European patent protection for medical uses of known products and drug repurposing
Published in Bioengineering & Biotechnology
Context & Article Excepts
Drug repurposing is a strategy for identifying new uses of approved or investigational drugs that are outside the scope of the original medical indications. Finding new therapeutic uses for existing medicines holds significant potential, as it is often faster to develop a drug for a new use if it is already known to be safe and well-tolerated by the human body. Repurposing de-risked compounds with previously known therapeutic effects is also an attractive drug development strategy because it can potentially reduce overall drug development costs and shorten timelines to market introduction. If it is successful, the public at large benefits from the repurposing of ‘old’ drugs (known products) to treat more common and rare diseases.
An ongoing debate surrounding pharmaceutical repurposing is whether the current intellectual property (IP) regime, particularly with regard to patent protection for medical uses of known compounds and substances, may be hindering repurposing activity. This debate is taking place on both sides of the Atlantic. Commentators often argue that the patent system fails to provide adequate incentives to promote innovation in the field of drug repurposing—medical use patents, they allege, are too difficult to obtain.
Are medical use patents difficult to obtain? Despite the clear policy importance of developing new treatments faster and more cost effectively, there is limited evidence-based research on the actual patent protection of new medical uses, and relatively little is known about the scale and characteristics of this ongoing patenting activity. Here, we examine commentators’ allegations about patent failure with the overall research question: Is drug repositioning being hindered by the difficulty of obtaining patent protection for medical uses of known products at the European Patent Office (EPO)?
In a recent study aimed at mapping the European patent landscape for medical uses of known products, we showed that, in fact, there are increasing numbers of medical use patents being granted at the EPO. This suggested that general patent failure for new medical uses was perceived, not actual. That said, several outstanding questions remained to be studied, including those related to(i) the relative difficulty of obtaining patent protection for medical uses of known products in the strict sense (i.e., patents for which the broadest independent claim is directed to a medical use) compared to other technical fields and (ii) the characteristics of medical use patents granted by the EPO.
Research Questions
More specifically, questions that remain unanswered include:
- What has been the patenting trend over the last 20 years for medical use patents in the strict sense?;
- Are these patents difficult to obtain (i.e., what is their current grant rate?);
- How does the number of medical use patents granted per year at the EPO compare to the number of European Medicines Agency (EMA) drug authorizations for “new uses of existing medicines”?;
- Which organizations own these medical use patents?;
- In how many and which jurisdictions have they been protected?; and What has been the patenting activity by therapeutic area?
Policy Relevance
Answering these types of questions helps clarify the patent protection challenges surrounding drug repurposing and makes it possible to provide sound, evidence-based recommendations. Without more evidence-based IP research to address these types of questions, it is challenging for scholars, legislators, patent offices and judges to propose, amend, interpret or apply the law of medical use patents with reasonable chances of achieving desirable social goals such as incentivizing drug development across therapeutic areas, including rare diseases.
Empirical studies looking at real-world patenting activity can provide valuable evidence for assessing whether the difficulty of patenting new medical uses in Europe is actual or perceived. For instance, if drug repositioning is being hindered by the difficulty of obtaining patent protection for medical uses of known products, we would expect (i) a small number of patents awarded relative to the number of repurposed drug authorizations, (ii) a low patent grant rate in this area relative to other technical fields, (iii) a lack of coverage for important therapeutic areas and (iv) indications of low value to the owners as measured by metrics of private value of these patents.
Summary of Results
There are currently over 4,086 patents with independent claims directed to medical uses of known products. Of these, in 1,590 (38.9%) patents, the only independent and broadest claim in the patent is directed to a medical use of a known product. Most of these patents (99.2%) are protected in Europe (EPO) and the United States (USPTO).
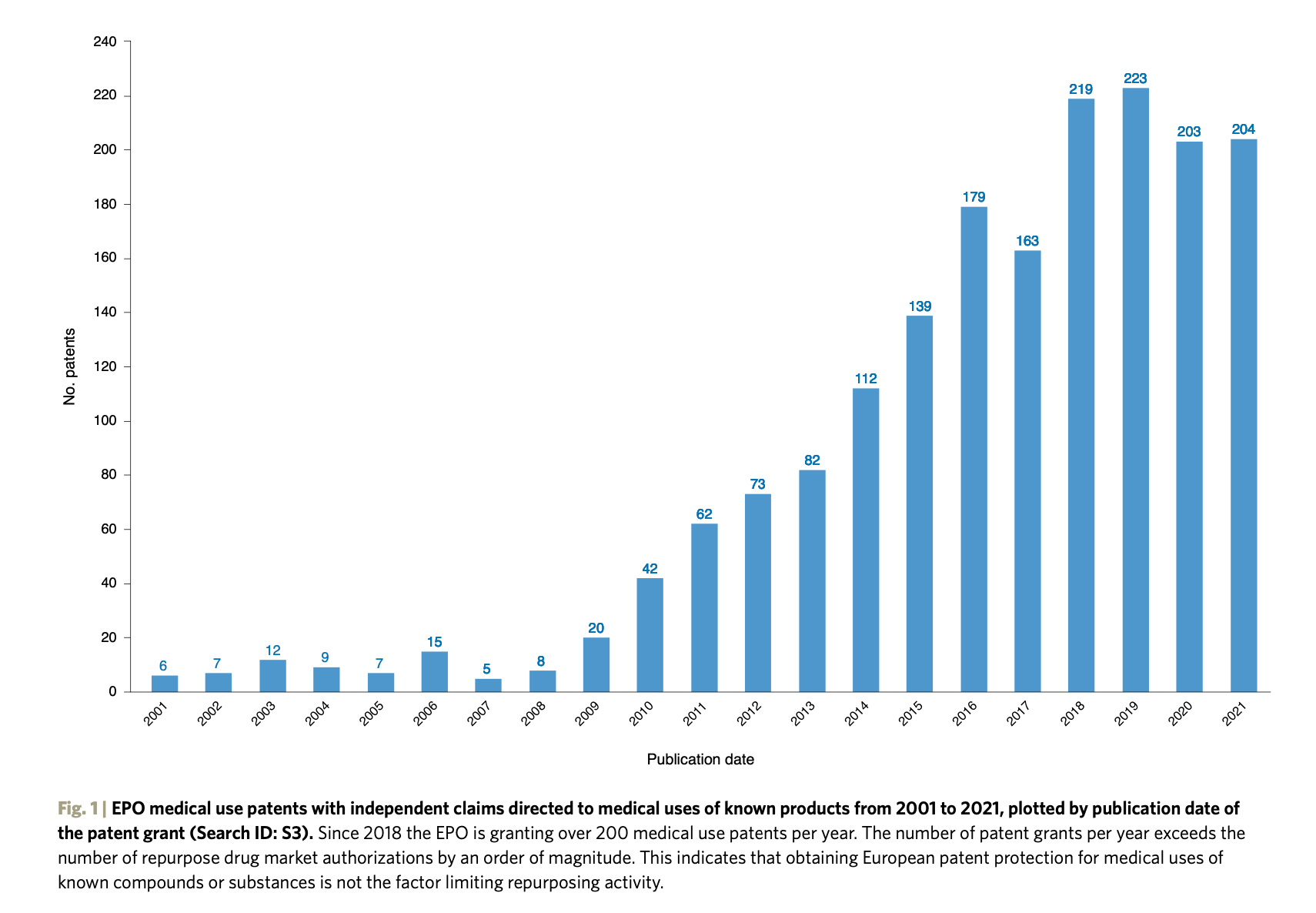
Since 2018, the EPO has been granting over 200 new medical use patents per year (with over 600 granted since 2018). The top 15% of medical use patents are protected in 20 or more jurisdictions (with the EPO counting as one). Although universities and publicly funded research institutes are active in protecting medical use inventions, the majority of patents protected in 20 or more jurisdictions are owned by pharmaceutical companies.
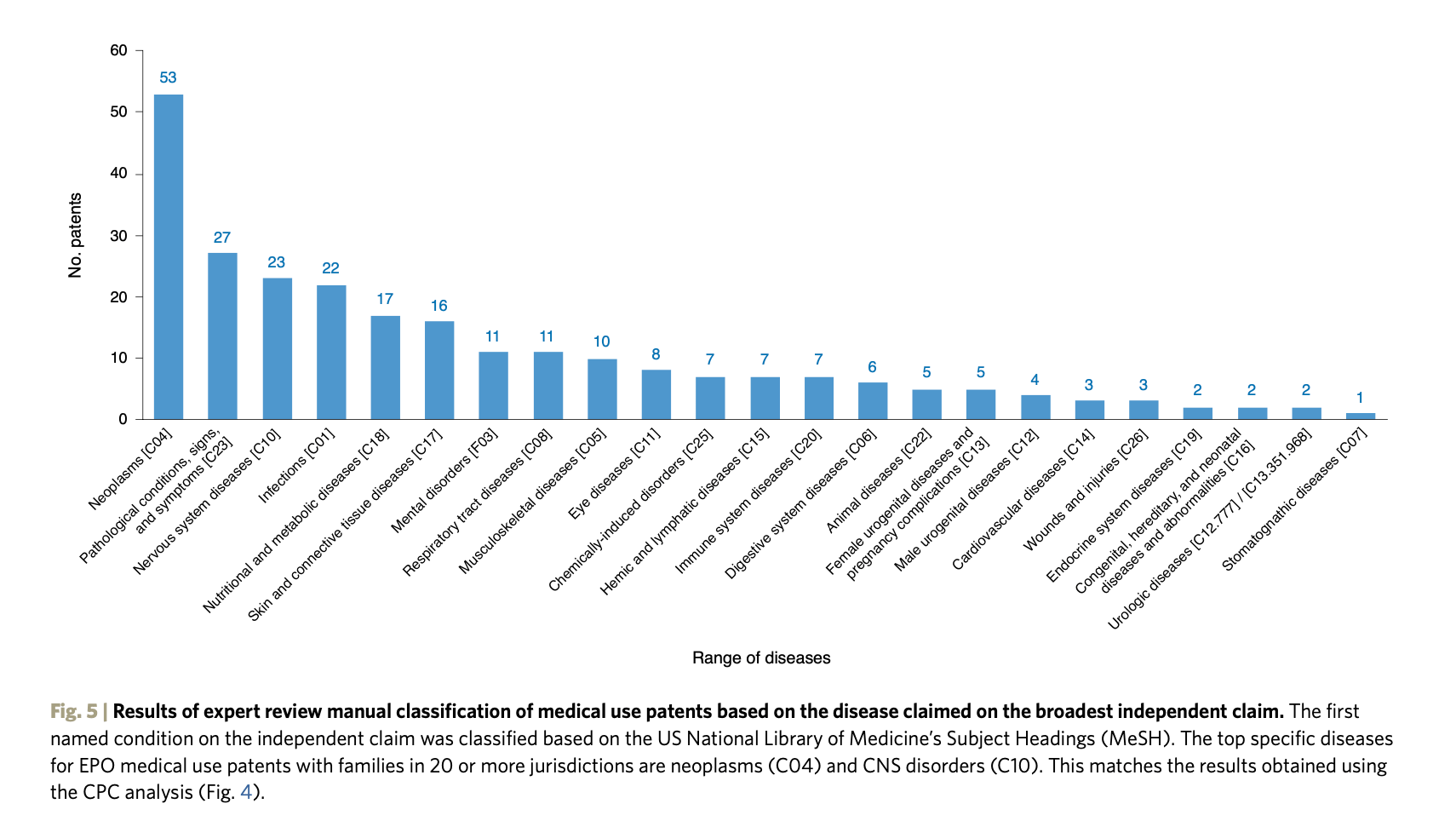
The top specific diseases for EPO medical use patents with families in 20 or more jurisdictions are cancer (MeSH C04) and diseases of the nervous system (MeSH C10). That said, our results show a broad coverage spanning a wide range of therapeutic areas.
Our finding that the number of in-force medical use patents is substantially higher than the corresponding number of repurposed drugs receiving EMA market authorizations suggests that, contrary to what other commentators have indicated, difficulty in obtaining patent protection for inventive activity involving second medical uses of known substances does not appear to be the key challenge or a bottleneck affecting repurposing activity. Furthermore, the majority of owners of these patents continue to pay the maintenance fees for the duration of the patent term, indicating that they consider their medical use patent portfolios to have strategic private value.
It appears that even if the medical use patents are not protecting a current repurposed drug with market authorization, the owners consider them to be of enough strategic long-term value to continue paying the maintenance fees, perhaps because that may protect forthcoming repurposed drugs in their pipeline.
Conclusions
In summary, we find (i) an increasing number of medical use patents relative to the number of repurposed drug authorizations, (ii) a relatively high grant rate for medical use patents indicating a clear legal basis for subject matter eligibility, (iii) a diversity of patent owners obtaining these medical use patents (public and private), (iv) a diversity of jurisdictions where they are protected and (v) coverage over all the major therapeutic areas. These findings indicate that obtaining patent protection for medical use inventions at the EPO does not appear to be a major challenge, and it is not likely to be hindering drug repurposing.
Our Hope
Given that there has been much debate and speculation about second medical use patents and that the patent system could be failing to provide suitable incentives for drug repurposing, we hope our results would be of interest to the readership of Nature and researchers working on drug repurposing and IP.
More Information
See the paper full-text (view-only version) or Nature Biotechnology article page (Nature Biotechnology, Volume 34, 465-471 (2022) – Published: 13 April 2022).
Behind the Paper
The idea behind the paper originated as part of the CeBIL (Collaborative Research Program in Biomedical Innovation Law), a joint collaborative research effort including University of Cambridge (LML, Faculty of Law), University of Copenhagen (CeBIL, Faculty of Law), Harvard Law School (Petrie-Flom), Harvard Medical School (PORTAL), and University of Michigan Law School. Preliminary results were first presented by Prof. Mateo Aboy & Prof. Kathleen Liddell at the 2019 CeBIL Retreat in Copenhagen.
Funding
The research was supported, in part, by a Novo Nordisk Foundation grant for a scientifically independent Collaborative Research Programme in Biomedical Innovation Law (Grant No. NNF17SA027784).
Follow the Topic
-
Nature Biotechnology
A monthly journal covering the science and business of biotechnology, with new concepts in technology/methodology of relevance to the biological, biomedical, agricultural and environmental sciences.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in