Developing electrokinetic mining technology for recovering rare earth elements from weathering crusts
Published in Earth & Environment
Rare earth elements (REEs), especially heavy REEs (HREEs), flourish modern industry by triggering innovation of materials in the field of aerospace, electronics, clean energy, national defense and so on. The irreplaceable role that HREE plays promotes the global demand of raw HREEs, which, further stimulates HREE mining industries. Currently, HREEs are dominantly mined from ion-adsorption rare earth deposits (IADs) discovered in South China, and the regional mining produces and supports 95% of the global demand of HREEs. However, the conventional mining applies excessive usage of ammonium-salt-based leaching agents to recover HREEs from IADs, such technique exhibits low efficiency and devastating environmental impact on the local eco-system. Shifting from the traditional “mountain scraping” type of mining to a new generation of green HREE mining has become the main focus of the REE research and development.
Electrokinetic technology involves the application of a direct or alternating electric field to accelerate the migration of movable species such as metals, organics, and water. There has been an increasing interest in using electrokinetics as an in situ clean technology to remove/separate contaminates from soils. HREEs exist primarily as hydrated trivalent cations in IADs, which can be leached by electrolyte solutions and transported by an applied field force, e.g., concentration gradient field in the current leaching technique. Inspired by these physical processes, we propose the concept of electrokinetic-mining (EKM) IADs for efficient and sustainable REE recovery (Fig. 1A), which aims at applying electric field to accelerate the migration of REEs and simultaneously reducing the quantity of chemical agents required to transport ions.
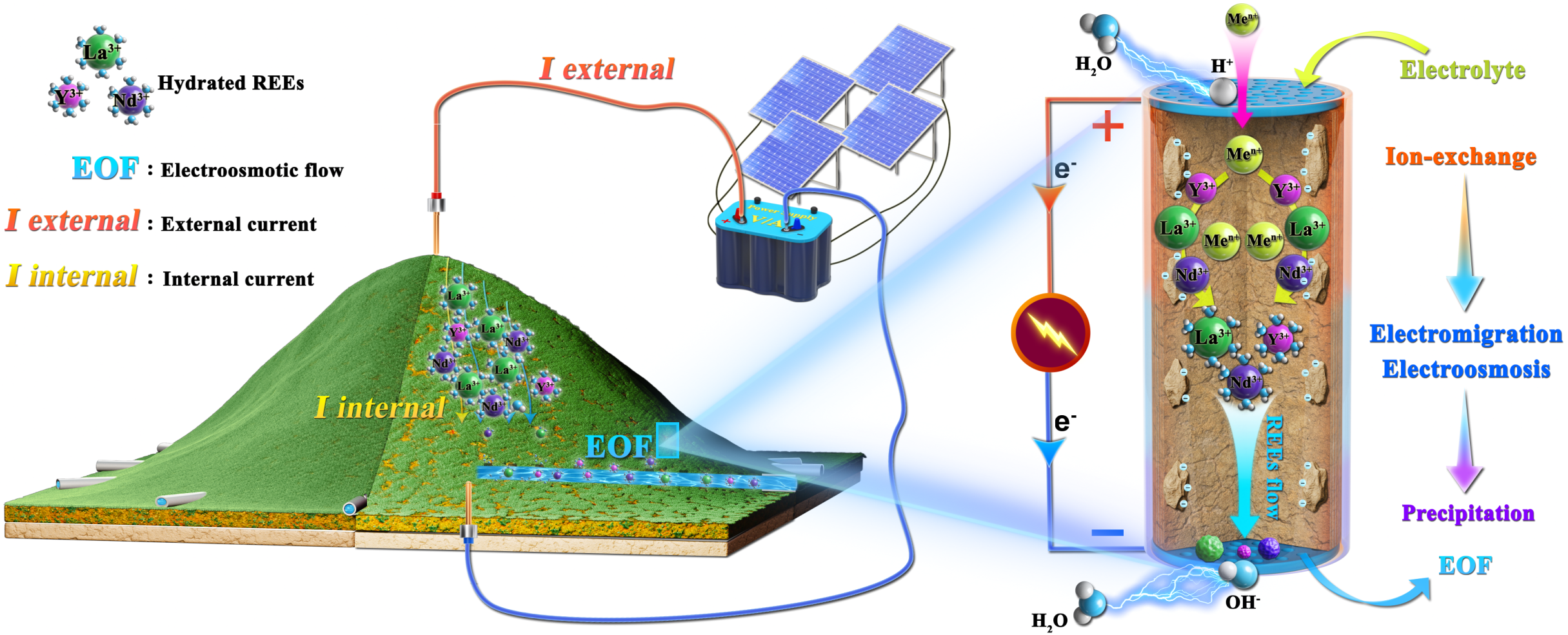
Fig. 1. Illustration of IAD mining via EKM. (left) 3D illustration of in situ EKM for an IAD. The hydrated REEs are expected to be mobilized and extracted from an IAD under electrokinetic effects. (right) Illustration of the EKM mechanisms for REE recovery, including ion exchange, electromigration, electroosmosis, and collection of electroosmotic flow (EOF) and precipitation.
To evaluate the feasibility of EKM for REE recovery, we first carried out bench-scale experiments in a homemade prototype with a simulated IAD. Results suggested that the REE recovery efficiency achieved by the EKM technique was 2.6 times higher than that achieved by the ammonium leaching technique (Fig. 2A). To assess the scalability of the EKM technique, we further carried out scaled-up experiments at the kilogram scale (20 kg) in a larger EKM setup. In the scaled-up experiments, we found that the current density was dramatically higher than that in the bench-scale experiments at the same voltage gradient, which was attributed to a lower total effective electric resistance (16ρ) in the scaled-up experiments compared with that (66ρ) in the bench-scale experiments. This finding can help reduce the applied voltage and the electrolyte dosage in larger-scale applications. The main findings in scaled-up experiments were consistent with in our bench-scale experiments, i.e., the EKM technique can achieve higher recovery efficiency and requires less treatment time. For instance, the scaled-up experiments achieved a recovery efficiency of 96% within 67 h by using the EKM technique, while that using the conventional technique was only ~62% at 130 h (Fig. 2B). Based on the successful bench-scale and scaled-up experiments, we applied the EKM technique to an actual IAD (~14 t-scale) via an on-site field experiment. Results suggested that the REE recovery efficiency achieved higher than 90% in 264 h (Figs. 2C and 2D).
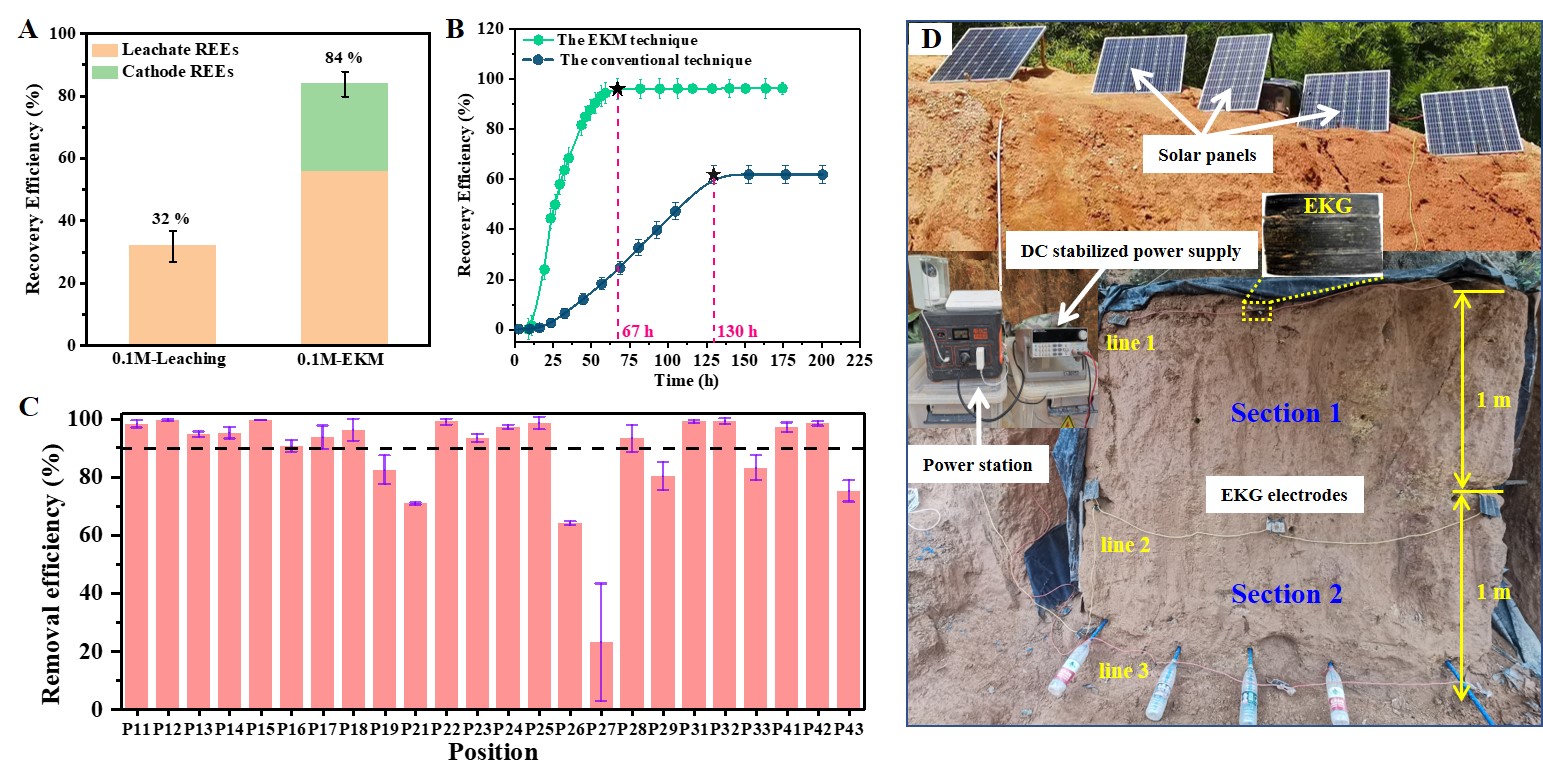
Fig. 2. The obtained results for EKM experiments at various scales. (A) Results for bench-scale EKM experiments. (B) Results for scaled-up EKM experiments. (C) Results for on-site field EKM experiment. (D) Photos showing the experimental setup for the on-site field EKM experiment.
As the feasibility of the newly developed EKM technique was systematically demonstrated via experiments at three scales. We then explored mechanisms underlying the high REE recovery efficiency of the EKM. We found that the electroosmosis flow (EOF) flux of the EKM technique (10.00×10-4 kg/m2·s at a voltage gradient of 0.07 V/cm) was approximately 3 times higher than the diffusive flux of the conventional leaching technique (3.54×10-4 kg/m2·s without an applied voltage gradient), which demonstrated that the applied electric field can markedly accelerate the migration of hydrated REEs. During IAD mining, REEs are primarily collected from the leachate. Thus, more REEs can be collected using EKM than using a conventional system based on such increment of the leachate flux, responsible for the improvement of the REE recovery efficiency. On the other hand, REEs were unidirectionally transported from the anode to the cathode, meaning that the leachate can be collected in a designed place. As demonstrated, the leakage of leachates into the fracture of a weathering crust under the action of gravity flow is the dominant reason for the low recovery efficiency of the current leaching techniques. The directional control of REE migration to a desired place during the EKM process can prevent or reduce leachate from leaking into offsite regions. Consequently, the REE recovery efficiency of the EKM technique is better than that of the conventional techniques wherein leakage usually occurs.
Interestingly, we identified an autonomous impurity purification (AIP) mechanism during the electrokinetic process, which substantially lowered the impurity content of the collected fluid. Specifically, the EKM technique led to a 66% and 61% decrease in metallic impurities in the collected contents compared with those using the conventional technique in bench-scale and scaled-up experiments, respectively. We investigated AIP by analyzing the contents of each impurity. Results suggested that high valent ions experience a larger electric field force compared with low valent ions experience in the same system. Consequently, high valent ions (e.g., REEs) arrive at the cathode more quickly than low valent metallic impurities (e.g., Ca2+ and K+), which forms a high potential barrier and impedes the transport of low valent metallic impurities toward the cathode. Therefore, we conclude that the first mechanism of AIP involves the mobility diversity (Fig 3). Furthermore, we noticed the formation of Al(OH)3 and CaCO3 precipitates near the cathode with assistance of energy-dispersive X-ray spectroscopy (EDS) and X-ray photoelectron spectroscopy (XPS), which decreased the impurity contents of Al and Ca. Thus, the second mechanism of AIP is induced by the reactivity diversity between REE and other active metal ions.
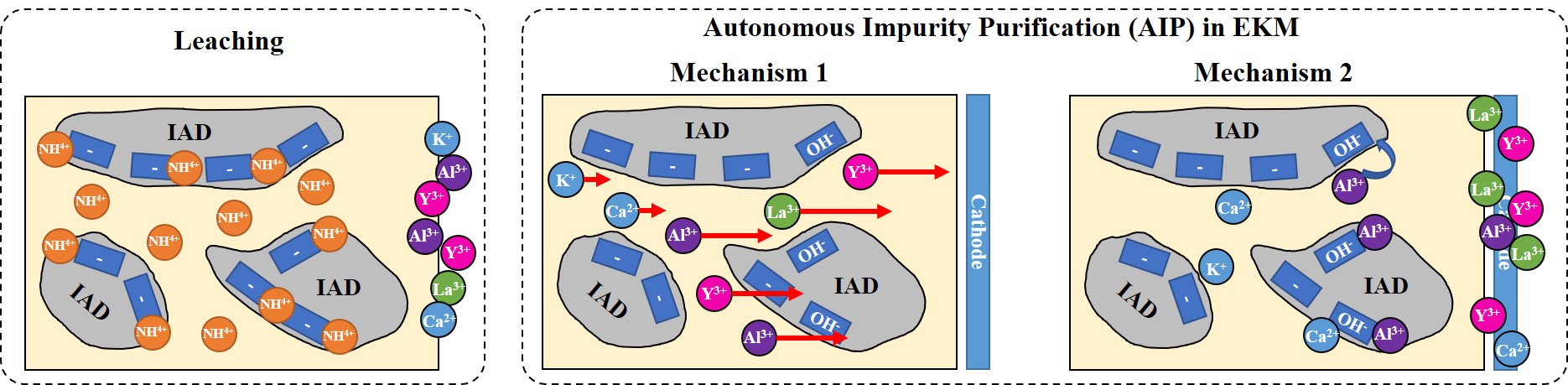
Fig. 3. Comparison between conventional leaching and the EKM technique and schematic of the AIP process.
Finally, we conducted a comparative techno-economic analysis (TEA) of EKM and conventional REE mining techniques, demonstrating the economic feasibility of EKM.
In summary, we conclude that the novel EKM is a sustainable technique, both environmentally and economically, and the newly developed technology enables green, efficient and selective recovery of REEs. The experimental results from different scales confirm that the EKM has great potential to be a new generation of mining technique not only for IADs for but also for other critical metals under conditions in which the metals exist in ionic and/or movable states, such as carbonate clay-type lithium, laterite nickel, and weathered crust elution-deposited scandium ores.
Follow the Topic
-
Nature Sustainability
This journal publishes significant original research from a broad range of natural, social and engineering fields about sustainability, its policy dimensions and possible solutions.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in