Mutations in the efflux pump regulator MexZ shift tissue colonization by Pseudomonas aeruginosa to a state of antibiotic tolerance
Published in Microbiology and Biomedical Research
Antibiotic treatment of bacterial infections is increasingly complicated by the development and spread of antimicrobial resistance (AMR). Long-term management of infected patients often involves monitoring resistance development through standardized laboratory antibiotic susceptibility assays conducted in clinical microbiology laboratories. However, our study reveals that antibiotic susceptibility can be conditional, with a proportion of bacterial variants in infected individuals evading antibiotic eradication through mechanisms not routinely tested in clinical practice. This highlights the importance of understanding how bacterial interactions with host tissues influence antibiotic efficacy, for optimizing infection management strategies and developing targeted therapeutics.
In this study, we specifically focused on Pseudomonas aeruginosa, a notorious human pathogen known for its resistance against antibiotics and its ability to establish chronic infections, particularly in people with cystic fibrosis (pwCF). Its high clinical relevance and propensity for acquiring AMR via various mechanisms, primarily chromosomal mutations, make it a top concern. Despite extensive efforts to combat P. aeruginosa infections, treatment remains challenging, with chronic lung infections often resulting in increased morbidity and mortality for affected individuals.
Multidrug efflux pumps play a central role in the AMR phenotype of P. aeruginosa, serving as key determinants of resistance, especially when overproduced due to loss-of-function mutations in their negative regulators. These protein complexes, located in the bacterial membrane, actively extrude a wide range of antibiotics outside the cell, contributing to P. aeruginosa's resistance to antimicrobial agents. Additionally, efflux pumps can impact bacterial physiology and virulence by modulating the extrusion of different types of compounds, including quorum sensing (QS) signalling molecules or their precursors, which regulate the expression of genes encoding different virulence factors.
Mutations in mexZ, encoding the negative regulator of the expression of mexXY efflux pump genes, are frequently acquired by P. aeruginosa during chronic infections. MexXY-OprM’s most prevalent substrates are aminoglycoside antibiotics like tobramycin, commonly used to treat P. aeruginosa infections. Consequently, mutations in mexZ have been traditionally considered to be selected for due to its contribution to tobramycin resistance through the associated mexXY overexpression. However, despite being highly prevalent in clinical isolates, mexZ mutations are associated with low levels of tobramycin resistance in standard laboratory tests. In fact, an analysis conducted at Rigshospitalet (Copenhagen, Denmark) and the Technical University of Denmark, revealed that among 500 P. aeruginosa isolates obtained from young early chronically infected pwCF, 40% of them carried mutations in mexZ, but only 7% of those mutants showed clinically relevant resistance to tobramycin.
Why are mexZ mutations so frequently acquired by P. aeruginosa during chronic respiratory infection despite only slightly increasing phenotypic AMR? This discrepancy made us hypothesise mexZ mutations to provide alternative adaptive advantages beyond straightforward AMR, which led us to uncover novel insights into the adaptation of this pathogen during chronic infection.
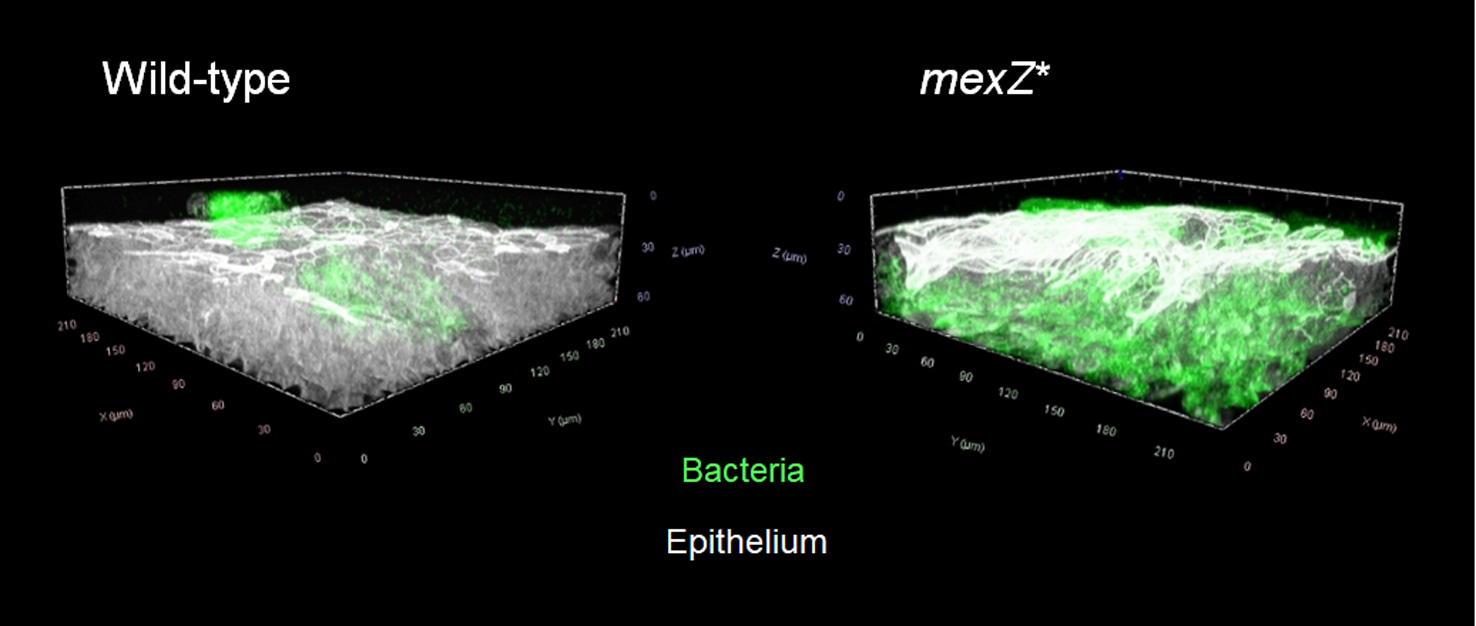
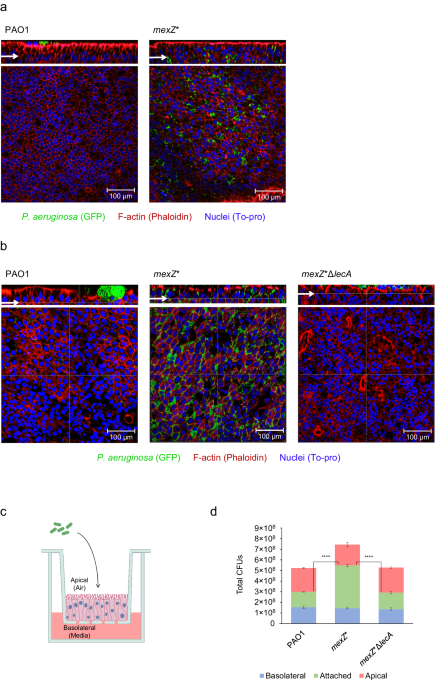
We investigated the colonization strategy of a mexZ mutant by using an in vivo-mimicking human airway epithelium infection model, revealing this mutation to induce changes in the infection behaviour of this bacterium. Notably, the mexZ mutant displayed enhanced tissue invasion capabilities, leading to an increased intraepithelial colonization. We observed that this behaviour confers protection from antibiotic eradication, as the intraepithelial compartment is impermeable to many antibiotic classes. This change in colonization and associated shielding from treatments may potentially explain the high frequency of mexZ mutations in clinical isolates from early timepoints of infection, since it could be contributing to treatment failure and persistence of P. aeruginosa infections.
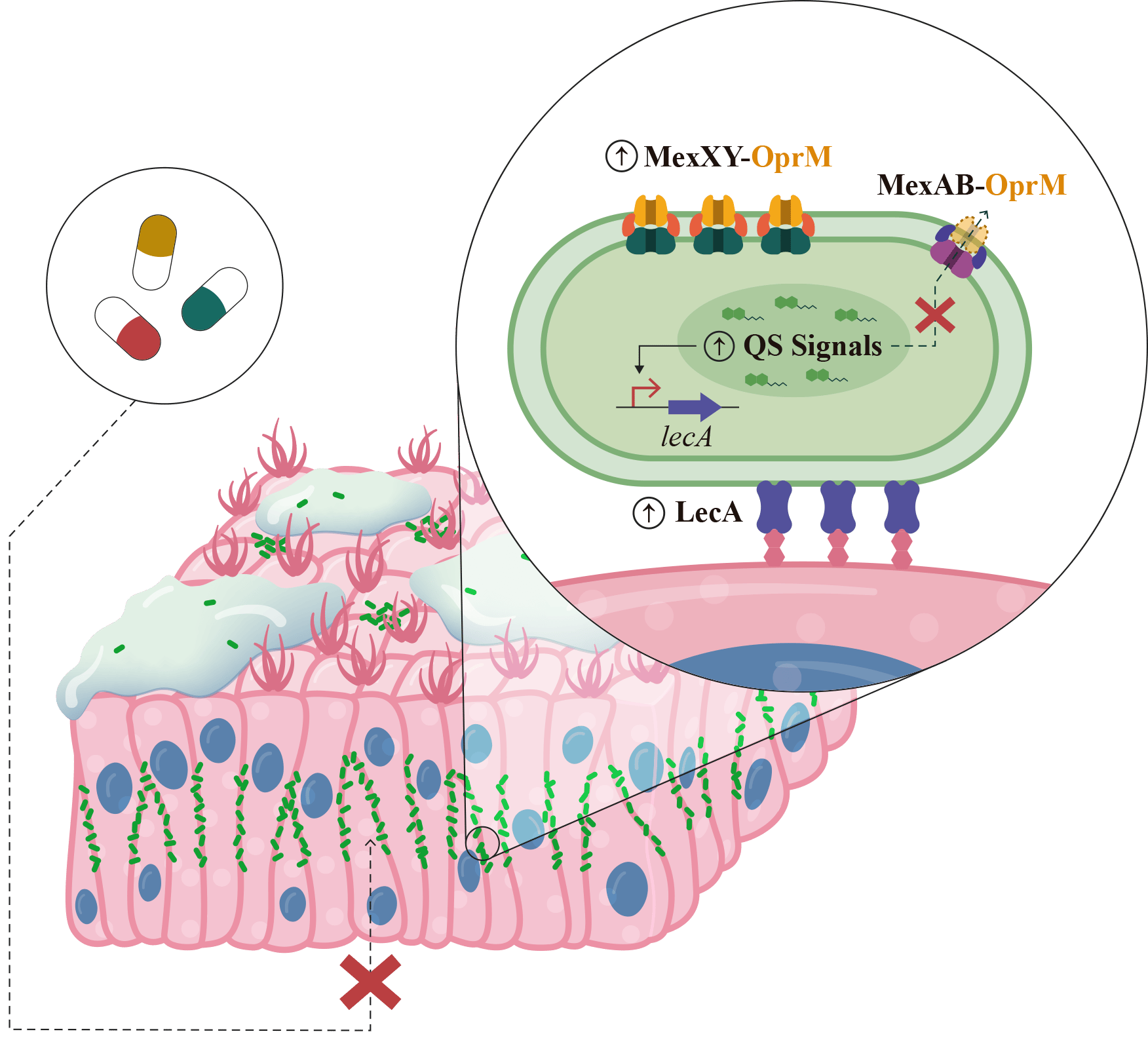
Additionally, our study elucidates the molecular mechanism underlying this intriguing phenotype associated with mexZ mutations. The overproduction of the MexXY-OprM efflux pump collaterally disrupts QS signalling, resulting in the upregulation of lecA, a QS-regulated gene encoding a lectin crucial for P. aeruginosa attachment to human tissues. This is rooted in the competitive dynamics for the porin OprM, shared by MexXY and MexAB efflux pumps, being the latter involved in the extrusion of QS signals. The mexZ mutation-associated overproduction of MexXY-OprM reduces the amount of MexAB-OprM, altering the extrusion of QS signals through this efflux pump.
Overall, our study provides novel insights into the complex relationship between AMR and bacterial colonization strategies in P. aeruginosa infections. mexZ mutations, while associated with low levels of AMR under standard laboratory conditions, confer additional adaptive advantages that potentially promote bacterial persistence and treatment failure. By elucidating the molecular mechanisms underlying these adaptive responses, we gained a deeper understanding of the challenges posed by P. aeruginosa infections and the need for comprehensive strategies to combat AMR. Our observations may precede more studies also pointing to integrating knowledge of host-bacteria interaction dynamics and bacterial adaptive responses into diagnostic and treatment approaches as a crucial step forward for improving outcomes in patients with infectious diseases.
Heading illustration made by Elena Contel Maza (@helen.mushu.science in instagram)
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in