Modulation of subthalamic beta oscillations by movement, dopamine and deep brain stimulation in Parkinson’s disease
Published in General & Internal Medicine
Parkinson’s disease (PD) is the most common movement disorder with the most pronounced symptoms being bradykinesia (i.e. slowness of movement) and rigidity (muscle stiffness), as well as tremor, postural instability and gait difficulties. The cause of PD lies in the loss of dopaminergic neurons in the substantia nigra, within the basal ganglia. PD is typically treated with dopaminergic medication. With increased motor fluctuations several years into the disease selected patients undergo Deep Brain Stimulation (DBS). DBS involves implanting two electrodes within a target region of the basal ganglia, which are connected to an implantable-pulse generator (IPG) placed below the clavicle. The most common DBS target for PD patients in the subthalamic nucleus (STN), a structure crucial for motor control.
Electrodes from the STN give us the opportunity not only to administer stimulation, but also to record Local Field Potentials (LFP). Subthalamic LFP recordings have revealed that beta band activity (13-35 Hz) is substantially exaggerated in patients with PD, while studies in the past two decades demonstrated that treatment (medical or DBS) reduce this activity. Notably, this reduction correlates with alleviation of parkinsonian motor symptoms, i.e. bradykinesia and rigidity. On a similar note, beta band activity significantly decreases during movement. For this reason, subthalamic beta is a robust biomarker for parkinsonian symptoms, and is more recently employed as a feedback signal for closed-loop DBS. In contrast to conventional DBS, which administers electric current continuously, closed-loop DBS uses a real-time electrophysiological signal and administers current only when this signal exceeds some predefined thresholds. This has substantial therapeutic benefits, as it potentially minimizes DBS-induced side effects (e.g. dyskinesia) and preserves the IPG battery.
Although beta band activity is a reliable predictor for parkinsonian symptoms, little is known about the different intertwined factors that modulate it. Moreover, most studies have been conducted in the acute post-operative state, when important confounds might not give a complete picture of the oscillatory dynamics in question. Here we studied the interaction of dopaminergic medication, DBS, and voluntary movement on subthalamic beta in seven PD patients (12 STNs) implanted with DBS 3-12 months after the implantation. Recordings were performed at rest, and during a repetitive finger tapping task in the following four conditions: after withdrawal of dopaminergic medication and DBS, with medication only, with DBS only, and with simultaneous medication and DBS. A triaxial accelerometer was placed on the index finger of the patients, in order to quantify movement. LFPs were acquired via the PERCEPT PC IPG with a sampling frequency of 250 Hz. Morlet wavelet (8 cycles, sampling rate = 20 Hz) was performed for signal transformation to the frequency domain. Paired-permutation tests and Linear mixed effects models were employed for statistical analysis.
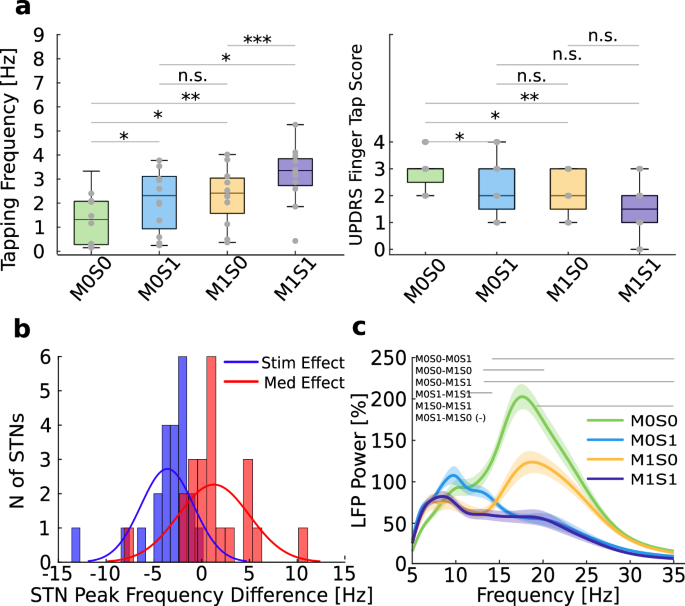
We found the dopaminergic medication and DBS improved motor performance, as measured with tapping frequency, with a similar effect size, while the combination of both treatments resulted in the optimal motor effect (Figure 1a). However, the two treatments suppressed different components of the beta band both during rest, and movement; dopaminergic medication primarily suppressed low beta (13-20 Hz), while DBS was associated with a broader decrease (Figure 1b-d). Movement suppressed beta in all four conditions and this decrease was larger with simultaneous medication and DBS. Last, beta band activity during movement correlated with motor performance within patients across all four states (Figure 1e).
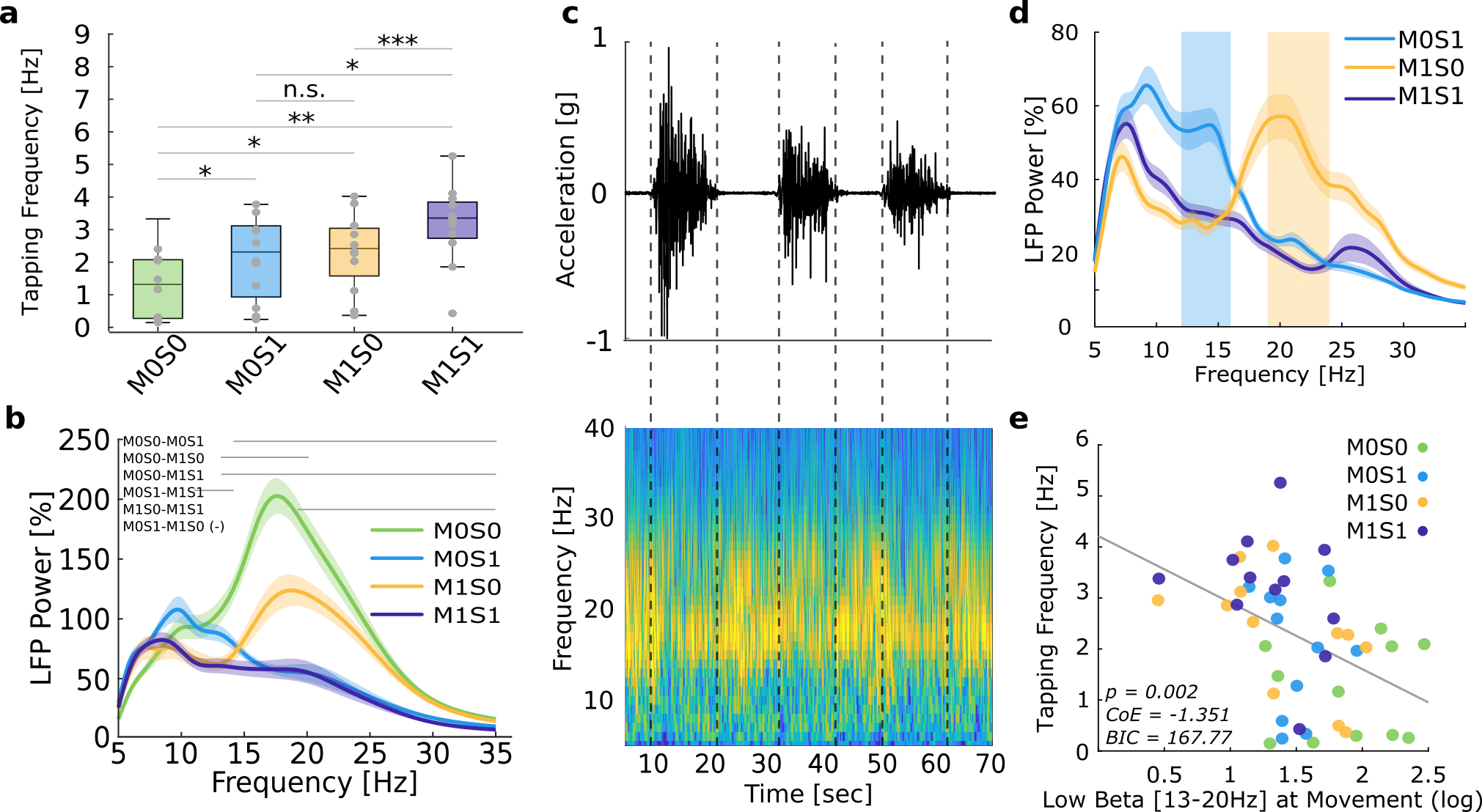
Figure 1. Medication, DBS, and movement modulate subthalamic beta band in PD. a Tapping frequency with paired-wise comparisons in four different conditions * p < 0.05, ** p < 0.01, *** p < 0.001, n.s.: not significant. b Averaged power spectra during rest across four recorded conditions. Horizontal lines indicate the statistically significant clusters between states. Shaded areas show the standard error of the mean. c Example case of desynchronization of beta band activity during repetitive finger tapping. Upper plot shows the movement trace as recorded from the accelerometer during three 10-second blocks of repetitive finger tapping (indicated by grey dotted lines), with 10 seconds of rest in between. The movement trace is synchronized to the neural activity (bottom spectrogram), which shows a notable desynchronization especially in the high beta component during movement. d Complementary effect of DBS and medication (orange and blue lines) when compared to the combined state (purple line) during movement. Shaded areas show the statistically significant cluster between these conditions. e Scatter plot showing the relation between low beta band activity during movement and motor performance across all conditions. M0S0: off-medication/off-DBS; M0S1: off-medication/on-DBS; M1S0: on-medication/off-DBS; M1S1: on-medication/on-DBS.
The differential modulation of beta band activity by medication and DBS suggest that the different beta components are linked to distinct neurophysiological mechanisms; low beta is mainly linked to the hypodopaminergic status, while high beta might be part of a physiological mechanism that promotes movement. Future studies could investigate whether persistent suppression of the broader beta band by DBS might lead to known DBS-induced side-effects, such as difficulties in motor inhibition. Nevertheless, the two therapies have a complementary effect with therapeutic benefits. Importantly, we show that subthalamic beta activity during movement significantly correlated with motor performance across all conditions, an important finding for future closed-loop DBS paradigms, which should aim at targeting beta band activity during treatment, as well as during rest and movement.
Varvara Mathiopoulou, Doctoral Candidate
Movement Disorders & Neuromodulation Section
Charité – Universitätsmedizin, Berlin
Follow the Topic
-
npj Parkinson's Disease
This journal publishes original basic science, translational and clinical research related to Parkinson's disease, including anatomy, etiology, genetics, cellular and molecular physiology, neurophysiology, epidemiology and therapeutic development and treatments.
Related Collections
With Collections, you can get published faster and increase your visibility.
The neuroimmune-axis and ageing in Parkinson’s Disease
Publishing Model: Open Access
Deadline: Jul 15, 2026
Cognition - preclinical models, and preclinical unmet need
Publishing Model: Open Access
Deadline: Jul 27, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in