Understanding the photogenerated hole traps in metal-organic-framework-based photocatalysts
Published in Chemistry
Photocatalytic water splitting for hydrogen evolution has been considered a promising approach to alleviate the current energy and environmental issues. Since Fujishima and Honda first reported solar energy conversion for hydrogen evolution reactions (HER) using semiconductor-based photocatalysts in 19721, there are many works related to this topic. In general, the primary drawback using a pure semiconductor was the fast recombination of photoinduced carriers, which seriously limited photocatalytic efficiency2,3. Thus, the design of the heterostructures with individual functionality of each nanomaterial has been urgency to improve the efficiency of photocatalytic activity.
Recently, metal-organic frameworks (MOFs) are porous crystalline materials with high specific surface area, tunable structure and multifunctionality. Therefore, MOFs are widely used in various fields, including photocatalysis, photodetector, electrolysis, and photovoltaics. Unfortunately, most of MOFs have low ability of photoresponse in the visible region, which largely hinders their application in solar energy conversion4,5. Furthermore, the investigation of photo-generated carrier dynamics in MOFs have been the key to understand the photocatalytic mechanism. In addition, the photoinduced electron dynamics in MOFs have been well investigated, but the kinetics of photogenerated holes and their effects on photocatalytic activity remain poorly understood. Generally, cadmium sulfide (CdS) has been the preferred visible-light photocatalyst for HER due to its suitable band structures. However, photoinduced corrosion and fast recombination of photoinduced electron−hole pairs have severely restricted improvements in its photocatalytic activity. Therefore, it is necessary to enhance the photocatalytic activity by fine-tuning the photo-induced carrier dynamics.
Here, we synthesized a spatial charge structure for the MOF-based photocatalyst (Figure 1). First, we encapsulate the Pt nanoparticles (NPs) into NH2-UiO-66, which not only shortened the migration path of photogenerated electrons, but also prevented the aggregation and leaching of the Pt NPs during the reaction6. Then, CdS was grown on the outer surface of NH2-UiO-66. Particularly, NH2-UiO-66 has a large specific surface area, which can promote the dispersion of CdS on the NH2-UiO-66 surface, thereby providing more adsorption sites and photocatalytic reaction centers7.
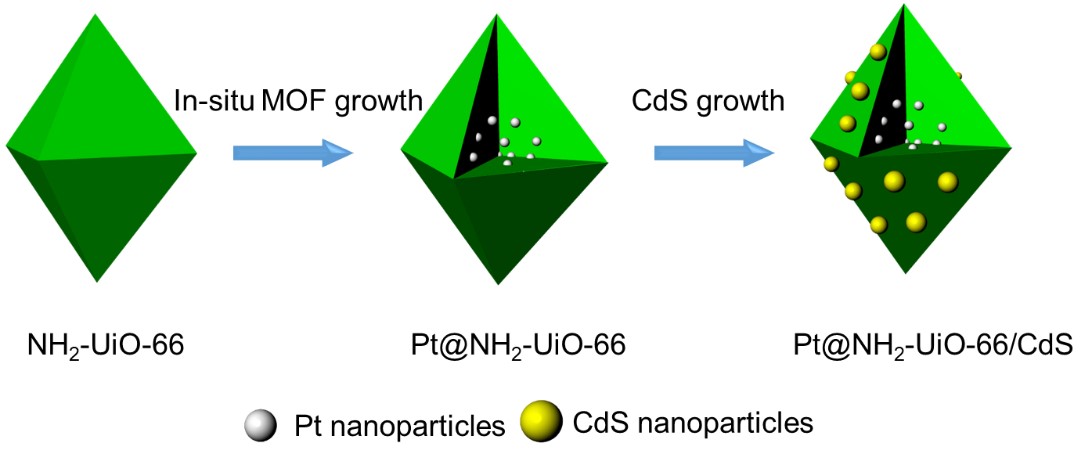
Figure 1. Illustrated scheme of synthetic procedures. Synthetic processes of Pt@NH2-UiO-66/CdS composites.
Through the testing of the photocatalytic activity in HER, we are excited to find that the photocatalytic HER rate of the Pt@NH2-UiO-66/CdS under visible light irradiation is 37.76 mmol h-1 g-1 (Figure 2), which is 3432 and 92 times that of NH2-UiO-66 (0.011 mmol h-1 g-1) and CdS (0.41 mmol h-1 g-1), respectively. The quantum efficiency of Pt@NH2-UiO-66/CdS at 400 nm is as high as 40.3%, which is the highest among MOF-based photocatalysts so far.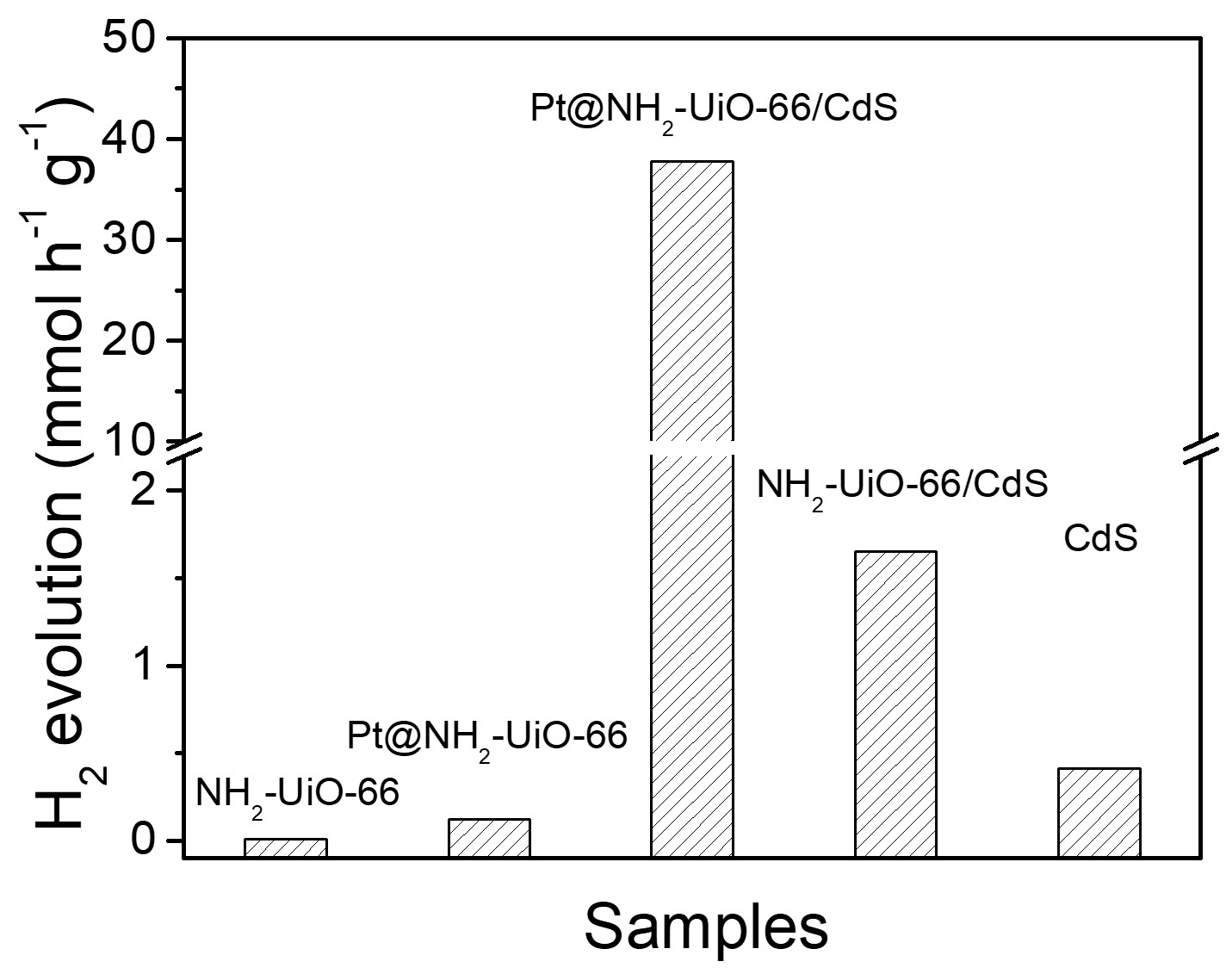
Figure 2. Photocatalytic hydrogen evolution. Comparison of the hydrogen evolution rate for NH2-UiO-66, Pt@NH2-UiO-66, Pt@NH2-UiO-66/CdS, NH2-UiO-66/CdS and CdS NPs under visible-light.
To study the transfer of photogenerated carriers in the photocatalytic process, we first performed the experiments of the Mott-Schottky curves of the samples and determined the positions of the conduction and valence bands of CdS and NH2-UiO-66. Then a hypothesis for the photocatalytic HER mechanism of the sample Pt@NH2-UiO-66/CdS was proposed. To confirm our hypothesis, we tracked photoinduced carrier dynamics by time-resolved transient absorption measurements to study electron and hole transfer. By performing the transient absorption experiments, we found that the high activity and stability of Pt@NH2-UiO-66/CdS are attributed to the spatial charge separation following an efficient photogenerated hole transfer band-well pathway (Figure 3). When visible light irradiated the Pt@NH2-UiO-66/CdS, both semiconductor phases were excited, the electron from the conduction band of CdS was transferred to the conduction band of NH2-UiO-66 through the electron trapped pathway. Then it was transferred to the Pt surface for HER under visible light irradiation. The photoinduced hole in NH2-UiO-66 was extracted from the trapped state and transferred to the CdS valence band. Then it migrated to the surface, where it was oxidized by the sacrificial reagent (lactic acid).
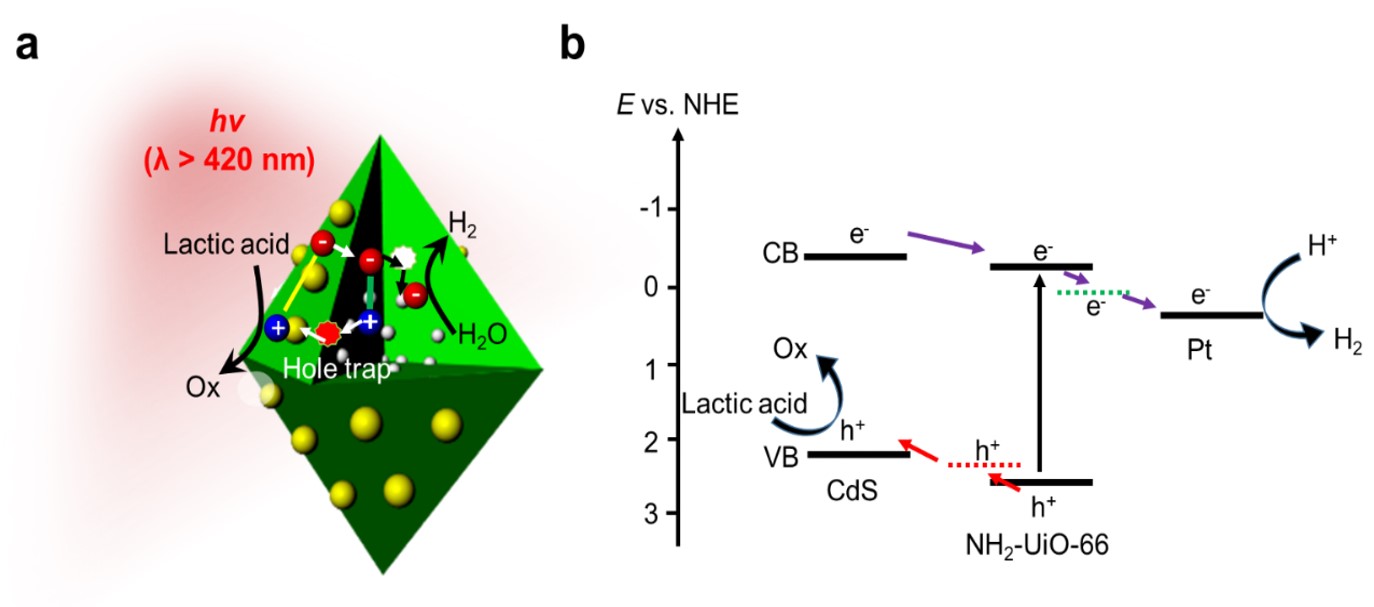
Figure 3. Mechanism of photocatalytic HER. a, Mechanism of photoinduced carrier dynamics for Pt@NH2-UiO-66/CdS, with a hole-trap transfer pathway for enhanced HER photocatalytic activity. b, Band diagram of photocatalytic processes in Pt@NH2-UiO-66/CdS.
Finally, the Pt@NH2-UiO-66/CdS photocatalyst separated photogenerated electrons and holes by combining Pt NPs and CdS NPs, which greatly prolonged the lifetime of the hole-trap-mediated pathway and improved the HER photocatalytic efficiency. This work provides a deeper understanding of electron and hole transfer in co-catalyst-NH2-UiO-66-semiconductor ternary composites with spatial-separation structures. We expect this work can provide more inspiration for scientific researchers, especially in the fields of materials and photocatalysis, and promote the development of efficient photocatalytic composites. We will do more extensive research in the field of photocatalysis, such as photocatalytic water splitting, CO2 reduction, etc. We hope that more scientific researchers can understand and join us to contribute to the strategies for solving the energy and environmental problems.
For more details, please read our recent publication in Communications Chemistry:
Lian, Z. et al. Photogenerated hole traps in metal-organic-framework photocatalysts for visible-light-driven hydrogen evolution. Commun Chem 5, 93 (2022).
https://www.nature.com/articles/s42004-022-00713-4.
References
- Fujishima, A. & Honda, K. Electrochemical photolysis of water at a semiconductor electrode. Nature 238, 37–38 (1972).
- Yan, H. et al. Visible-light-driven hydrogen production with extremely high quantum efficiency on Pt–PdS/CdS photocatalyst. J. Catal. 266, 165–168 (2009).
- Li, J., Yang, J., Wen, F. & Li, C. A visible-light-driven transfer hydrogenation on CdS nanoparticles combined with iridium complexes. Chem. Commun. 47, 7080–7082 (2011).
- Su, Y., Zhang, Z., Liu, H. & Wang, Y. Cd2Zn0.8S@UiO-66-NH2 nanocomposites as efficient and stable visible-light-driven photocatalyst for H2 evolution and CO2 reduction. Appl. Catal. B: Environ. 200, 448–457 (2017).
- Yang, Q., Xu, Q. & Jiang, H.-L. Metal–organic frameworks meet metal nanoparticles: synergistic effect for enhanced catalysis. Chem. Soc. Rev. 46, 4774–4808 (2017).
- Xiao, J.-D. et al. Boosting photocatalytic hydrogen production of a metal–organic framework decorated with platinum nanoparticles: the platinum location matters. Angew. Chem. Int. Ed. 55, 9389–9393 (2016).
- Xu, H.-Q. et al. Unveiling charge-separation dynamics in CdS/metal−organic framework composites for enhanced photocatalysis. ACS Catal. 8, 11615–11621 (2018).
Follow the Topic
-
Communications Chemistry
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Jun 30, 2026
Experimental and computational methodology in structural biology
Publishing Model: Open Access
Deadline: Jul 31, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in