Benefits of a Quadruplet Regimen Containing Isatuximab: Deep Responses in Patients With Newly Diagnosed Multiple Myeloma Ineligible/With No Immediate Intent for Transplant
Published in Cancer
Despite treatment advances contributing to improved outcomes in patients with multiple myeloma (MM), those who are ineligible for autologous stem cell transplantation (ASCT) have lower survival rates, which is due in part to receiving less-intensive therapy as a result of advanced age and/or comorbidities.1,2 The achievement of minimal residual disease negative (MRD-) may represent an early marker of good prognosis in the newly diagnosed MM (NDMM) setting, as it has been shown to be associated with prolonged progression-free survival (PFS) and overall survival (OS).3-5
Bortezomib-lenalidomide-dexamethasone (VRd) has become a standard-of-care regimen in patients with NDMM ineligible for/with no intent for immediate ASCT based on the Phase 3 SWOG S0777 study, in which the administration of VRd until disease progression resulted in significantly longer OS than with the administration of lenalidomide-dexamethasone (Rd) alone.6 Results from the DETERMINATION study also provide support for the VRd combination plus ASCT.7 Patients ineligible for ASCT may also benefit from backbone regimens plus novel agents (eg, monoclonal antibodies) in the frontline setting, resulting in deep and durable responses.1,8
In our Phase 1b study, we evaluated the preliminary efficacy, safety, and pharmacokinetics (PK) of the quadruplet regimen of the anti-CD38 monoclonal antibody isatuximab combined with VRd (Isa-VRd) in transplant-ineligible patients with NDMM (Part A cohort). In the Isa-VRd Part B cohort, patients with no intent for immediate transplant were also included.
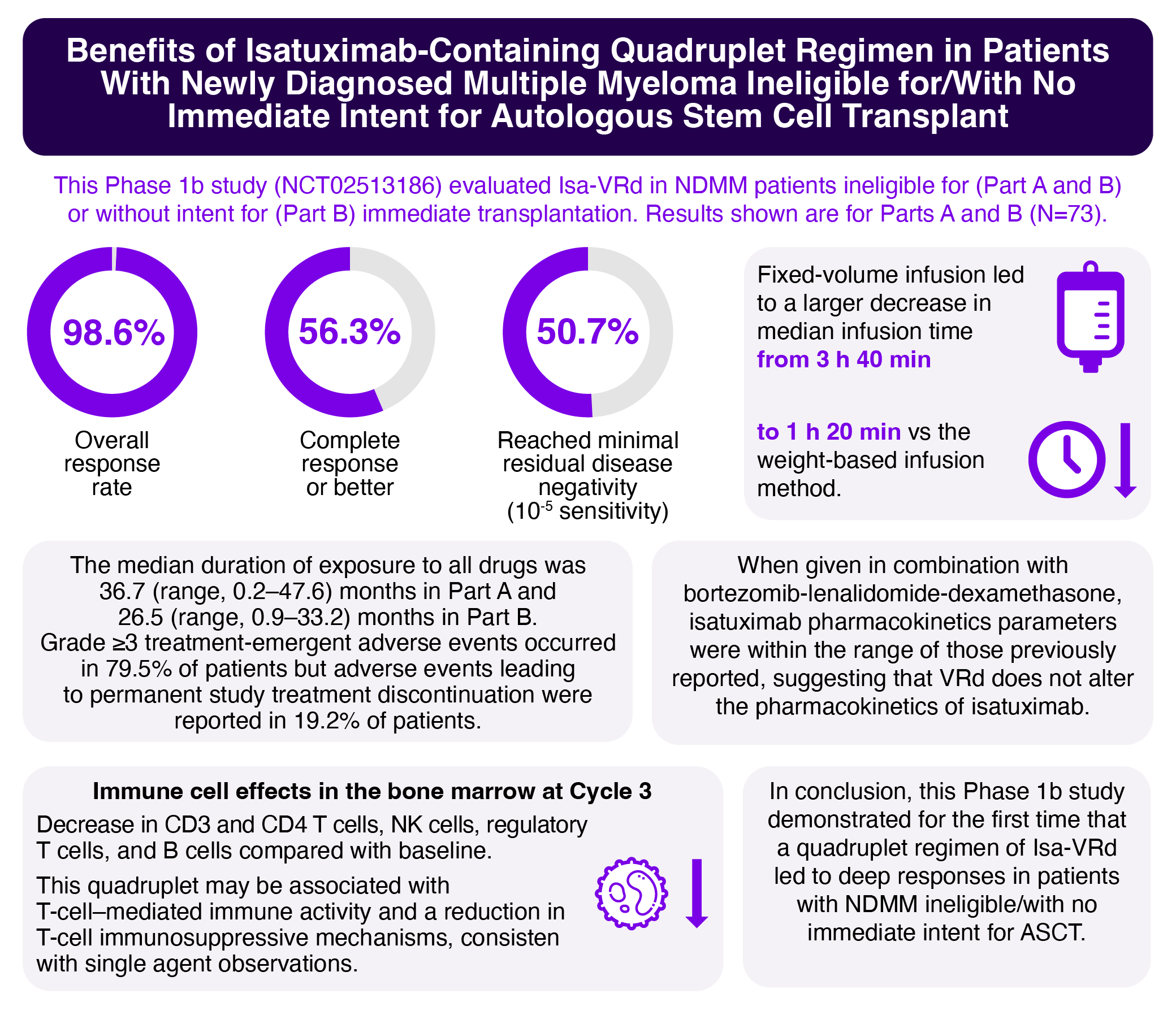
Overall, 73 patients received four 6-week induction cycles of Isa-VRd, then maintenance with Isa-Rd in 4-week cycles until disease progression, unacceptable toxicity, or patient withdrawal. In Part A, a weight-based volume infusion of isatuximab 10 mg/kg was used based on the most recent patient weight available on the day of infusion preparation. In Part B, isatuximab 10 mg/kg was diluted and administered intravenously from a fixed-volume infusion bag containing 250 mL of 0.9% sodium chloride solution. At the time of analysis, all ongoing patients from Part A had switched to the fixed-volume infusion method.
In the efficacy population (n = 71), the overall response rate was 98.6%, with 56.3% achieving a complete response or better (sCR/CR), and 36/71 (50.7%) patients reaching MRD- (10-5 sensitivity). Notably, this study demonstrated for the first time that a quadruplet regimen of isatuximab plus VRd led to deep responses in patients with NDMM ineligible/with no immediate intent for ASCT.
Grade ≥3 treatment-emergent adverse events (TEAEs) and treatment-emergent serious AEs occurred in 79.5% (58/73) and 53.4% (39/73) of patients, but those leading to permanent study treatment discontinuation were reported in only 14 (19.2%) patients. In the overall safety population, there were 7 TEAEs leading to death, including 2 events of COVID-19 and 1 event each of listeremia, progressive disease, metastatic breast cancer, metastatic melanoma, and diverticulitis. Isa-VRd exhibited a safety profile consistent with that of each individual drug, suggesting that this quadruplet combination is both feasible and effective in this patient population.
Isatuximab PK parameters were within the previously reported range, suggesting that VRd does not alter the PK of isatuximab. Data investigating the impact of immunogenicity on safety, efficacy, and PK profile are forthcoming.
This study also evaluated the duration of isatuximab infusion. With the fixed-volume infusion, all patients in Part B and patients in Part A who switched from the weight-based infusion method exhibited a larger decrease in the median infusion time from approximately 3 h 40 min during the first infusion to approximately 1 h 20 min after the third infusion. This is in contrast with patients in Part A with the weight-based infusion method (approximately 3 h 40 min at first infusion to approximately 2 h 35 min after the third infusion), and with no increase in infusion reactions reported in Part B vs Part A.
Descriptive analysis of blood immune cell subpopulations in our study showed a decrease in CD3 and CD4 T cells, NK cells, regulatory T cells, and B cells at Cycle 3, which coincides with previous observations following single-agent anti-CD38 antibody treatment.9 Our results suggest that Isa-VRd may be associated with T-cell–mediated immune activity and a reduction in T-cell immunosuppressive mechanisms in this patient population.
Overall, these data support additional confirmatory Phase 3 studies of this isatuximab quadruplet in the NDMM setting that are currently ongoing, such as NCT03319667 (IMROZ) and NCT03617731 (GMMG-HD7), with the objective to explore outcomes in transplant-ineligible and -eligible patients, respectively. Furthermore, the Phase 3 BENEFIT study (NCT04751877) was designed to investigate isatuximab-based triplet (Isa-Rd) vs quadruplet (Isa-VRd) regimens in patients with NDMM ineligible for ASCT. This study will help determine any added value of the quadruplet vs triplet regimen in this patient population, considering the safety profile of each regimen.
In conclusion, despite therapeutic advances that prolong survival and improve patient outcomes, patients with NDMM still eventually relapse; therefore, there is high demand for newer treatment regimens to combat disease progression. Our results demonstrated that the addition of isatuximab to the standard-of-care triplet backbone, VRd, improved outcomes in NDMM and could represent a promising frontline treatment option for transplant-ineligible and -eligible patients.
References
- Djebbari F, Sharpley FA, McLain-Smith S, Vallance G, Eyre TA, Kothari J, et al. Treatment-free interval as an additional measure of efficacy in a large UK dataset of transplant ineligible myeloma patients. PLoS One. 2020;15:e0229469.
- Mateos MV, San Miguel JF. Management of multiple myeloma in the newly diagnosed patient. Hematology Am Soc Hematol Educ Program. 2017;2017:498-507.
- Landgren O, Devlin S, Boulad M, Mailankody S. Role of MRD status in relation to clinical outcomes in newly diagnosed multiple myeloma patients: a meta-analysis. Bone Marrow Transplant. 2016;51:1565-8.
- Perrot A, Lauwers-Cances V, Corre J, Robillard N, Hulin C, Chretien ML, et al. Minimal residual disease negativity using deep sequencing is a major prognostic factor in multiple myeloma. Blood. 2018;132:2456-64.
- Avet-Loiseau H, Ludwig H, Landgren O, Paiva B, Morris C, Yang H, et al. Minimal residual disease status as a surrogate endpoint for progression-free survival in newly diagnosed multiple myeloma studies: a meta-analysis. Clin Lymphoma Myeloma Leuk. 2020;20:e30-e7.
- Durie BGM, Hoering A, Abidi MH, Rajkumar SV, Epstein J, Kahanic SP, et al. Bortezomib with lenalidomide and dexamethasone versus lenalidomide and dexamethasone alone in patients with newly diagnosed myeloma without intent for immediate autologous stem-cell transplant (SWOG S0777): a randomised, open-label, phase 3 trial. Lancet. 2017;389:519-27.
- Richardson PG, Jacobus SJ, Weller EA, Hassoun H, Lonial S, Raje NS, et al; DETERMINATION Investigators. Triplet therapy, transplantation, and maintenance until progression in myeloma. N Engl J Med. 2022;387:132-147.
- Yimer H, Melear J, Faber E, Bensinger WI, Burke JM, Narang M, et al. Daratumumab, bortezomib, cyclophosphamide and dexamethasone in newly diagnosed and relapsed multiple myeloma: LYRA study. Br J Haematol. 2019;185:492-502.
- Dimopoulos M, Bringhen S, Anttila P, Capra M, Cavo M, Cole C, et al. Isatuximab as monotherapy and combined with dexamethasone in patients with relapsed/refractory multiple myeloma. Blood. 2021;137:1154-65.
Follow the Topic
-
Leukemia
This journal publishes high quality, peer reviewed research that covers all aspects of the research and treatment of leukemia and allied diseases. Topics of interest include oncogenes, growth factors, stem cells, leukemia genomics, cell cycle, signal transduction and molecular targets for therapy.







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in