After the Paper: How and Where HIV Hides
Published in Microbiology
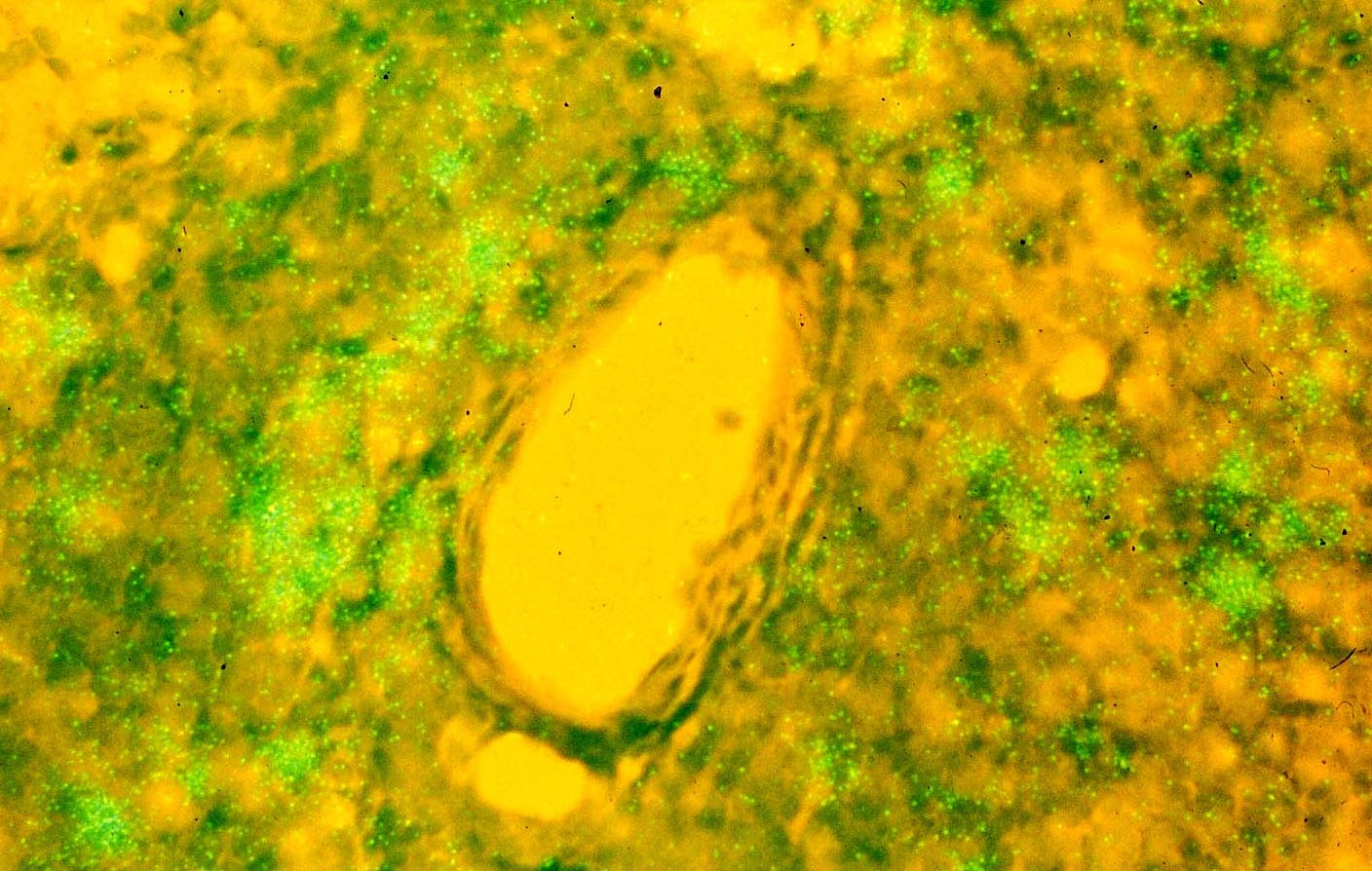
I have always been fascinated by the central question of how lentiviruses elude host defenses, and, now in the case of the human lentivirus, HIV, the mechanisms by which the virus persists despite currently effective anti-retroviral therapy (ART). In our early studies of the prototypic lentivirus and distant relative of HIV, visna virus, we thought that the answer was that visna virus escaped the immune response in infected sheep by “hiding” in cells in a quiescent state, analogous to bacteriophage lysogeny, but on a grand scale, in an animal (1-3). So when Simon Wain Hobson linked HIV to visna and the ungulate lentiviruses (4), we set out to see if HIV persisted in this way.
But, where and how to look? The where seemed obvious-most of the then known CD4+ T cell and macrophage targets would be concentrated in lymphoid tissues-and the how to search for 10 attomoles of proviral DNA in single cells became possible with the development of in situ PCR (5). Armed with the technology and lymphoid tissue samples, I was astonished to see one night how many DNA+ cells there were in our lymphoid tissue samples, and could only find the floor’s janitor to share the discovery. We published the story in Nature in back-to back papers with Tony Fauci’s lab (6, 7), both papers confirming earlier work by Paul Racz that the lymphoid tissues were the principal sites of HIV infection (8). We emphasized covert (latent) infection in our paper, while Pantaleo et al. reached the opposite conclusion. HIV infection was always active. This Janus-faced pairing is actually a recurrent theme in lentivirus infections (9) from Visna to HIV-silent infection to hide and persist, and active infection to maintain and spread infection and indirectly cause disease from immunopathological responses to active infection.
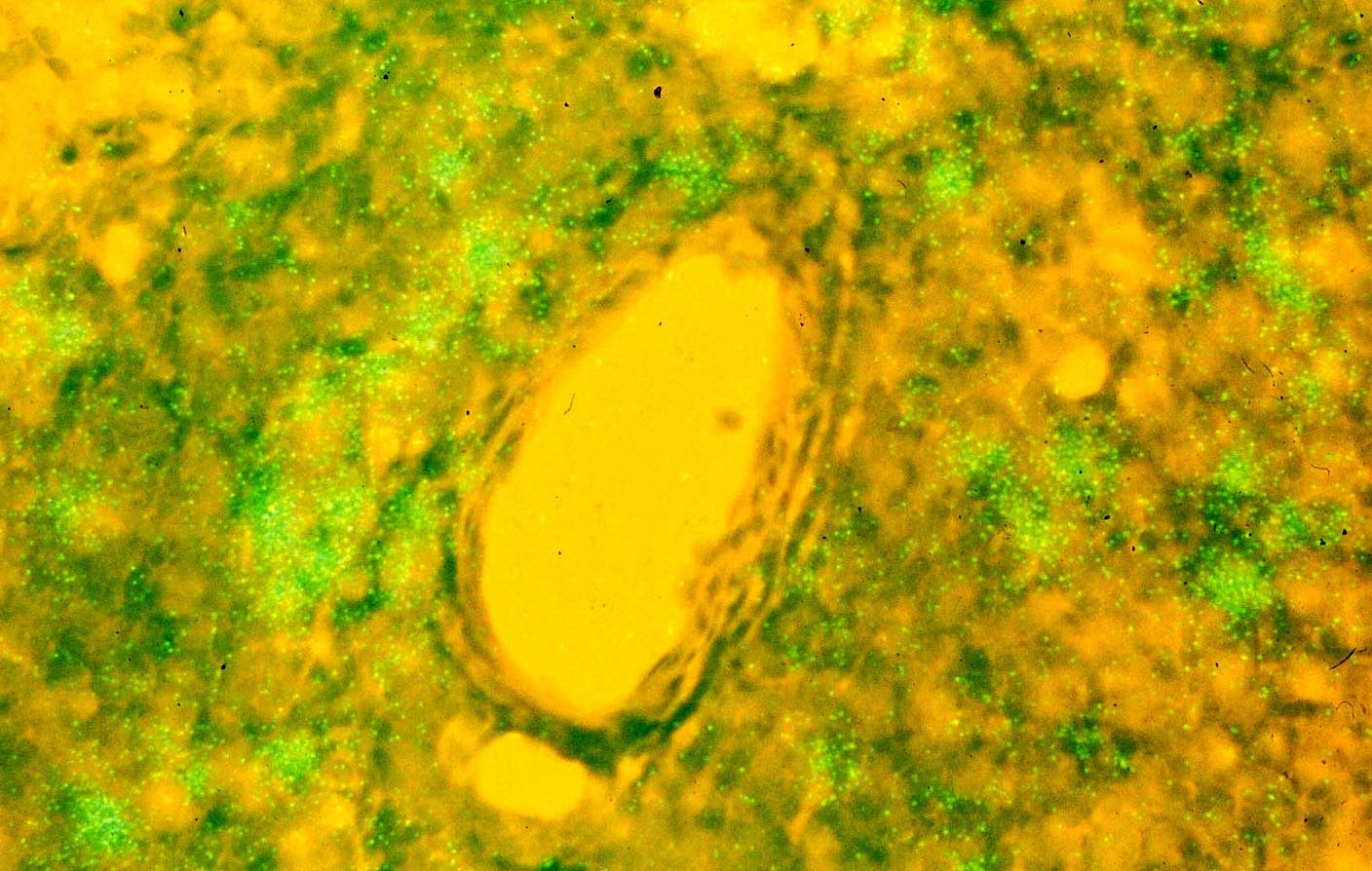
The two papers caused a stir at the time. John Maddox, then Nature’s editor, and Howard Temin and Dani Bolognesi wrote Commentaries (10, 11). All agreed that we must now think of lymphoid tissues as the reservoir where HIV had been hidden and was now fully revealed. Maddox focused on the implications for refuting Peter Duesberg’s “HIV-hypothesis” that HIV was not the cause of AIDS, while the Temin/ Bolognisi prescient piece anchored the findings to the likelihood in any retrovirus infection of defective proviruses accounting for a substantial portion of transcriptionally silent cells.
The advent of effective ART and discovery of HIV dynamics by the Ho and Shaw labs (12, 13) quickly shifted attention to the active nature of HIV infection, only to be soon followed by the diametrically opposed theme of HIV latent infection by Siliciano (14) and others. Fast forward to the present and the quest for curing HIV infections. Lymphoid tissues are the principal reservoirs in HIV and SIV infections (15), but these reservoirs harbor populations of largely defective proviruses (16). So, how are we to count the replication competent proviruses from which infection rebounds when ART is interrupted? And, can we discount immune and drug sanctuaries in lymphoid tissues where active infection continues (17, 18) as a source of virus for rebound and stimulus for immune activation and its associated contribution to morbidity and mortality (19)? Engaging questions, a quarter century after the initial discovery of HIV’s two faces in lymphoid tissues.
1. Haase, AT. The slow infection caused by visna virus. Curr. Top. Microbiol. Immunol. 72,101-156 (1975).
2. Haase, AT, Stowring, LS, Narayan,O, Griffin, D and Price, D. Slow persistent infection caused by visna virus: Role of host restriction. Science 195: 175-177 (1977).
3. Brahic, M, Stowring, L, Ventura, P and Haase, AT. Gene expression in visna virus infection in sheep. Nature 292, 240-242 (1981).
4. Sonigo, P, Alizon, M, Staskus, K, Klatzmann, D S, Cole, S O Danos, O E, Retzel, E, Tiollais, EP, Haase,AT and Wain-Hobson, S. Nucleotide sequence of the visna lentivirus: Relationship to the AIDS virus. Cell 42, 369-382 (1985).
5. Haase, AT, Retzel, E, and Staskus, K. Amplification and detection of lentiviral DNA inside cells. Proc. Natl. Acad. Sci. USA 87, 4971-4975 (1990).
6. Embretson, J, Zupancic, M, Ribas, JL, Burke, A, Racz, P, Tenner-Racz, K, and Haase, AT. Massive covert infection of helper T lymphocytes and macrophages by human immunodeficiency virus during the incubation period of AIDS. Nature 362: 359-362 (1993).
7. HIV infection is active and progressive in lymphoid tissue during the clinically latent stage of disease. Pantaleo, G, Graziosi,G, Demarest, JF, Butinit, L, Montronit, M, Fox, CH, Orenstein, JM, . Kotlerll,DP and Fauci, AS. Nature 362: 355-359 (1993).
8. Racz. P. Prog Allergy 37, 81-181 (1986).
9. Haase, AT. Pathogenesis of lentivirus infections. Nature 322: 130-136 (1986).
10. Maddox, J. Where the AIDS virus hides away. Nature 362, 287 (1993).
11. Where has HIV been hiding. Temin, H M and Bolognesi, DP. Nature 362, 292-293 (1993).
12. Ho DD, Neumann AU, Perelson AS, Chen W, Leonard JM, Markowitz M. Rapid turnover of plasma virions and CD4 lymphocytes in HIV-1 infection. Nature 373,123-6 (1995).
13. Wei, X., Ghosh, S., Taylor, M. et al. Viral dynamics in human immunodeficiency virus type 1 infection. Nature 373, 117–122 (1995).
14. Finzi, D, Hermankova, M, Pierson, T, Carruth, LM, Buck, C, Chaisson, RE, Quinn, TC, Chadwick, K, Margolick, J R, Brookmeyer, JR, Gallant, J, M, Markowitz, M, Ho, DD, Richman, D, and Siliciano, RF. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science 278, 1295–1300 (1997).
15. Estes JD, Kityo C, Ssali F, Swainson L, Makamdop KN, Del Prete GQ, Deeks SG, Luciw PA, Chipman JG, Beilman GJ, Hoskuldsson T, Khoruts A, Anderson J, Deleage C, Jasurda J, Schmidt TE, Hafertepe M, Callisto SP, Pearson H, Reimann T, Schuster J, Schoephoerster J, Southern P, Perkey K, Shang L, Wietgrefe SW, Fletcher CV, Lifson JD, Douek DC, McCune JM, Haase AT, Schacker TW. Defining total-body AIDS-virus burden with implications for curative strategies. Nat Med. 2017. Epub 2017/10/03.
16. Capoferri, AA, Lai, J, Strain, MC, Lada, SM, Hoh, R, Y.-C. Ho, Y.-C, Richman, DD, S. G. Deeks, SG, Siliciano, JD, Siliciano, RF Defective proviruses rapidly accumulate during acute HIV-1 infection. Nat. Med. 22, 1043–1049 (2016).
17. Fletcher, CV, Staskus, K, Wietgrefe, S, Rothenberger, M, Reilly, C, Chipman, J, Beilman, G, Khoruts, A, Thorkelson, A, Schmidt, T, Anderson, J, Perkey, Stevenson, M, Perelson, AS, Douek, DC, Haase, AT, Schacker. TW. Persistent HIV-1 replication is associated with lower antiretroviral drug concentrations in lymphatic tissues. Proc. Natl. Acad. Sci. USA. 111, 2307-2312 (2014).
18. Connick E, Mattila T, Folkvord JM, Schlichtemeier R, Meditz AL, Ray MG, et al. CTL fail to accumulate at sites of HIV-1 replication in lymphoid tissue. J Immunol. 178, 6975–83 (2007).
19. Kuller LH, Tracy R, Belloso W, De Wit S, Drummond F, Lane HC, Ledergerber B, Lundgren J, Neuhaus J, Nixon D, Paton NI, Neaton JD. Inflammatory and coagulation biomarkers and mortality in patients with HIV infection. PLoS medicine 5,e203 (2008).
Follow the Topic
-
Nature
A weekly international journal publishing the finest peer-reviewed research in all fields of science and technology on the basis of its originality, importance, interdisciplinary interest, timeliness, accessibility, elegance and surprising conclusions.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in