The Serology Puzzle: Untangling Cross-Reactivity in Arbovirus Diagnostics
Published in Bioengineering & Biotechnology, Microbiology, and Biomedical Research
In regions where multiple arboviruses such as dengue, Zika, and chikungunya co-circulate, diagnostic clarity is a luxury. Cross-reactivity, where antibodies against one virus bind to another, has long hampered our ability to identify the true causal agent of fever.
The project began in 2021 with the simple idea of creating a multi-antigen panel for arbovirus serology using a bead-based multiplex immunoassay (MIA) on the Luminex platform. We adapted our in-house methods for malaria and SARS-CoV-2 serology studies and added antigens reported in the arbovirus literature.
First challenge: Search for gold standards
To verify our panel, we needed "gold standard" antibody reagents. Since these aren’t commercially available for many targets, we examined antibody responses in samples of individuals in cross-sectional surveys from dengue-endemic sites in Senegal, New Caledonia, Peru, and French Guiana to find hyperimmune individuals whose serum could serve as positive controls. However, finding samples from individuals with a confirmed history of viral exposure was a hurdle. To overcome this, we travelled to Senegal to collaborate with our partners at the Institut Pasteur de Dakar by applying our assay to samples with confirmed arbovirus infection.
One challenge after another
At the end of 2022, we had tested most of the study samples and realised we had hit a wall. Our data showed that antibody responses were highly correlated; individuals were testing positive for proteins across similar viral families. It became clear that simply expanding our antigen panel wasn't enough to distinguish a true infection from a cross-reactive "echo." We needed a way to filter the signal from the noise.
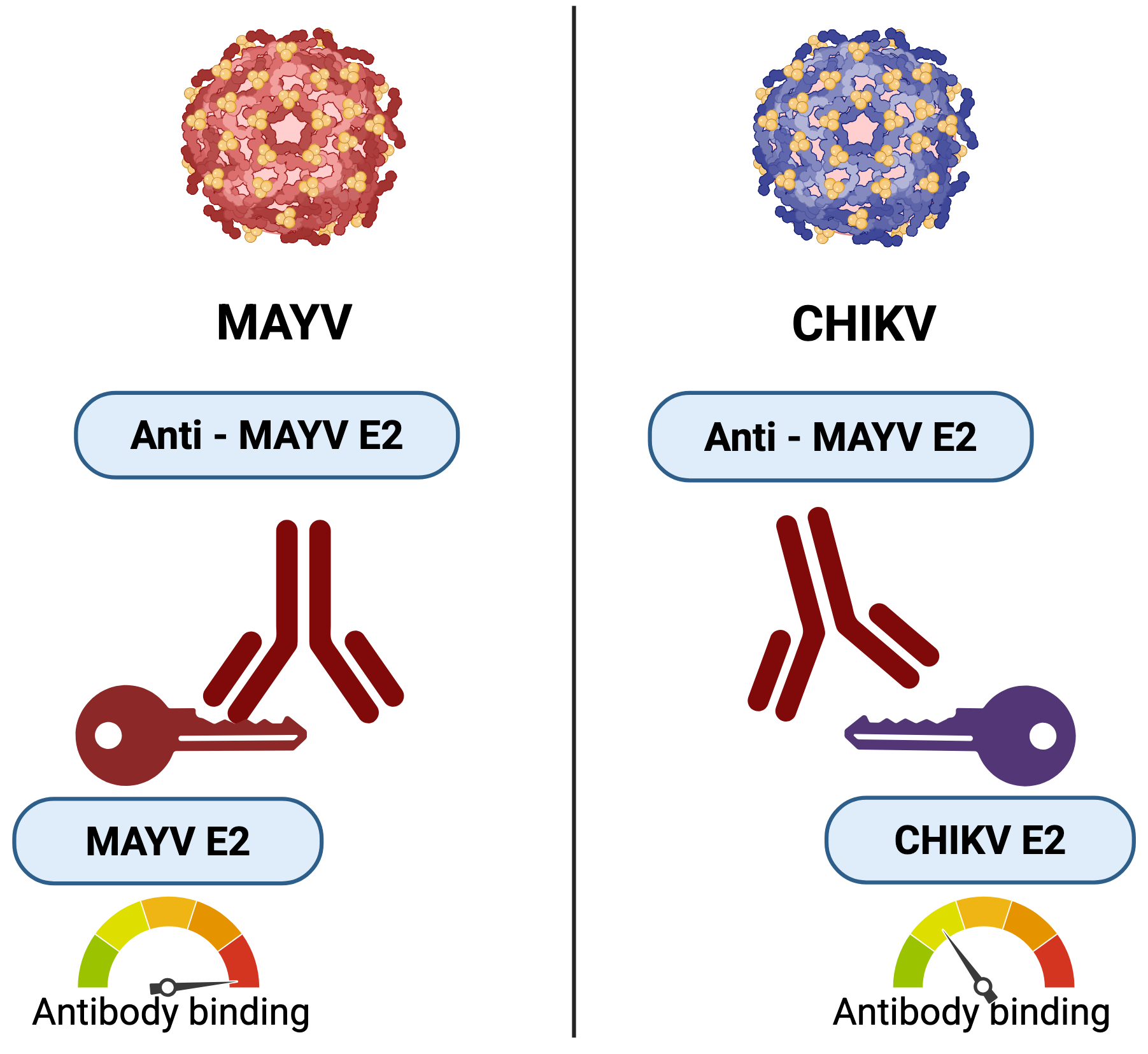
The Cross-Reactivity Dilemma: In regions where similar viruses, such as Mayaro (MAYV) and Chikungunya (CHIKV), co-circulate, standard antibody tests (ELISA or RDTs) often struggle to distinguish them. Antibodies against MAYV E2 antigen (shown as red keys) can cross-react and bind to CHIKV E2 antigen (purple key). While true, specific binding is stronger (high-scale reading), this "noisy" cross-reactive signal makes it impossible to define positive samples without more advanced methods.
Statistical models brought clarity
In collaboration with colleagues working on serological modelling, we brainstormed ways to define seropositivity thresholds. We turned to Bayesian finite mixture models to move beyond arbitrary "one-size-fits-all" thresholds. This allowed us to define seropositivity cut-offs that accounted for the specific epidemiological context of our study populations, giving us a more nuanced view of the data.
To truly distinguish between the two viruses, we used an in-house competitive assay to strip away cross-reacting antibodies. By pre-incubating samples with specific 'competitors,' we could see which antibodies remained active in the supernatant. We were encouraged to find that, in some cases, individuals who initially appeared to have been infected with both chikungunya and the closely related Mayaro virus had been infected only with Mayaro and had developed cross‑reactive antibodies that resembled those induced by chikungunya virus infection. had developed cross‑reactive antibodies that resembled those induced by chikungunya virus infection.
We were happy to see that, by combining both competitive assays and statistical models, we were able to disentangle cross-reactivity from true exposure and define seropositivity.
Reference
Yman, V., Rosado, J., Ochida, N. et al. Investigating antibody cross-reactivity and transmission dynamics of alphaviruses and flaviviruses using a multiplex serological assay. Nat Commun 17, 3491 (2026). https://doi.org/10.1038/s41467-026-71451-7
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in