Behind the Paper: Unraveling the Metabolic Alterations in Parkinson’s Disease Through Comprehensive Blood Metabolomics
Published in Chemistry, Neuroscience, and Biomedical Research
What Did the Researchers Discover?
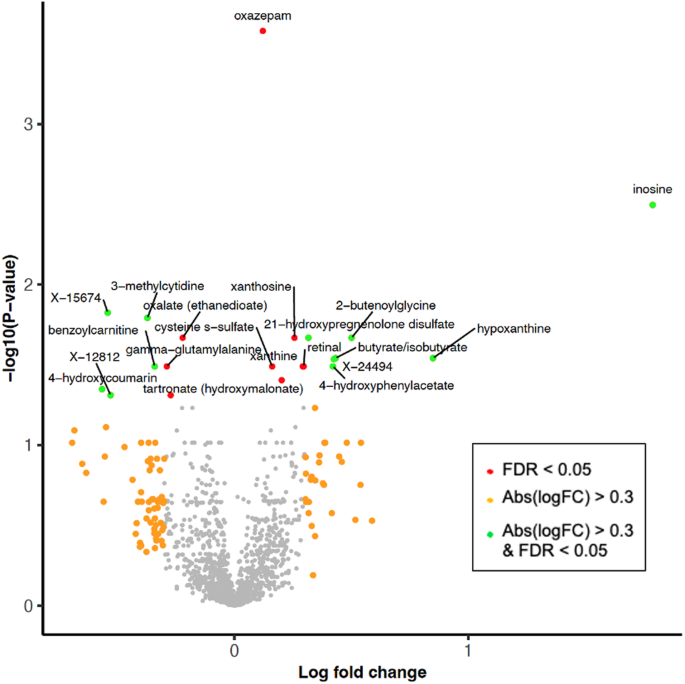
A team of scientists worked on decoding the metabolic alterations associated with Parkinson’s Disease (PD). By analyzing the blood plasma of PD patients and healthy controls within the Luxembourg Parkinson's Study, they unveiled significant changes in metabolite levels and pathway activities, particularly highlighting a pronounced alteration in xanthine metabolism. Their analysis revealed that certain groups of functionally associated metabolites, including those related to xanthine, catecholamine, and fatty acid metabolism, were significantly altered in PD patients compared to controls. Furthermore, their integrative network analysis pinpointed the enzyme hypoxanthine phosphoribosyltransferase 1 (HPRT1) as a potential regulatory hub connecting these metabolic disruptions to the pathological loss of cellular adenosine triphosphate (ATP), offering new insights into PD's molecular landscape.
How Does This Affect Our Understanding of Parkinson’s Disease?
This study deepens our understanding of PD at a molecular level, showcasing how systemic metabolic changes are intricately linked to the disease’s pathology. The pronounced alterations in xanthine metabolism and the identification of HPRT1 as a potential key regulators underscore the complexity of PD’s metabolic disruptions. These findings suggest that PD's pathogenesis could involve a multifaceted network of metabolic pathways, going beyond the traditionally emphasized dopaminergic system. By shedding light on these metabolic perturbations, the research opens new avenues for exploring diagnostic markers and therapeutic targets that could pave the way for innovative treatment strategies targeting the disease’s metabolic underpinnings.
Why Is This Important?
The importance of this study lies in its potential to change the diagnostic and therapeutic landscape of PD. The identification of specific metabolic alterations and their connection to PD’s pathology provides valuable biomarkers for potential early diagnosis, enabling interventions at an earlier stage. Moreover, unraveling the role of HPRT1 in regulating these metabolic changes offers a promising target for developing new disease-modifying treatments. By moving beyond the dopamine-centric view of PD, this research offers hope for more effective and personalized management strategies that address the disease's broader biological impact.
What Challenges Did the Researchers Face?
Conducting such a comprehensive metabolomic analysis presented several challenges, including the task of distinguishing between disease-specific metabolic changes and those resulting from environmental factors or other confounding variables. The team navigated these complexities by employing robust statistical and bioinformatics techniques, ensuring the reliability of their findings. Additionally, integrating metabolomic data with transcriptomic insights to unravel the complex network of interactions underscored the importance of interdisciplinary approaches in understanding multifaceted diseases such as PD.
Any Surprising Findings?
One of the most intriguing aspects of this study was the discovery of HPRT1's potential role in connecting metabolic alterations to PD's pathology, suggesting a link between xanthine metabolism and cellular energy dysregulation. This finding opens new research directions exploring HPRT1's role in PD and its potential as a therapeutic target, highlighting the study's capacity to unveil novel insights into the disease's underlying mechanisms.
What’s Next?
Looking forward, the researchers aim to validate these findings in larger, independent cohorts and through experimental models, enhancing our understanding of these metabolic pathways' roles in PD. Further exploration of HPRT1's regulatory mechanisms and its impact on cellular metabolism and PD pathology is also on the horizon. Ultimately, this research lays the groundwork for developing new diagnostic tools and therapeutic strategies for Parkinson’s Disease.
Follow the Topic
-
npj Parkinson's Disease
This journal publishes original basic science, translational and clinical research related to Parkinson's disease, including anatomy, etiology, genetics, cellular and molecular physiology, neurophysiology, epidemiology and therapeutic development and treatments.
Related Collections
With Collections, you can get published faster and increase your visibility.
The neuroimmune-axis and ageing in Parkinson’s Disease
Publishing Model: Open Access
Deadline: Jul 15, 2026
Cognition - preclinical models, and preclinical unmet need
Publishing Model: Open Access
Deadline: Jul 27, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in