Unveiling Pseudomonas aeruginosa's Hidden Arsenal of Quinolones
Published in Chemistry
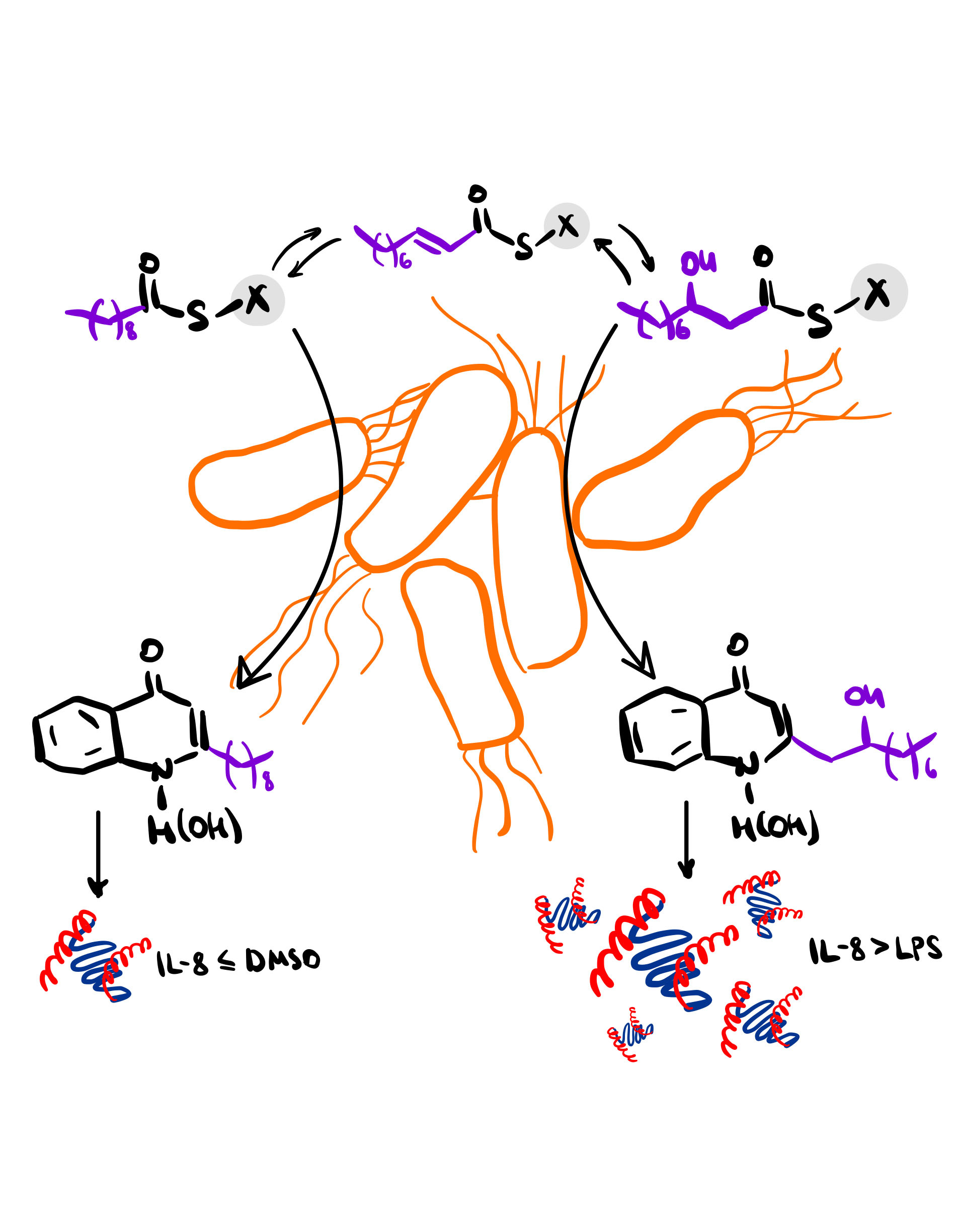
The research group of Prof. Dr. Thomas Böttcher at the Faculty of Chemistry, University of Vienna, is dedicated to studying the chemistry of microbial interactions, exploiting their chemical properties, and employing synthetic chemistry techniques to manipulate and control microbial behavior (JACS 2021, 143, 8344; Chem. Sci. 2021, 12, 6908; ACS Cent. Sci. 2020, 6, 241; Chem. Sci. 2019, 10, 6624; JACS 2018, 140, 14019; ACIE 2017, 56, 7271; Cell Chem. Biol. 2017, 24, 598). They particularly focus on pathogenic bacteria, such as Pseudomonas aeruginosa, which possess a diverse array of small molecules with functions ranging from virulence factors and immune modulators to siderophores, quorum sensing signals, and antibiotics. Among these secondary metabolites produced by P. aeruginosa, two closely related classes known as 2-alkyl-4(1H)-quinolones (AQs) and 2-alkyl-4(1H)-quinolone N-oxides (AQNOs), have attracted significant interest. Although both classes share a common structural core, the N-oxidation of AQNOs leads to a distinct and profound alteration in their bioactivity. While AQs, such as 2-heptyl-4-hydroxyquinoline (HHQ), primarily function as quorum sensing molecules for P. aeruginosa, AQNOs exhibit highly structure-dependent and species-specific antimicrobial activities, particularly against gram-positive bacteria (Chem. Commun. 2020, 56, 6328; ACIE 2017, 56, 7271). The realm of structural diversity within AQs and AQNOs is extensive, yet not all their variations, biological activity, as well as their physiological and medicinal significance, have been thoroughly investigated. Considering the observation that the structural diversity of AQs and AQNOs arises from the flexible incorporation of fatty acids by the PqsB/C complex during biosynthesis, our research aimed to predict novel classes of AQ and AQNO compounds.
In our recent publication titled "Biosynthetic flexibility of Pseudomonas aeruginosa leads to hydroxylated 2-alkylquinolones with proinflammatory host response" published in Communications Chemistry, we embarked on investigating whether P. aeruginosa synthesizes previously unreported quinolone classes, namely 2'-hydroxy and 2'-oxo-substituted quinolones and quinolone N-oxides, by incorporating the corresponding beta-hydroxy and beta-keto fatty acids derived from the fatty acid metabolism.
The identification and quantification of potential 2'-hydroxy (2'-OH) and 2'-oxo compounds among the complex mixture of AQs and AQNOs produced by P. aeruginosa present challenges due to their structural similarity, resulting in similar polarity and identical masses compared to other AQs and AQNOs. While our in-house synthetic standard library and MS2 database have previously facilitated the detailed identification and quantification of quinolones from Burkholderia species (Chem. Commun. 2020, 56, 6328), these resources lacked standards for the newly proposed AQs and AQNOs classes, and no synthetic strategies were available for these highly functionalized quinolones. Existing literature on synthetic strategies for quinolone synthesis proved unsuitable for our target molecules, as they often required harsh reaction conditions such as high temperatures or extreme pH values, particularly for the cyclization of the quinolone ring (key review: Arkivoc 2021, ix, 218).
Hence, we initiated our investigations by developing a modified and unconventional approach for synthesizing quinolones, employing a catalytic reductive cyclization method that could be conducted at room temperature and neutral pH. Throughout our synthetic endeavors, we developed protocols to precisely control the degree of reduction by utilizing different catalysts and reaction times, leading to a versatile method for synthesizing either AQs or AQNOs from the same initial substrate.
By utilizing these standards for our desired molecules, we successfully confirmed the production of 2'-OH-NQ and 2'-OH-NQNO by P. aeruginosa. Surprisingly, we found that 2'-OH-NQ was present in culture supernatants at remarkably high concentrations comparable to NQ. Moreover, the addition of exogenous β-hydroxydecanoic acid further enhanced the production of 2'-OH-NQ, confirming the origin of the hydroxylated substrate from the fatty acid metabolism. In contrast to NQ, 2'-OH-NQ exhibited potent induction of the cytokine IL-8 in a human cell line even at a concentration as low as 100 nM, suggesting its potential involvement in modulating the host immune response. In patients with cystic fibrosis, ongoing inflammation in the lungs is influenced by IL-8, a cytokine that is known to be triggered by P. aeruginosa infections. Therefore, our recent discovery of hydroxylated quinolone derivatives with immune-modulating properties may have unveiled one of the metabolites responsible for driving the interactions between microbes and the human host.
More details of this work can be found here: “Biosynthetic flexibility of Pseudomonas aeruginosa leads to hydroxylated 2-alkylquinolones with proinflammatory host response” in Communications Chemistry. https://doi.org/10.1038/s42004-023-00937-y
Follow the Topic
-
Communications Chemistry
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Jun 30, 2026
Sustainable waste management through polymer upcycling
Publishing Model: Open Access
Deadline: May 31, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in