Suppression of lysosome metabolism-meditated GARP/TGF-β1 complexes specifically depletes regulatory T cells to inhibit breast cancer metastasis
Published in Cancer
Background and Introduction:
Regulatory T cells (Tregs), a subset of CD4+ T cells, play a critical role in maintaining immune homeostasis and self-tolerance by suppressing self-reactive T cells, thus preventing autoimmunity. However, an overactive function of Tregs can contribute to cancer progression, particularly by inhibiting antitumor immune responses, which has been correlated with a poor prognosis in clinical settings.
In our initial experiments, we synthesized naphthalenelactam-platinum complexes (NPt) by coordinating naphthalenelactam with cisplatin, which are potent and selective inhibitors of the extra-terminal domain (SET). These complexes have demonstrated selectivity for the first bromodomain and have shown potential for high efficiency in therapies against triple-negative breast cancer (TNBC) and in the study of Treg biology.
The differentiation and function of Tregs are dependent on the transcription factor FOXP3, whose stable expression is a hallmark of fully differentiated Tregs. Despite this, the lack of a specific surface marker presents a significant challenge in the depletion of human Tregs. Transforming growth factor beta 1 (TGF-β1), found on the surface of Treg cells, is typically in an inactive form bound to the membrane protein GARP. The mechanism by which it becomes activated remains to be fully elucidated. Additionally, metabolic programs are known to be crucial for Treg stability and function.
Our research, which could be categorized under the keywords "Metabolism", "Immune-therapy" ,"Molecular mechanism" and "Pt-based anti-tumor metallodrugs", has raised intriguing questions: Is there a link between metabolism and the differentiation of Tregs? And does the TGFβ1 signaling pathway play a role in this process?
Lysosomes, essential organelles in Treg metabolism and immune tolerance regulation, are known to provide necessary substances for the transcription and stability of Tregs. However, the mechanisms that inhibit the production of active TGF-β1 within Treg cell metabolism are not yet clear.
The TGF-β pathways are known to be significant in TNBC, epithelial–mesenchymal transition (EMT), and metastasis. Cells responding to TGF-β1 reprogram their antioxidant metabolism, reducing the effectiveness of anticancer therapeutics, including platinum-based chemotherapy. This resistance to anti-TNBC therapy can lead to tumor recurrence. The blockade of BRAFi-induced transcriptional activation of lysosomal function can cause metastatic dissemination and chemo-resistance, associated with an enhanced TGF-β signaling pathway. Therefore, modifying the lysosomal metabolism-mediated TGF-β pathway presents a significant challenge and a potential breakthrough in the development of chemotherapeutic agents for anti-TNBC therapies and Treg biology.
In our recently published article, "Suppression of lysosome metabolism-mediated GARP/TGF-β1 complexes specifically depletes regulatory T cells to inhibit breast cancer metastasis" (https://www.nature.com/articles/s41388-024-03043-y), we have presented a novel synergistic effect for breast cancer immunotherapy and drug discovery. Our findings indicate that suppressing TRAF3IP3 in lysosome-mediated GARP/TGF-β1 complex activation can specifically deplete Tregs and increase the sensitivity of TNBCs to chemotherapy.
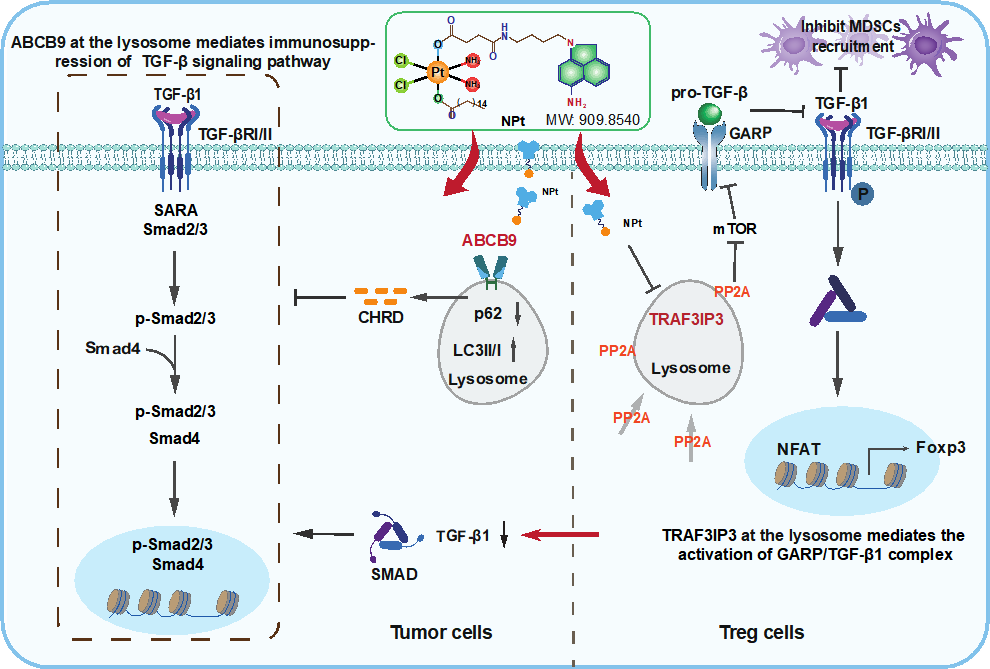
Through the exploration of a novel naphthalamide-platinum drug (NPt), which acts as an immune modulator, we discovered that NPt can specifically reduce the number of CD4+CD25+Foxp3+ Tregs in the tumor microenvironment by inhibiting the production of active GARP/TGF-β1. This effect is distinct from that of cisplatin and currently available immune-modulatory antibodies in clinical use. Mechanistically, TRAF3IP3 within lysosomes promotes the production of active TGF-β1 in Tregs, but not in other cell types, thereby facilitating the recruitment of myeloid-derived suppressor cells (MDSCs) and the progression of breast cancer. The upregulation of ABCB9 in lysosomes also contributes to the autophagy-lysosome-mediated TGF-β pathway in TNBCs. Interestingly, we synthesized nine naphthalenelactam-modified platinum complexes and identified one that, when localized in lysosomes, specifically activates ABCB9 in TNBC and TRAF3IP3 in Tregs. This complex engages with GARP through the PP2A/mTOR pathway, subsequently blocking the activation of human Tregs by TGF-β1.
.png)
Scheme 1. The schematic illustration of antitumor mechanisms of NPt. The scheme highlights TRAF3IP3 at the lysosome inhibits the production of active GARP/TGF-β1 in Tregs but not in other cell types to deter MDSCs (myeloid-derived suppressor cells) recruitment and breast cancer progression. NPt suppresses ABCB9 and TRAF3IP3-mediated divergent lysosomal metabolic programs in TNBC and Tregs for remodeling the tumor microenvironment.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in