ATP, often hailed as the cellular powerhouse, has captivated scientists for decades with its pivotal role in energizing essential cellular processes. It drives molecular transport, powers muscle contractions, and facilitates intricate biochemical reactions, such as DNA and protein synthesis. Beyond its energy-transfer prowess, ATP's enigmatic abundance within cells, typically ranging from 2 to 12 millimolars, has intrigued researchers (1).
Recent scientific revelations have shattered the limits of ATP's influence, revealing it as a multifaceted regulator of protein homeostasis. One remarkable role involves ATP's dissolution of protein liquid-liquid phase separation (LLPS) and amyloidosis associated with diseases like Amyotrophic Lateral Sclerosis (ALS) (2). ATP's interaction with amino acids like Arginine and Lysine exerts nuanced control over LLPS of intrinsically-disordered regions (IDRs), including key proteins like FUS, TDP-43, and the SARS-CoV-2 N protein (3-6). Surprisingly, ATP also counters the destabilizing effects on folded proteins by cellular crowding, a phenomenon linked to cataract formation (7,8).
Even more astonishingly, recent studies have unveiled ATP's remarkable ability to induce protein folding, making it the most efficient folding inducer known so far. This is exemplified in its role in folding the C71G mutant of human profilin-1 (C71G-hPFN1) and nascent Cu/Zn-Superoxide Dismutase 1 (hSOD1), both implicated in ALS (9). ATP's power to induce folding traces back to its triphosphate moiety, but free triphosphate has an adverse ability to trigger aggregation. What's truly marvellous is that ATP and triphosphate seem to enhance protein folding by interacting with the most mysterious molecule to both science and religion: Water, which coats protein surfaces (10). Related molecules rank in inducing capacity as: ATP = ATPP = PPP > ADP = AMP-PNP = AMP-PCP = PP, while AMP, Adenosine, P, TMAO, and NaCl show no induction. However, when Adenosine and triphosphate join forces, ATP gains the integrated ability to initiate folding, inhibit aggregation, and enhance stability of proteins.
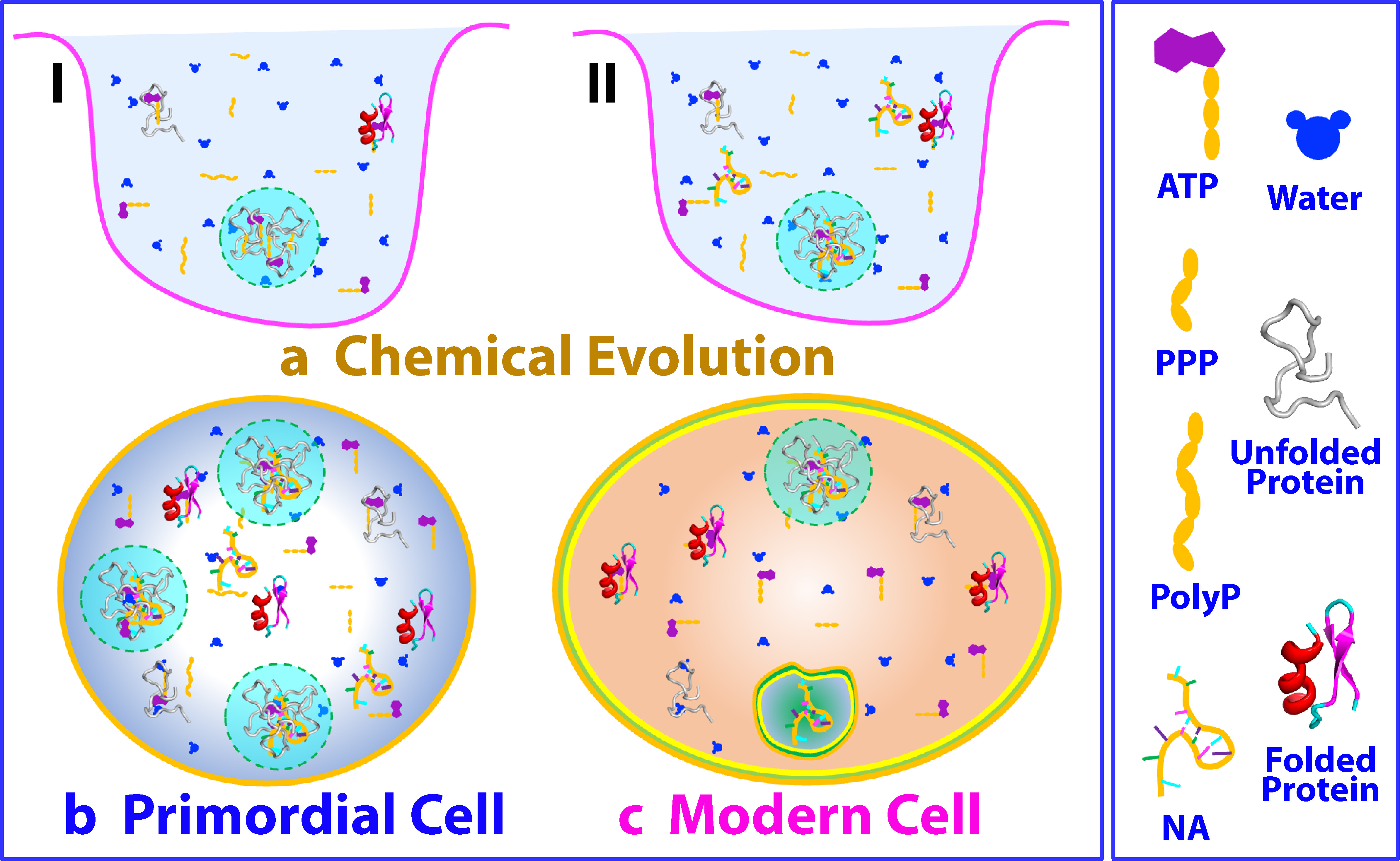
These newfound insights beckon us to ponder the broader significance of ATP and its constituent triphosphate/polyphosphates. It is becoming increasingly apparent that their influence could extend to the very origins of life itself where triphosphate is a molecule with deep-seated implications even in prebiotic chemical evolution (11). These molecules could have served as foundational building blocks in the formation of early structures, while simultaneously acting as primitive chaperones that facilitated protein folding with unparalleled efficiency. In the context of prebiotic chemical evolution, triphosphate/polyphosphates might have fueled the creation of liquid droplets by driving LLPS, setting the stage for the eventual emergence of rudimentary cellular entities (I of Fig. 1a). As life's narrative evolved and oligonucleic acids emerged, ATP and triphosphate/polyphosphates found renewed purpose in enhancing protein folding (II of Fig. 1a). This intricate interplay could have paved the way for the creation of primordial cells, the early cellular forms devoid of the intricate protein folding and degradation machineries we observe in modern cells (Fig. 1b). Even today, ATP continues to hold hidden roles, shaping protein homeostasis and mitigating aggregation through diverse mechanisms (Fig. 1c).
However, the story doesn't end here. The complex relationship between triphosphate/polyphosphates and protein behavior has a caveat. The concentrations of triphosphate/polyphosphates in modern cells are notably lower, likely due to their potential to induce aggregation of partially-folded and intrinsically-disordered proteins. Eukaryotic cells, in particular, seem to be rich in such proteins, and this revelation could offer a clue as to why the concentrations of these molecules have dwindled. Interestingly, however, some single-cell organisms have still harnessed polyphosphates as primordial chaperones (12), suggesting that the intricate relationship between energy currency and protein regulation extends deep into the evolutionary past.
While the mysteries of our primordial past are intriguing, ATP's significance extends to our present reality. Neurodegenerative diseases like ALS are universally associated with protein misfolding and aggregation. Here's where ATP steps in again. In modern cells, ATP appears also to energy-independently function as a guardian against misfolding and aggregation, enhancing the intrinsic folding capacity of proteins encoded in the sequences. This revelation might help us understand the perplexing onset of familial ALS and the increased risk of neurodegenerative diseases as we age, a period when cellular ATP levels decline.
In conclusion, ATP's multifaceted role transcends the bounds of energy exchange. Its influence over protein homeostasis, from folding to aggregation prevention, paints a richer picture of cellular orchestration. Moreover, the origins of life itself might have been shaped by ATP's unique abilities, carving a path for the emergence of life's first building blocks. As we continue to decipher the mysteries of this marvellous molecule, we uncover not only the intricacies of cellular functions but also the enigmatic origins of existence.
References
- M. M. Cox. Lehninger’s Principles of Biochemistry, 5th edn (W.H. Freeman and Company, New York, 2005).
- A. Patel et al. ATP as a biological hydrotrope. Science 356, 753–756 (2017).
- J. Kang, et al. A unified mechanism for LLPS of ALS/FTLD-causing FUS as well as its modulation by ATP and oligonucleic acids. PLoS Biol 17, e3000327 (2019).
- M. Dang, et al. Arg/Lys-containing IDRs are cryptic binding domains for ATP and nucleic acids that interplay to modulate LLPS. Commun. Biol. 5, 1315 (2022).
- M. Dang, et al. ATP biphasically modulates LLPS of TDP-43 PLD by specifically binding arginine residues. Commun Biol. 4, 714. (2021).
- M. Dang, et al. ATP and nucleic acids competitively modulate LLPS of the SARS-CoV2 nucleocapsid protein. Commun. Biol. 6, 80 (2023).
- J. Kang, et al. ATP binds and inhibits the neurodegeneration-associated fibrillization of the FUS RRM domain. Commun Biol. 2, 223 (2019).
- J. Song. Adenosine triphosphate energy-independently controls protein homeostasis with unique structure and diverse mechanisms. Protein Sci. 30, 1277-1293 (2021).
- J. Kang, et al. ATP induces folding of ALS-causing C71G-hPFN1 and nascent hSOD1. Commun. Chem. 6, 80 (2023).
- J. Song. Insight into "insoluble proteins" with pure water. FEBS Lett. 583, 953-9 (2009).
- F. H. Westheimer, Why nature chose phosphates. Science. 235, 1173-1178 (1987).
- M. J. Gray, et al. Polyphosphate is a primordial chaperone. Mol Cell. 53:689–699 (2014).
Follow the Topic
-
Communications Chemistry
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Sep 30, 2026
Sustainable waste management through polymer upcycling
Publishing Model: Open Access
Deadline: Aug 31, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in