2 with an expression construct that allowed the Doxycycline (Dox)-dependent expression of the DNAJB1-PRKACA fusion protein. HLA presentation was subsequently proven by liquid chromatography-coupled tandem mass spectrometry (LC-MS/MS) of the DNAJB1-PRKACA transduced cell lines as well as in differentiated and maturated monocyte-derived dendritic cells after antigen loading.
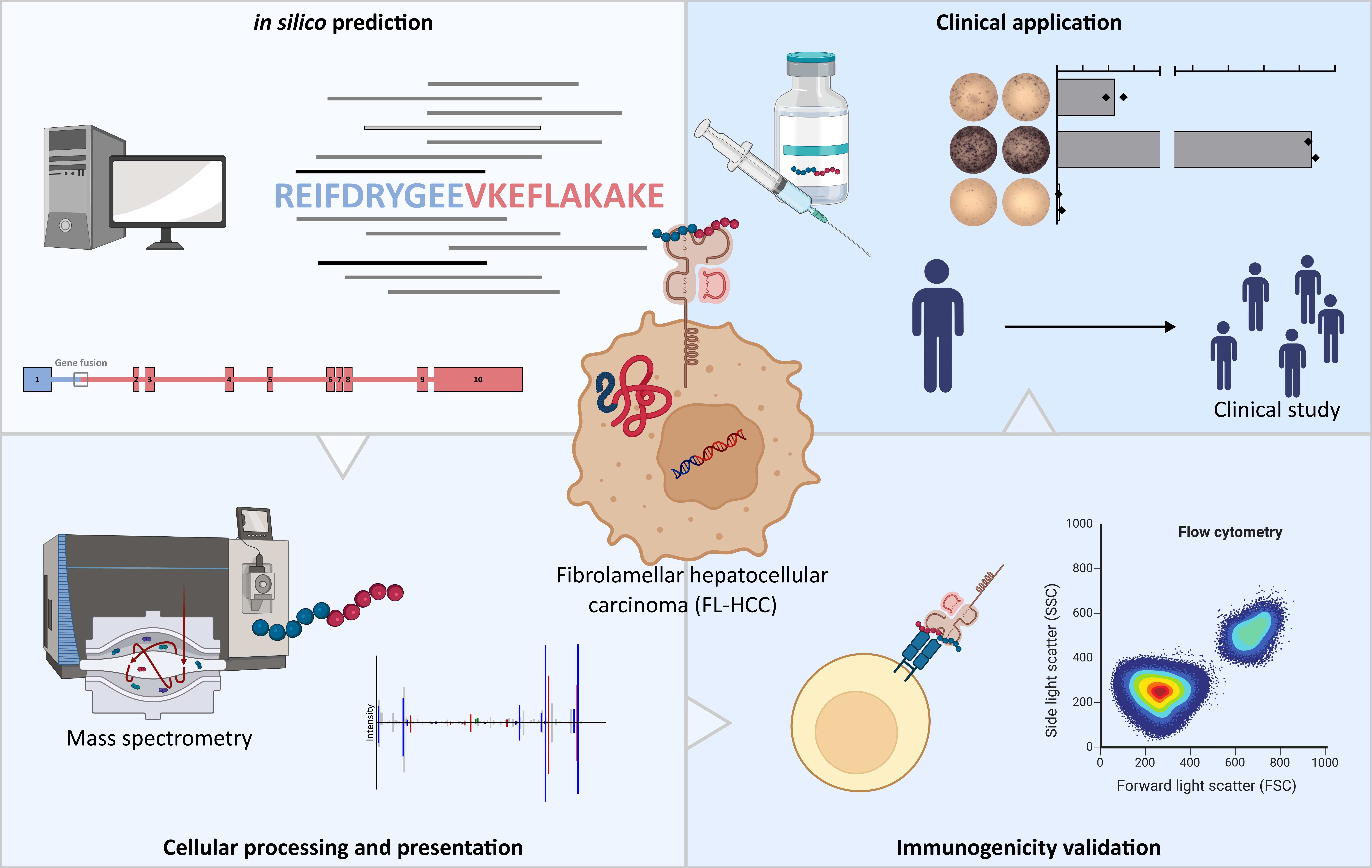
Figure 1. Schematic overview of the four main milestones of DNAJB1-PRKACA-based peptide vaccine development for FL-HCC patients. (Top left panel) In silico prediction of possible HLA-ligands derived from the DNAJB1-PRKACA fusion protein followed by (bottom left panel) the identification of naturally processed and presented HLA-ligands by mass spectrometry, and (bottom right panel)validation of immunogenicity by in vitro T-cell assays. (Top right panel) Finally a DNAJB1-PRKACA-based peptide vaccine was evaluated in a single-FL-HCC patient, which will be followed by an upcoming clinical study in a larger cohort of FL-HCC patients and other tumor entities carrying the DNAJB1-PRKACA fusion transcript.
DNAJB1-PRKACA-derived HLA class I and HLA class II ligands were further tested for immunogenicity by in vitro T-cell assays. De novo priming of CD4+ T cells from healthy volunteers with peptide-loaded mature moDCs induced multifunctional peptide-specific CD4+ T cells, with expression of CD107a and CD154 as well as production of interleukin-2 (IL-2), interferon-γ (IFN-γ), and tumor necrosis factor (TNF). In addition, artificial antigen presenting cells (aAPCs) were used for priming of CD8+ T cells of healthy volunteers to validate the immunogenicity of HLA class I presented peptides. aAPC-based priming induced peptide-specific multifunctional CD8+ T cells reflected by IFN-γ, TNF, and CD107a production/expression. Single-cell RNA sequencing further identified multiple T cell receptors from DNAJB1-PRKACA-specific multifunctional CD8+ T cells.
Finally, we designed a personalized DNAJB1-PRKACA-derived peptide vaccine for a young patient with histologically confirmed unresectable FL-HCC, who suffered from multiple tumor relapses, after initial liver transplantation. The vaccine was applied twice within a 6-week interval and was adjuvanted with the toll-like receptor (TLR) 1/2 agonist XS15 (Pam3Cys-GDPKHPKSF) emulsified in MontanideTM ISA51 VG to endorse activation and maturation of antigen presenting cells and prevent vaccine peptides from immediate degradation. Vaccination induced multifunctional CD4+ T cells, with an activated T-helper 1 phenotype and high T cell receptor clonality. Vaccine-induced DNAJB1-PRKACA-specific T cell responses persisted over time and, in contrast to various previous treatments, were accompanied by durable relapse free survival of the patient for more than 21 months post vaccination.
Our preclinical and clinical findings identify the DNAJB1-PRKACA fusion protein as source for immunogenic neoepitopes and corresponding T cell receptors and provide efficacy in a single-patient study of peptide-based immunotherapy specifically targeting this oncogenic fusion. These findings will further be followed in a upcoming clinical trial evaluating the here defined DNAJB1-PRKACA neoepitopes adjuvanted with the TLR1/2 agonist XS15 emulsified in MontanideTM ISA51 VG in combination with the PD-L1 antibody atezolizumab in a Phase I vaccine study, recruiting patients with various malignant disease expressing the DNAJB1-PRKACA fusion transcript.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in