Blood coagulation testing using smartphones
Published in Bioengineering & Biotechnology

For millions of people, medical conditions such as atrial fibrillation, mechanical heart valves and genetic mutations increase the risk of morbidity and mortality from blood clotting. These individuals require lifelong administration of anticoagulation drugs such as warfarin, an effective and cheap medication but also one of the most common causes of hospitalization due to adverse drug events. Hence, medication effects must be closely monitored via frequent prothrombin time (PT) or international normalized ratio (INR) tests to assess coagulation properties due to the drug’s narrow therapeutic index and interactions with food and other medications. While newer anticoagulants that do not rely on regular PT/INR testing are increasing in popularity, studies show that warfarin remains the most commonly prescribed outpatient blood thinner.
We have several family members in India and Hong Kong who frequently delay their monitoring of clotting time because it often involves a visit to a clinic laboratory. While devices for at-home testing exist, they are costly, and cost is often cited as the primary barrier to self-testing.
In our recently published Nature Communications paper, we present a frugal smartphone-based test for measuring blood clot time using just a single drop of blood. Demo code showing our system in action can be found here.
How does it work?
The vibration motor on the phone shakes a tiny cup of blood (10μl) containing a copper particle and an activator. The phone’s camera then tracks the motion of the particle which slows down as the clot forms. Back in the day, doctors used to manually rock tubes of blood back and forth to monitor how long it took a clot to form. This, however, requires a lot of blood, making it infeasible to use in home settings. The creative leap we make here is that we’re showing that by using the vibration motor on a smartphone, our algorithms can do the same thing, except with a single drop of blood. And we get accuracy similar to the best commercially available techniques.
Check out the video below to see it in action:
To calculate PT and INR, we run algorithms to compute two time stamps: 1) when the user inserts the blood and 2) when the particle stops moving. For the first time stamp, we're looking for when the user inserts a capillary tube containing the sample in the frame. For the end of the measurement, we look directly at the interior of the cup so that the only movement within those frames is the copper particle. The particle stops moving abruptly because blood clots very quickly, and you can observe that difference between frames. From there we can calculate the PT, and this can be mapped to INR.
We tested our system across 159 whole blood samples, 79 of which had a clotting disorder, and 140 plasma samples. The results of our test were strongly correlated with traditional measurement devices used in the lab or at-home.
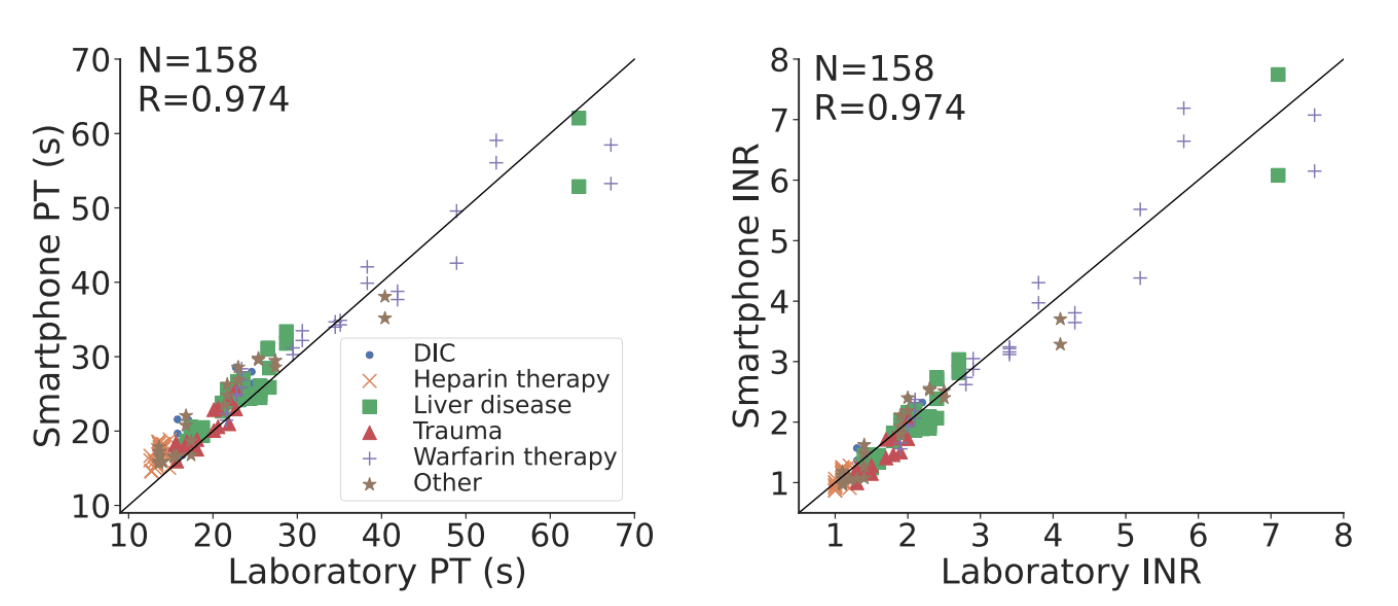
Looking forward, further work is required to make our proof-of-concept system easy to use and durable in the hands of patients. Future studies are also required to evaluate the performance of the device in real world scenarios including with patients at home, as well as in more resource-limited countries.
Check out our Nature communications paper for more details.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in