Classifying Activation Energies in Heterogeneous Catalysis
Published in Chemistry
The paper in Communication Chemistry is here: http://go.nature.com/2G7WuOG
When screening for catalysts computationally, energies of reaction intermediates and transition states are the key parameters that determines the catalytic activity and selectivity. The introduction of linear relations between energies of reaction intermediates and transition states, such as the BEP method, makes it viable to efficiently screen thousands of catalyst structures. However, establishing these relations is very time-consuming and requires significant computational power, thus limiting the application of the method. An electronic strategy that involves bond order conservation can be applied to determine the evolution of the interaction between the fragments of a reaction intermediate and the catalyst surface along the bond-breaking or bond-forming reaction coordinate. Based on the principle of bond order conservation, two functions (gamma and xi) can be extracted that uniquely define the potential energy surface along the reaction coordinate, from which the transition state energy can be obtained easily.
I have always been interested in the modeling aspects of heterogeneous catalysis. When I started in the field, a lot of the work was done by brute force, that is calculations on a single material with a well-defined orientation. The results that are achieved are interesting for this particular system but it never provides information how one should proceed from there. Linking materials properties to their reactivity based on the underlying electronic structure is important for developing descriptive models and today a number of such models exist in heterogeneous catalysis – d-band center approaches, Brønsted-Evans-Polanyi relations, linear scaling relations, Generalized coordination number approaches to name a few. With this work we add a new tool to the toolbox from which transition state energies for catalyzed chemical reactions can be obtained much faster than with existing approaches. This work was done together with Dr. Liang Yu and Dr. Laia V. Arribas
In this paper we show that the two functions gamma and xi depend entirely on the local electronic configuration of the bond being formed or broken, hence reaction steps involving similar bond types like sp3 hybridized C-H bonds in e.g. CH4, CH3*, CH2*, CH* can be considered part of the same class. Using such classification of reactions allows gamma and xi functions derived for a single member in the class to be applied to all its members.
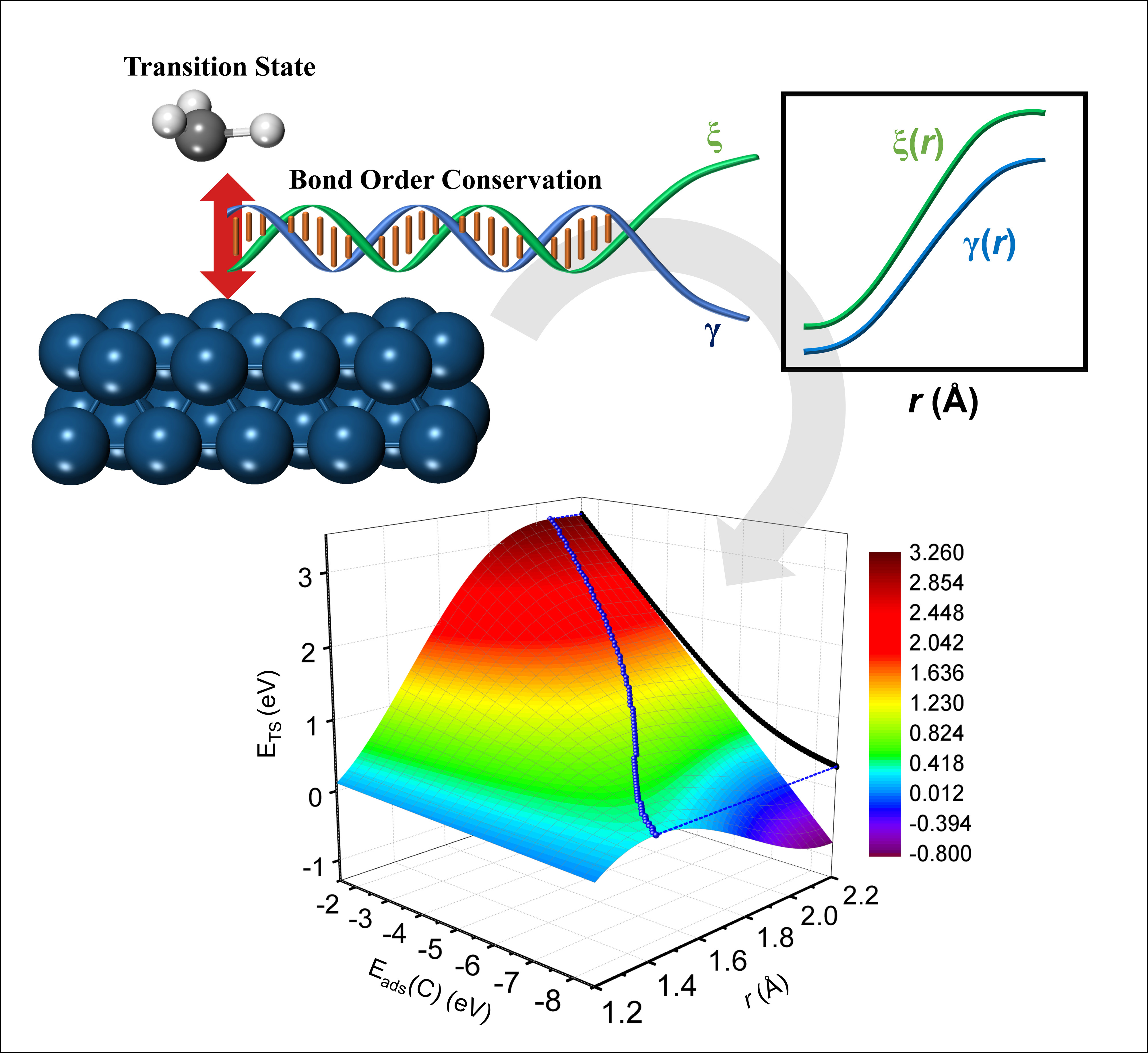
Image caption: Schematics showing how unique functions gamma and xi determined through a bond order conservation approach can be linked to the estimation of activation energies important in chemical reactions on heterogeneous surfaces.
Once template functions for gamma and xi for a given class have been derived, the additional data needed to transfer it to a large number of reactions is very limited and more importantly no complicated constrained maximization simulations like Nudged Elastic Band calculations are needed. With this approach a new method is introduced with which transition state energy relations can be established at a much lower computational cost compared to traditional methods. As such this will substantially accelerate the pace at which computer-aided screening of new catalysts can be performed.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in