Peptidoglycan Saccharides from gut bacteria play a protective role in gut health
Published in Chemistry and Microbiology
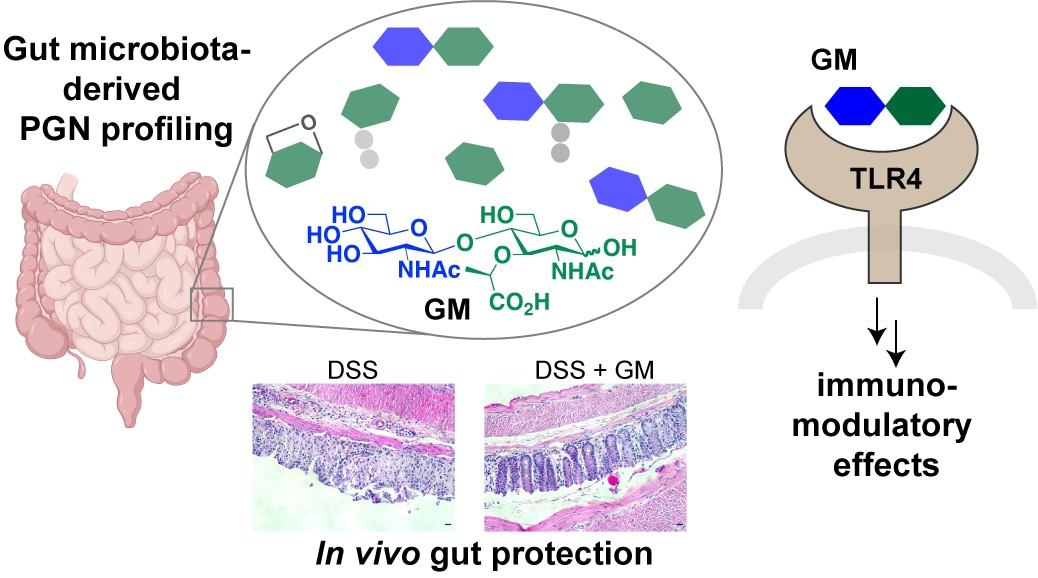
As the natural habitat for trillions of microorganisms, the mammalian gut is often regarded as an extraordinary interface for microbial-host crosstalk. As chemical biologists, we are particularly drawn to understanding how the gut microbiota communicates with the host at the molecular level.
Our lab focuses on gut bacteria–derived peptidoglycan fragments (PGNs), soluble components of the peptidoglycan polymer that represent a unique and conserved feature of the bacterial cell wall absent in mammalian cells. Intriguingly, the ubiquitous presence of natural PGNs in mammalian serum and gut has been demonstrated using ELISA with a PGN-specific antibody (see study by Wang and coworkers) and recently through the administration of radiolabeled PGNs (see study by Boneca and coworkers), supporting the biological significance of gut microbiota-derived PGNs in hosts. Despite the growing recognition of gut microbiota–derived PGNs as emerging gut effector molecules, the fundamental question of the exact structures of PGNs present in the host body has yet to be addressed. Determining the structures of natural PGNs could be a game-changer in the field, opening the door to further biological insights.
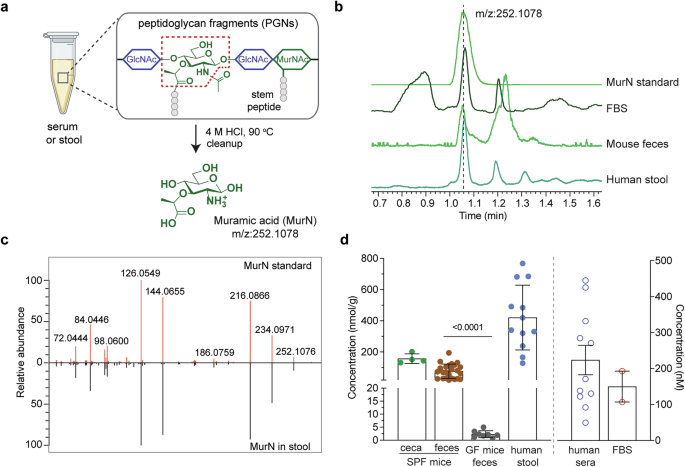
Grateful to have started my independent lab at NTU in 2019 with generous start-up funding from the National Research Foundation Fellowship (NRFF), I made the largest purchase of my life at that time: an Orbitrap Exploris 120 mass spectrometer, dedicated to PGN analysis studies. Our first goal is to develop a high-resolution LC-MS/MS workflow for structural elucidation of natural PGNs in the host. After numerous trials and optimizations, we established two LC-MS/MS pipelines: a MurN-based quantification method of PGNs, as well as a workflow for profiling various PGN subtypes. To validate our LC-MS methods for PGN discovery and identification, we tested them on standard PGNs, as well as on samples from mice and humans. In a pilot study approved by the IRB, we aimed to recruit 20 healthy human volunteers to provide stool and serum samples for PGN analysis. As a newly minted PI, I was warmed by the willingness of my lab members and their friends to participate by donating stool and blood samples, which eased our sample collection process and contributed meaningfully to the study.
With the unveiling of natural PGN subtypes in the host using our LC-MS/MS pipeline, we discovered an abundance of PGN-derived saccharide-only motifs, including N-acetylglucosamine-β1,4-N-acetylmuramic acid (GM) and N-acetylmuramic acid (M), which constituted over 90% of the total PGN pool in the healthy gut. To confirm the structural identity of these saccharide PGNs in the gut, we rightfully proceeded to chemically synthesize standard compounds—a notable achievement given the inherent difficulty of carbohydrate synthesis. Our robust and large-scale synthesis of natural PGNs paves the way for further biological studies.
The presence of natural PGN saccharides in the host gut is particularly intriguing, as these molecules do not activate the mammalian NOD1 and NOD2, the canonical innate immune sensors for bacterial PGNs. Nevertheless, instead of outright dismissing these saccharides as inactive bystander molecules within the host, we chose to embark on an uncharted path to explore their potential roles. Our initial observations were pleasantly surprising: the disaccharide GM is immunologically active—it modestly stimulates proinflammatory cytokine production in murine macrophages and monocytes and upregulates CD80/86 surface expression in dendritic cells, which are completely unexpected findings! After months of repeated experiments and diverse assays to validate the reproducibility of the results, we proceeded to the next bigger question: if not NOD1 or NOD2, then what is the receptor in host cells that mediates GM’s effects?
In collaboration with immunology colleagues, we tested a panel of TLR inhibitors to evaluate their ability to block GM-induced cytokine production in immune cells. Among the inhibitors tested, only the specific TLR4 inhibitor (TAK-242) effectively blocked GM’s effects. Retrospectively, this observation marked a key point that guided the direction of our subsequent research; however, at the time, we were highly skeptical of our results, particularly because TLR4 is a well-established sensor of bacterial LPS, not PGN. To gather more evidence and validate our findings, we procured TLR4 knockout cell lines and mice—an important (and costly) step for our young chemical biology lab to commit and delve deeper into immunology. Utilizing TLR4−/− BMDMs, we confirmed that GM-induced cytokine activation is mediated through TLR4 signaling. To demonstrate direct binding between GM and TLR4, we performed both in vitro pulldown assays and SPR binding analyses, providing key biochemical evidence of direct ligand–protein interaction. Moreover, we employed site-directed mutagenesis to reveal potential residues in the TLR4 ectodomain in GM recognition. Concurrently, we synthesized a series of structural analogues of GM to investigate the structural requirements of disaccharide ligands for TLR4 activation.
Despite multiple lines of experimental evidence supporting GM as a TLR4 ligand, its mild immunological activity still drew critical skepticism from reviewers concerning potential endotoxin contamination in our synthetic GM sample. The enduring publishing process also served as a valuable learning experience, through which we learned that polymyxin B titration and LAL detection assays can be used to rule out endotoxin contamination., these assays conclusively addressed the contamination concerns, validating our discovery of the gut microbiota–derived disaccharide GM as a natural ligand of the host TLR4 receptor that elicits mild immunological responses.
Does the in vitro finding of GM as a TLR4 ligand have any in vivo implications? To explore this, we investigated whether GM affords any protective effects against the DSS-induced colitis model in mice. First of all, we showed that daily intraperitoneal administration of GM does not trigger inflammation in healthy mice alone; nevertheless, it effectively mitigates colonic inflammation in wild-type mice with DSS-induced colitis, in terms of immune cell infiltration, proinflammatory marker expressions, and histological analysis. Importantly, the protective effects of GM were abolished in TLR4-/- mice, consistent with our in vitro results of GM as a TLR4 ligand. Overall, these in vivo findings underscore the therapeutic potential of natural gut microbiota–derived disaccharides in alleviating intestinal inflammation in the host.
Looking back, this work has been five years in the making, during which our young lab established expertise across multiple disciplines, including analytical chemistry, synthetic chemistry, biochemistry, immunology, and animal models. While it is gratifying to see this work finally published and to share our findings with the community, we are excited about what’s coming next as we delve deeper into understanding the molecules underlying gut microbiota–host crosstalk.
https://www.nature.com/articles/s41467-025-60678-5
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in