You can find our paper in Nature Catalysis here: https://www.nature.com/articles/s41929-018-0085-6
“What about stability?” - A question, increasingly emerging at conferences or in peer review of scientific works in the field of electrocatalysis. Needless to say, the kinetic hindrance of many important reactions is still one of the major bottlenecks towards efficient conversion of energy from renewable sources. In recent years, this fact has led to increased interest towards this topic, resulting in an abundance of publications reporting on innovative catalyst materials for electron transfer reactions. Ideally, the catalyst should not be consumed in these processes; yet, reality shows that harsh reaction conditions or side reactions in the catalytic mechanism can cause severe degradation.
The community is aware of these issues, however, often does not hold the right means for effective stability evaluation and benchmarking. In addition to the obvious material properties, several parameters like catalyst loading, duration of the measurement, applied potential/current, catalyst size and porosity, or support material interaction do influence stability significantly. Moreover, degradation can proceed over various mechanisms that affect the catalyst structure, composition and electronic properties, which complicate the picture drastically. Hence, our question - “What about stability”- is difficult to be answered precisely. Even more so, as stability is a “long-term characteristic”, its thorough evaluation hampers fast breakthroughs in catalyst development.
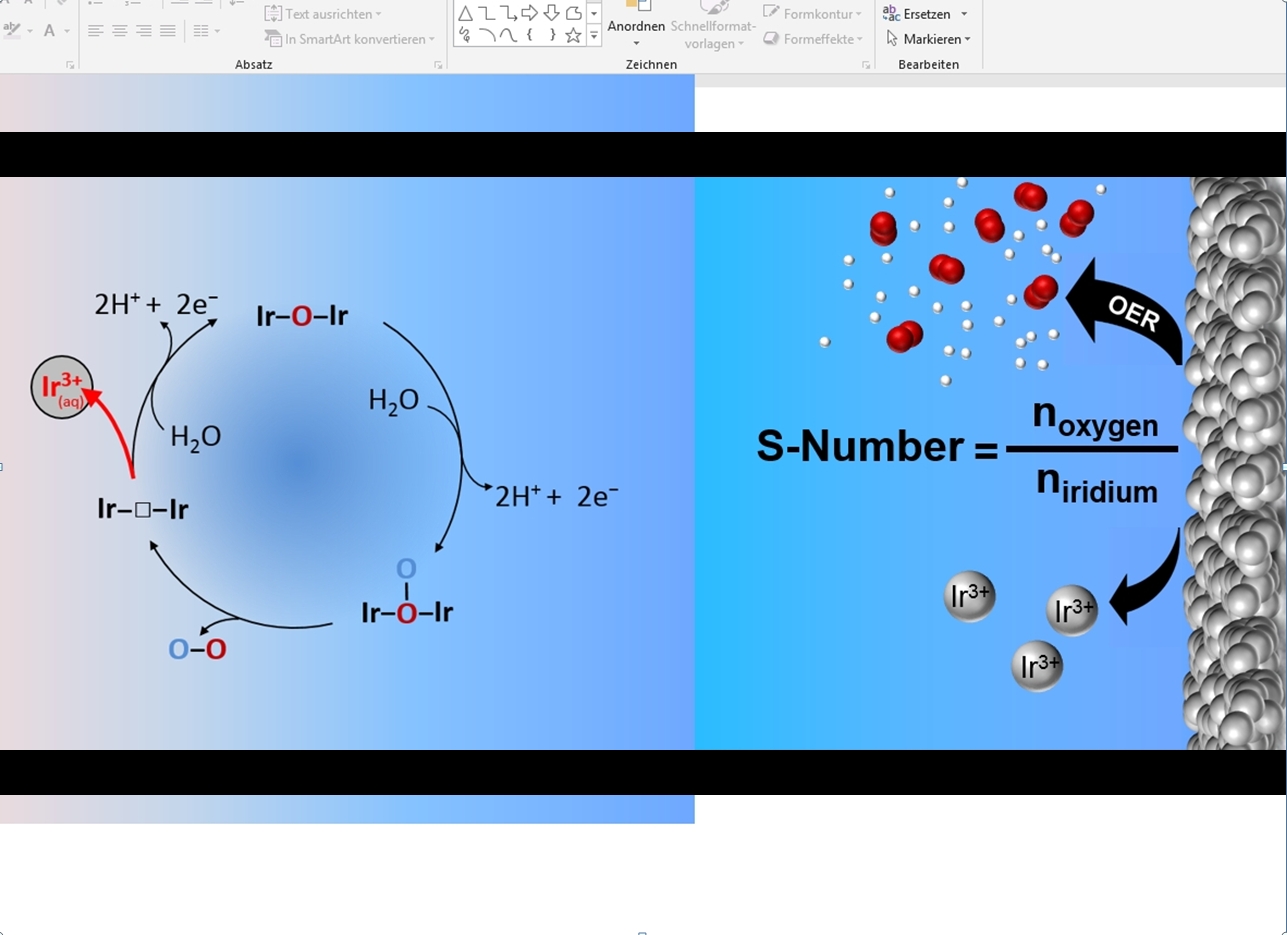
To overcome this issue particularly for degradation by dissolution of elements, we propose by the example of the oxygen evolution reaction (OER) to normalize the flux of produced molecules by the flux of dissolved catalyst atoms. We refer to this metric as the Stability-number (S-number). The S-number in principle describes how many molecules are evolved before one catalyst atom dissolves into the electrolyte. In this manner, it is similar to the well-established turn over number, with the major advantage that the amount of active sites does not need to be known. Moreover, it can be assessed very quickly and in a time-resolved manner, so that combined with high-throughput capabilities rapid screening of material libraries becomes possible. For our work we used some of the most prominent and highly discussed modifications of iridium oxide to demonstrate the S-number as a useful benchmarking tool for OER-catalysts, and even succeeded to identify fundamental differences in between material classes. Along the same line, other electrocatalytic reactions that suffer from dissolution will greatly benefit from this metric as well (e.g. chlorine evolution).
Overall, our research facilitates communication between research groups regarding their achievements in catalyst stability, which is particularly important for the development of efficient electrochemical energy conversion devices. The S-number is an easy accessible metric for benchmarking and provides important quantitative data for insights into dissolution mechanism, both important factors for future catalyst design.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in