Photoswitchable gating of non-equilibrium enzymatic feedback in chemically communicating polymersome nanoreactors
Published in Chemistry
One of the most captivating questions concerning the interface between chemistry and biology is “How did inert matter transition into living matter?” Current living matter is far more evolved than primitive life forms: it is thought that the first cells did not contain proteins and were composed mainly of lipids, amino acids, and nucleic acids.1 It is intriguing to consider whether it is possible to engineer alternative life forms (or observe life-like behaviour) using alternative chemical building blocks. Successful recreation of behaviours like energy transformation, minimalistic metabolism, replication, growth, division, and “rudimentary” logic functions2 could facilitate experimental abstraction of the physical requirements of living systems.
In this work, we chose to emulate the circadian rhythm’s modulation of living organism metabolism in an inanimate matter system. We were specifically inspired by dynamic oscillations of non-equilibrium metabolite signalling in response to the day and night cycle, which are fundamental to achieving homeostasis and other physiological functions.
When we started this project, the first examples of hollow spheroidal bilayer membrane ensembles (polymersomes) capable of modulating (bio)catalytic rates in response to external stimuli were appearing in the literature.3 Such transitions are of high importance in living systems: in the presence of stimuli, cells adapt their functions to out-of-equilibrium states and when stimuli are removed, cells return to basal equilibrium conditions. In one of the examples, polymersome membranes were functionalised with Donor Acceptor Stenhouse Adducts (DASA)4 which, in the presence of light, isomerise from a non-polar, colourful triene-enol to a more polar, colourless cyclopentenone (Figure 1A). This isomerisation permeabilizes polymersome membranes to water-soluble fuel molecules, which are then transformed into products by encapsulated enzymes. In cells, the formation of products occurs through similar, albeit more complex, processes that that are also regulated by hierarchical feedback loop networks. As a step towards realisation of inanimate matter systems that imitate non-equilibrium processing in living cells, we asked ourselves how to engineer self-modulation into the light-responsive DASA-polymersome systems. We concluded that this might be achieved by encapsulating enzymes that produce coloured products which match the absorbance of the DASA in the polymersome membrane. These products would then compete for light absorption throughout the catalytic process. Accordingly, we encapsulated an esterase enzyme into the polymersome (DASA nanoreactor). This enzyme transforms esters into acids in the presence of a pH pigment that transitioned to a photomask. Throughout exposure to light, we observed an increase in photomask formation which was gradually interrupted (Figure 1B). We attributed this phenomenon to the inhibition of light absorption by the DASA-polymersomes leading to resealing of the membrane and blocking of the ester influx, which indicated that a light-mediated negative feedback loop had been established.
Though we added a new level of regulation to these cell-like systems, natural cells are capable of responding to stimuli multiple times. Therefore, we built a second polymersome nanoreactor with an intrinsically permeable membrane and encapsulated urease (urease nanoreactor, Figure 1C). In the presence of urea, urease produces ammonia under acidic conditions, which results in an increase in pH and switches off the enzyme in mild alkaline conditions. Urease becomes active again when the medium is re-acidified (Figure 1D), providing a pH-mediated negative feedback loop.
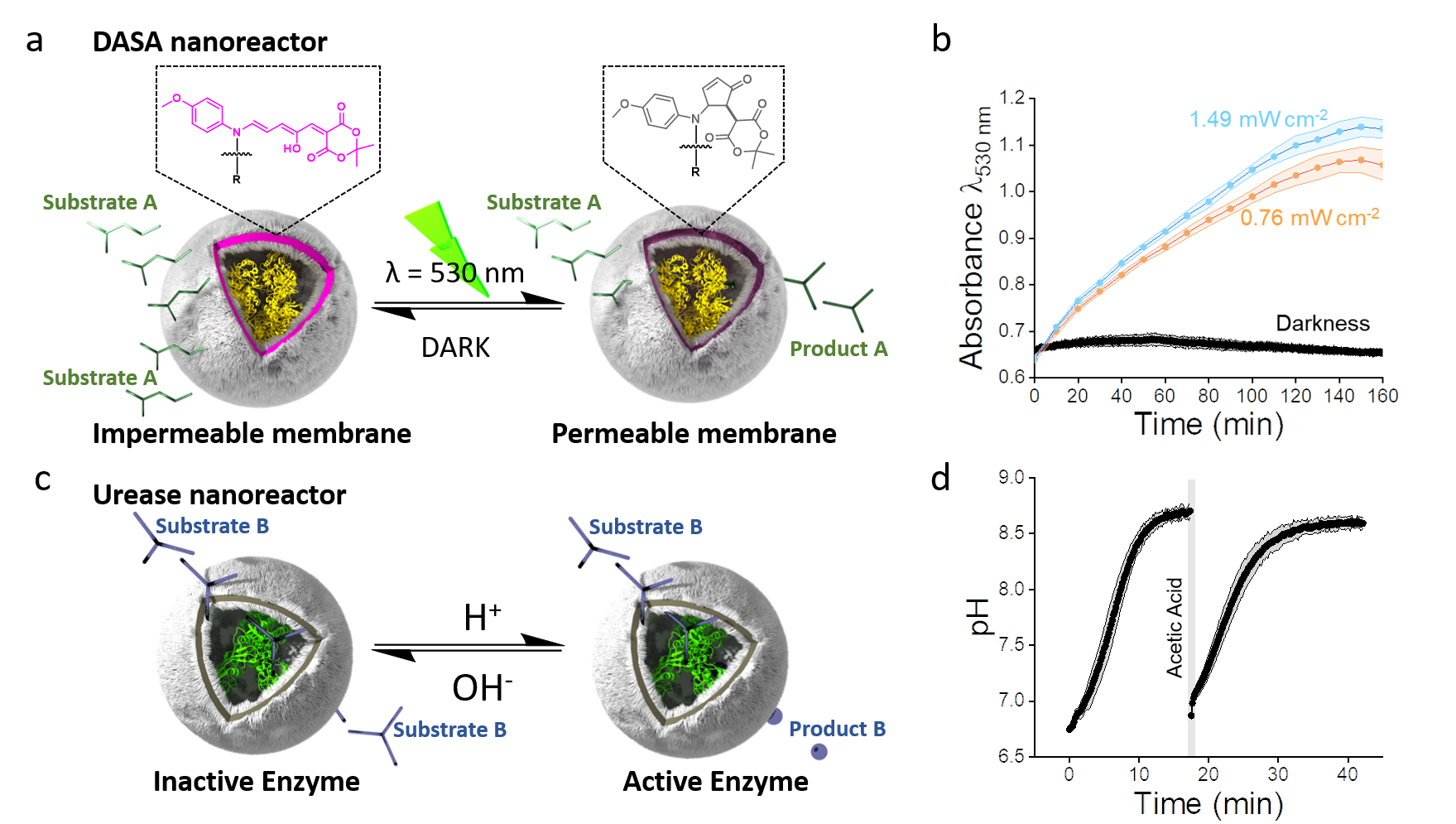
To imitate how circadian rhythms regulate metabolic rates in response to sunlight, we exposed our two antagonistic polymersome nanoreactors to alternating green light and darkness cycles (Figure 2A). In the absence of light, the system remained at an equilibrium basal level corresponding to alkaline conditions (Figure 2B). This was expected, since the DASA nanoreactors are not permeable in darkness. In contrast, in the presence of light, the DASA system was activated, leading to ester influx, gradual acidification of the medium and subsequent accumulation of the photomask, which attenuates the light reaching the DASA. Over time, the acidification process was interrupted by the antagonistic formation of hydroxide ions in the urease nanoreactors such that the system reached a plateau in which the formation of acid was equivalent to the formation of base. Importantly, higher light intensities modulated the plateau so that it occurred at more acidic conditions. When the system was again subjected to darkness, the non-equilibrium activity of the DASA nanoreactors was interrupted whilst the urease nanoreactors continued to produce hydroxide ions. As expected, the system then returned to the equilibrium basal level (alkaline conditions) at which the urease activity was self-inhibited. Overall, our results demonstrated the formation of a feedback-regulated reaction network that enabled communication between two nanoreactors under non-equilibrium conditions.
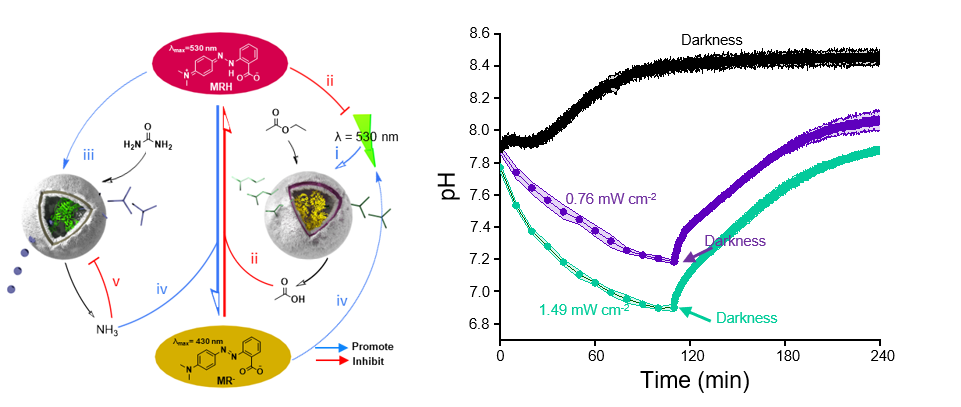
In addition, we employed the fluctuations of pH to generate chemomechanical work by cyclically swelling and deswelling a pH-responsive hydrogel as a rudimentary representation of the modulation of tissue matrices by the circadian rhythm. Importantly, we achieved this in biologically relevant buffers, which opens an avenue to modulating cellular behaviour5 and establishing cell/nanoreactor communication networks. Our paper demonstrates that polymersomes are an excellent platform to generate experimental protocell models from inanimate matter and to abstract chemical bias from the roles of essential biomolecules in the construction of living entities.
For the full story of our research, please see the article published in Nature Chemistry (https://www.nature.com/articles/s41557-022-01062-4).
References
- Gilbert, W., Origin of life: The RNA world. Nature 1986, 319 (6055), 618-618.
- Gao, N.; Li, M.; Tian, L.; Patil, A. J.; Kumar, B. P.; Mann, S., Chemical-mediated translocation in protocell-based microactuators. Nat. Chem. 2021, 1-12.
- a) Rifaie-Graham, O.; Ulrich, S.; Galensowske, N. F.; Balog, S.; Chami, M.; Rentsch, D.; Hemmer, J. R.; Read de Alaniz, J.; Boesel, L. F.; Bruns, N., Wavelength-Selective Light-Responsive DASA-Functionalized Polymersome Nanoreactors. J. Am. Chem. Soc. 2018, 140, 8027−8036; b) Rifaie-Graham, O.; Galensowske, N. F. B.; Dean, C.; Pollard, J.; Balog, S.; Gouveia, M. G.; Chami, M.; Vian, A.; Amstad, E.; Lattuada, M.; Bruns, N., Shear Stress-Responsive Polymersome Nanoreactors Inspired by the Marine Bioluminescence of Dinoflagellates. Angew. Chem., Int. Ed. 2021, 60 (2), 904-909.
- Stricker, F.; Sanchez, D. M.; Raucci, U.; Dolinski, N. D.; Zayas, M. S.; Meisner, J.; Hawker, C.; Martínez, T.; Read de Alaniz, J., A multi-stage single photochrome system for controlled photoswitching responses. Nat. Chem. 2022, 1-7.
- Mager, M. D.; LaPointe, V.; Stevens, M. M., Exploring and exploiting chemistry at the cell surface. Nat. Chem. 2011, 3 (8), 582-589.
Follow the Topic
-
Nature Chemistry
A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in