Inhibition of glycosphingolipid synthesis with eliglustat in combination with immune checkpoint inhibitors in advanced cancers: preclinical evidence and phase I clinical trial
Published in Biomedical Research

Glycosphingolipids (GSLs) are usually aberrantly expressed in cancer cells and undergo structural rearrangements that interfere with membrane proteins and anti-tumour immune responses during cell transformation, metastasis and multidrug resistance (1). Metabolic genes involved in lipid synthesis and uptake are downstream effectors of oncogenic alterations and are typically upregulated in tumours (2). GSL interferes with membrane proteins and anti-tumour immune responses, suggesting that GSL is a viable target for the treatment of tumours. Although a number of therapies targeting GSL are currently in clinical use, the efficacy of these agents remains limited. In addition, the specific effects of cancer immunotherapy on multiple GSLs remain unknown.
Our previous research has shown that loss of SPPL3, an upstream B3GNT5 shearing enzyme, inhibits the anti-tumour cytotoxicity of CAR T cells, suggesting that GSLs may protect cells presenting certain antigens from immune cell killing (3). It should be noted that glucose ceramide is the precursor of all GSLs and its synthesis is regulated by the key enzyme UGCG (4). Eliglustat, a drug approved by the FDA in 2014 for the treatment of type 1 Gaucher disease, can target UGCG and thereby alter GSL metabolism. We therefore hypothesise that eliglustat may restore tumour sensitivity to anticancer drugs by inhibiting GSL synthesis.
We first demonstrated that eliglustat mediates anti-tumour effects through the immune system rather than killing tumour cells directly. Using in vivo antibodies to deplete CD4 T, CD8 T and NK cells, it was found that the anti-tumour effect of eliglustat was counteracted by depletion of CD8 T, demonstrating that eliglustat acts primarily through CD8 T cells. Furthermore, our team found that the use of eliglustat inhibits the synthesis of GSLs and increases the level of exposure to antigens on the surface of the tumour cells, thereby enhancing the accessibility and anti-tumour activity of CD8 T cells. In addition, this therapy not only enhanced T-cell recognition of tumours, but also altered the composition of immune cells in the tumour microenvironment, increasing the number of antigen-specific T cells. Mechanistically, we demonstrated that eliglustat mediated the migration of CD8 T cells from lymph nodes to tumours and increased the number of antitumour-specific CD8 T cells and the diversity of T-cell receptors (TCRs). And, in a range of tumour models, we observed that eliglustat was able to significantly enhance the therapeutic efficacy of immune checkpoint inhibitors.
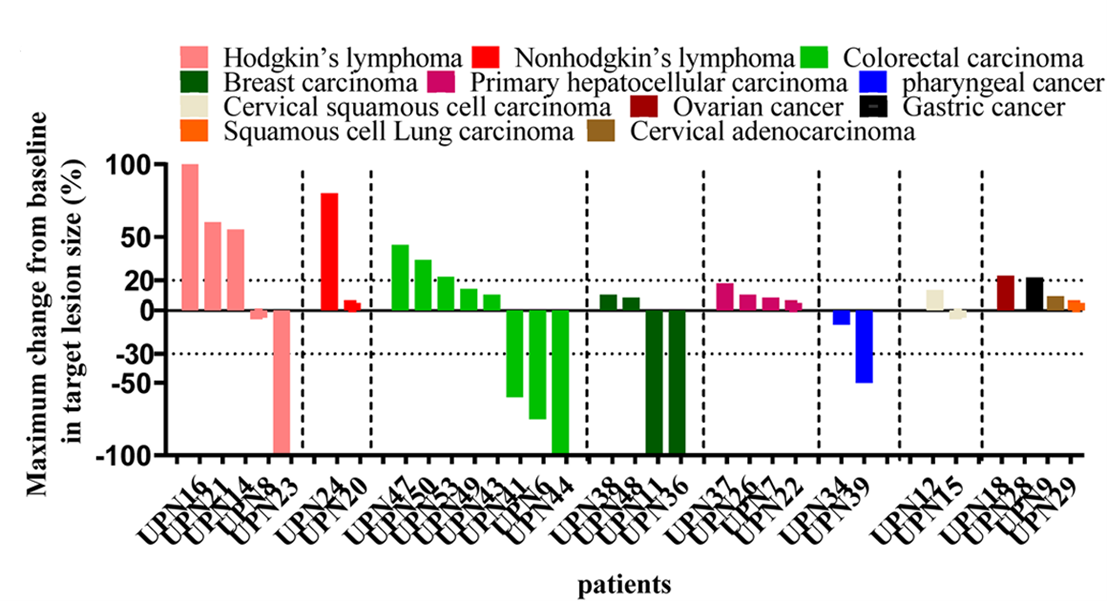
Based on this finding, we designed and conducted a proof-of-concept phase I clinical trial (NCT04944888) to evaluate the safety and feasibility of eliglustat in combination with a PD-1 antibody. The results of the trial showed that all prespecified primary endpoints were met, with only 1 out of 31 patients with advanced cancer experiencing a grade 3 adverse event and no grade 4 adverse events. Encouragingly, the therapy achieved a disease control rate of 71 percent, particularly in patients with mismatch repair proficient/microsatellite stable (pMMR/MSS) colorectal cancer, with one patient achieving complete remission, two patients in partial remission, and two patients with stable disease, which demonstrated that eliglustat combined with a PD-1 antibody exhibited a favourable safety profile(Fig. 1)
Interestingly, on the 7th of this month, the journal NATURE published an online study by Professor Kivanc Birsoy's team at Rockefeller University titled Glycosphingolipid synthesis mediates immune evasion in KRAS-driven cancer (5). Using functional genomics and lipidomics approaches, the article identifies the sphingolipid de novo synthesis pathway as an important pathway mediating KRAS-driven immune evasion in cancer cells. It has also been shown that blocking sphingolipid production enhances the anti-proliferative effect of natural killer cells and CD8+ T cells on tumour cells. This is consistent with what we have observed with eliglustat at the in vivo and in vitro levels. Furthermore, the research team also investigated the potential of UGCG, a pivotal enzyme in sphingolipid synthesis, as a therapeutic target. They observed that while treatment with eliglustat or CBT alone had minimal impact on tumour load in an in vivo model, the combination of eliglustat and CBT demonstrated a notable reduction in tumour load. This finding is in close alignment with the results of our experimental investigation. The authors suggest that this poor effect of eliglustat alone may be due to incomplete targeting of GSLs or effects on other cell types (including immune cells) in the TME, which strongly supports the findings in our paper. To confirm that it is indeed the depletion of glycosphingolipids by eliglustat, rather than the effects of eliglustat on other metabolic pathways, that leads to the immune response, we also used another drug, miglustat, which inhibits glycosphingolipid synthesis, and obtained the same results as eliglustat. These results highlight the potential of GSL deficiency in combination with immune checkpoint blockade therapy as a novel cancer immunotherapy strategy.
While differing in terms of the in vivo model employed, the surface molecules of interest, and the manner in which these molecules interact with immune cells, our study and theirs together confirm that inhibition of glycosphingolipids reverses tumour immune escape. It is particularly noteworthy that, although this research is still in its infancy, it has the potential to make significant contributions to the field of cancer therapy. Further clinical trials are planned to further validate the safety and efficacy of eliglustat in combination with immune checkpoint inhibitors and to explore its potential application in other types of cancer. Furthermore, as additional clinical data on the utilization of GSL inhibitors in conjunction with immunotherapy emerges, our comprehension of the mechanisms underlying immune evasion in cancer will be enhanced. This will also furnish a scientific foundation for the advancement of more efficacious personalized immunotherapy regimens.
References
- Hakomori S. Aberrant glycosylation in tumors and tumor-associated carbohydrate antigens. Advances in cancer research 1989;52:257-331 doi 10.1016/s0065-230x(08)60215-8.
- Biellmann F, Hülsmeier AJ, Zhou D, Cinelli P, Hennet T. The Lc3-synthase gene B3gnt5 is essential to pre-implantation development of the murine embryo. BMC developmental biology 2008;8:109 doi 10.1186/1471-213x-8-109.
- Yan X, Chen D, Ma X, Wang Y, Guo Y, Wei J, et al. CD58 loss in tumor cells confers functional impairment of CAR T cells. Blood Adv 2022;6(22):5844-56 doi 10.1182/bloodadvances.2022007891.
- Merrill AH, Jr. Sphingolipid and glycosphingolipid metabolic pathways in the era of sphingolipidomics. Chemical reviews 2011;111(10):6387-422 doi 10.1021/cr2002917.
- Soula, Mariluz et al. Glycosphingolipid synthesis mediates immune evasion in KRAS-driven cancer. Nature, 10.1038/s41586-024-07787-1. 7 Aug. 2024, doi:10.1038/s41586-024-07787-1.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in