Minimal Residual Disease in Myeloma is Here to Stay
Published in Cancer
Over the last decade, advances in next generation sequencing (NGS) technology have broadened its applicability for minimal residual disease (MRD) testing in a wide variety of malignancies. In the context of multiple myeloma (MM), improving therapies have led to deeper responses, beyond the traditional quantitation by classic disease biomarkers such as monoclonal protein determination. In these cases, MRD has allowed for better stratification and quantification of possible residual disease. The International Myeloma Working Group (IMWG) updated its response criteria in 2016 and include MRD as a parameter of response, using either NGS or next-generation flow cytometry (1). It has become increasingly clear that subclassification of MM responses less than MRD negativity may be merely an academic exercise since there is no clear distinction between their outcomes. The specificity of the B cell receptor rearrangement makes NGS MRD detection not only disease specific, or even patient specific, but rather clone specific (2).
Three meta-analyses have demonstrated the prognostic significancy of MRD negativity in MM for both progression-free and overall survival (3, 4, 5). Moreover, the higher the sensitivity level of the MRD test used the more discriminatory power it has for outcomes. The first generation of MRD assays were flow cytometry-based and achieved sensitivities ranging from 10-4 to 10-5, detecting one cell in 10,000 or 100,000, respectively. Recent advances in flow cytometry, along with the incorporation of NGS, allow routine measurement at sensitivities of 10-6. MRD testing is now supported by various platforms of assays, most are standardized, and some (NGS) even have FDA approval and have coverage by CMMS and other payers.
Despite the strong evidence supporting its prognostic impact, surprisingly, MRD is still not widely used in routine clinical practice. Some claim that additional clinical trials are needed before there is widespread acceptance of MRD as routine in the clinic. The stringency of this request is exceptional and was never applied to other very useful routine tests such as the serum free light chains. For now, MRD results should not be considered as ultimate deciders for therapeutic decisions, but rather be considered as an augmentative knowledge about a disease state for a given patient. Current MRD assays solely represent an improvement in technology that allows more accurate measurements than was previously done with other less-sensitive assays. As an example, some groups have demonstrated that CR patients who remain MRD-positive have similar outcomes as patients achieving partial response (6, 7, 8).
It is important to note, that MRD negativity does not imply absence of disease – it is impossible to prove a negative. But as technology evolves, we will most likely be able to detect disease at even greater sensitivities. Sustainability of MRD negativity seems to be an even better predictor of outcome. However, taken to the extreme the argument is tautologic (circular) as it simply reports on patients who have extraordinary outcomes (e.g., yearly sustained MRD negativity for 10 years). Becoming MRD negative at the first point of measurement is a desirable clinical outcome, and preferrable than remaining positive. But patience is important as various studies now show that later conversions to MRD negativity may be just as valuable. And of course, a word of caution, we all know of patients who can have long term disease control even with measurable residual disease. But bear in mind this last statement can only be done retrospectively.
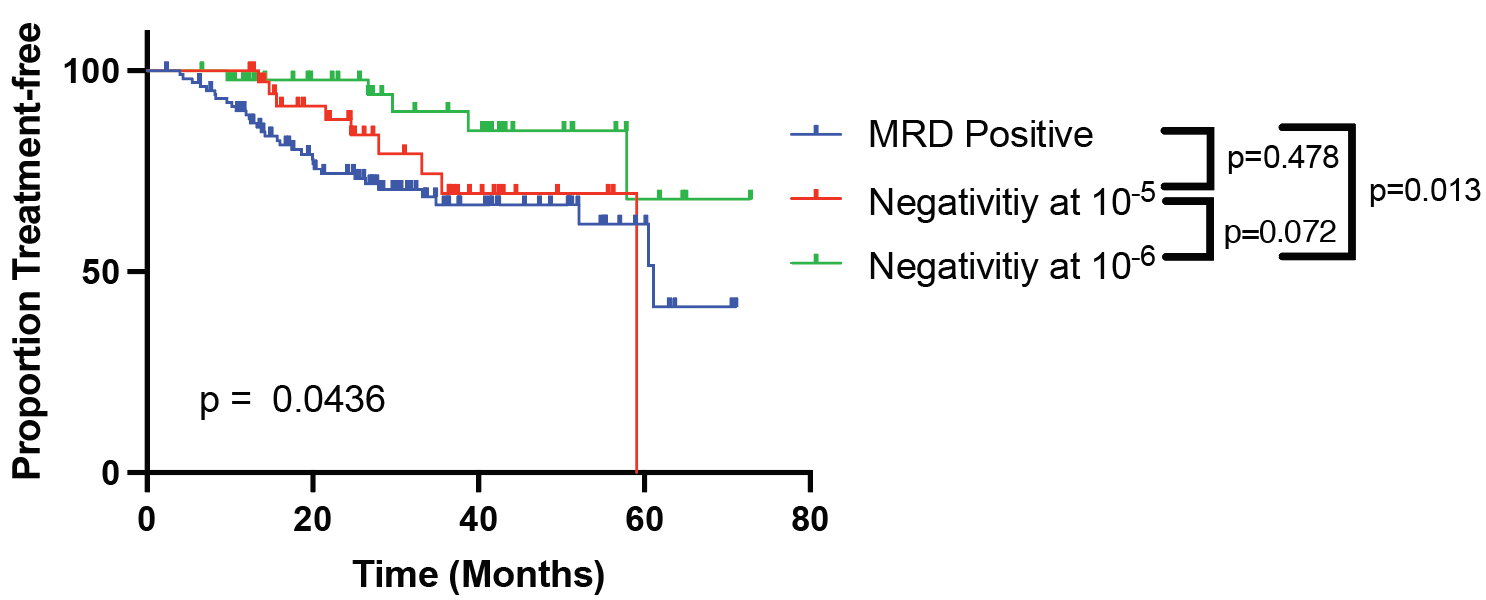
MRD testing has been incorporated as standard of care for other hematological malignancies, such as in ALL, where negativity at 10-6 following induction/transplant is associated with extraordinary survival outcomes, and seems to be an ultimate threshold for disease “cure” (9, 10). Our findings exhibit that in MM, said threshold has not yet been reached – but we are on our way. We demonstrate that achieving negativity at 10-6 offers a significant advantage over achieving negativity at lower threshold, including 10-5. We have documented that the deeper the response, the longer the time to progression. It has previously been shown that with each log-reduction in disease, there is a proportional advantage in survival outcomes (11). Having said that, a minority of patients can still relapse even after achieving MRD negativity at 10-6, showing that 10-6 may still not be the ultimate universal threshold. The right clinical behavior after resurgence of disease from MRD negativity is still a matter of debate, and needs to be studied further.
A potential limitation of MRD testing for MM is its reliance on local sampling, and the possibility of heterogeneous marrow involvement, as well as the technique of sample procurement. Imaging might alleviate such concerns? In our cohort, we show that PET-CT offers little added benefit to MRD testing, given that almost none of the patients who achieved MRD-negativity had positive PET-CT scans. The interobserver variability, and the subtle nature of changes observed with PET CT also adds to the uncertainty of value derived of functional imaging analysis. To overcome these limitations other blood-based methods might be incorporated into the future. Peripheral blood MRD NGS testing is possible but has a lower sensitivity (1-2 logs). Developments in mass spec protein identification (i.e., MALDI-TOF or LC-mass spec) and quantification may further aid in this process, but those are only available at selected academic centers.
Our findings further validate attainment of MRD negativity as the most important prognostic factor in myeloma - the deeper the response the better the outcome. Only with improved technologies and extended follow-up will we be able to determine the ultimate threshold quasi synonymous with a predictable cure. In the meantime, achieving negativity at 10-6 should be the ultimate goal of therapy, by all safe means possible. Many questions remain, including using MRD-negativity as an endpoint for consolidation and/or maintenance therapy, and as a finite boundary allowing patients to live free of the burden of chronic maintenance.
As more data is generated other applications for MRD testing will emerge. But at the minimum MRD negativity has beyond dispute prognostic significance. Furthermore, MRD negativity is “actionable.” A clinician actions are much more than prescribing therapy. A conversation (verb) with a patient who has achieved MRD negativity is of high mutual regard- joy. And in selected cases it can contribute to the decision to discontinue burdensome therapy.
There is no denying the clinical significance of MRD testing, and its ready to start its primetime! Lastly, as we all want to accelerate the process by which drugs reach patients, the FDA should finally consider MRD negativity a validated surrogate markers for clinical trials.
References
- Kumar S, Paiva B, Anderson KC, Durie B, Landgren O, Moreau P, et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. The Lancet Oncology. 2016;17(8):e328-e46.
- Ching T, Duncan ME, Newman-Eerkes T, McWhorter MME, Tracy JM, Steen MS, et al. Analytical evaluation of the clonoSEQ Assay for establishing measurable (minimal) residual disease in acute lymphoblastic leukemia, chronic lymphocytic leukemia, and multiple myeloma. BMC Cancer. 2020;20(1):612.
- Munshi NC, Avet-Loiseau H, Rawstron AC, Owen RG, Child JA, Thakurta A, et al. Association of Minimal Residual Disease With Superior Survival Outcomes in Patients With Multiple Myeloma. JAMA Oncology. 2017;3(1):28.
- Munshi NC, Avet-Loiseau H, Anderson KC, Neri P, Paiva B, Samur M, et al. A large meta-analysis establishes the role of MRD negativity in long-term survival outcomes in patients with multiple myeloma. Blood Adv. 2020;4(23):5988-99.
- Landgren O, Devlin S, Boulad M, Mailankody S. Role of MRD status in relation to clinical outcomes in newly diagnosed multiple myeloma patients: a meta-analysis. Bone Marrow Transplant. 2016;51(12):1565-8.
- Paiva B, Puig N, Cedena MT, Rosinol L, Cordon L, Vidriales MB, et al. Measurable Residual Disease by Next-Generation Flow Cytometry in Multiple Myeloma. J Clin Oncol. 2020;38(8):784-92.
- Paiva B, San-Miguel J, Avet-Loiseau H. MRD in multiple myeloma: does CR really matter? Blood. 2022;140(23):2423-8.
- Perrot A, Lauwers-Cances V, Corre J, Robillard N, Hulin C, Chretien ML, et al. Minimal residual disease negativity using deep sequencing is a major prognostic factor in multiple myeloma. Blood. 2018;132(23):2456-64.
- Berry DA, Zhou S, Higley H, Mukundan L, Fu S, Reaman GH, et al. Association of Minimal Residual Disease With Clinical Outcome in Pediatric and Adult Acute Lymphoblastic Leukemia. JAMA Oncology. 2017;3(7):e170580.
- Maloney KW, Devidas M, Wang C, Mattano LA, Friedmann AM, Buckley P, et al. Outcome in Children With Standard-Risk B-Cell Acute Lymphoblastic Leukemia: Results of Children’s Oncology Group Trial AALL0331. Journal of Clinical Oncology. 2020;38(6):602-12.
- Rawstron AC, Gregory WM, de Tute RM, Davies FE, Bell SE, Drayson MT, et al. Minimal residual disease in myeloma by flow cytometry: independent prediction of survival benefit per log reduction. Blood. 2015;125(12):1932-5.
Follow the Topic
-
Blood Cancer Journal
This journal seeks to publish articles of the highest quality related to hematologic malignancies and related disorders.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in