The microbial trafficking in genetic parts – within us
Published in Microbiology
There are more things in heaven and earth, Horatio,
Than are dreamt of in your philosophy.
- Hamlet (Act 1, Scene 5, 167-8)
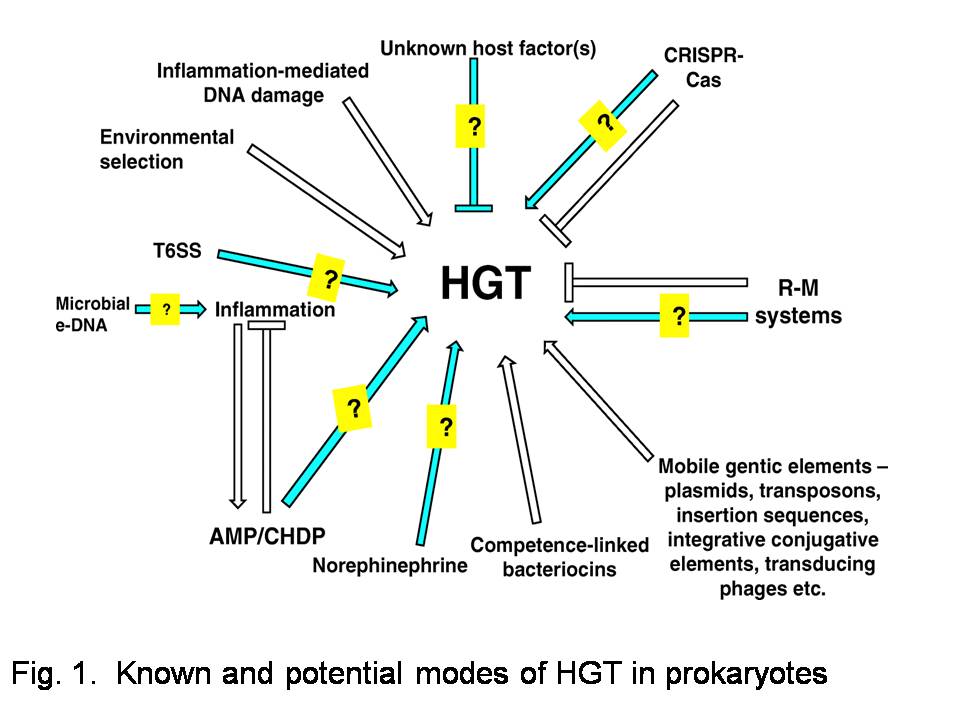
Humans harbor microbes in numbers comparable to that of their own cells [1]. This assemblage, specifically when considered as a unit of natural selection, has been termed as the (human) ‘holobiont’ [2]. Diverse microbial communities in various contexts exhibit horizontal genetic transfer (HGT) via a variety of mechanisms. Horizontal, because this transfer of genetic material occurs among unrelated individuals, as opposed to vertical transfer through lines of descent. Within the prokaryotic component of the microbiota, HGT can occur not only through the ‘classical’ modes – transformation, conjugation or transduction – but also via more recently discovered ones such as virus-like gene transfer agents, nanotubes, membrane vesicles and so on. An incidental outcome of microbiome studies fueled by advances in high-throughput sequencing technologies is the realization that HGT can occur and be stabilized across phylogenetically distant groups of prokaryotes, especially under appropriate selection pressures. Several of these findings and possibilities for HGT are summarized in Fig. 1 (Fig. 2 in the paper).
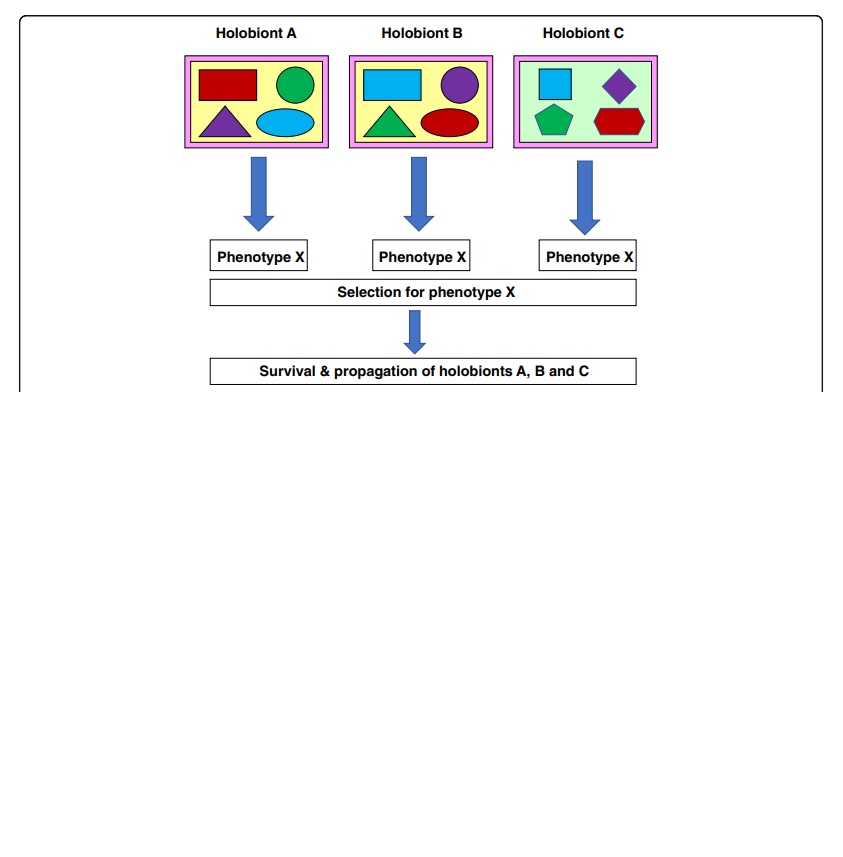
Now, as the microbiome is part of the human holobiont, do these instances of HGT have consequences for the holobiont? In my article, I have attempted to synthesize information from diverse sources that could have a bearing on this question. Ever since Robert Koch conducted his seminal studies and formulated the germ theory of disease, we have studied microbes as pests rather than partners for a longer period of time. Nevertheless, lurking in the data derived from the pests (decidedly a minuscule minority relative to their benign or indifferent kinsfolk) are tantalizing indications that aspects of human physiology can influence HGT among the resident microbiota that can, in turn, influence host physiology, again impacting HGT. It is conceivable that such reciprocal influences result in the ‘tightening’ of the association of men and microbes. By way of a case study, I have dwelt at some length on the chronic association between the stomach and duodenal ulcer-causing bacterium Helicobacter pylori and humans, its only known natural host. Given the potential importance of such associations between humans (or other macroscopic organisms) and their resident microbiota, we suggest that the holobiontic perspective may be important in specific contexts. However, other contexts may not require it, and may be effectively examined in the ‘traditional,’ reductionist standpoint of a microbial community in a given habitat, or even that of individual members of the community.
The above writing admittedly seems more ‘about the paper’ rather than something ‘behind’ it as this section would have you believe. Not to disappoint, we make amends for our readers who take the time to access the article at Microbiome: Mark Twain’s acerbic view of the microbial world and the mysterious ways of Providence at the beginning. It also happens to be the only section that remained untouched by any criticism during the review process, and was not censured by the handling editor either.
Note: This article is part of the holobiont-themed collection titled "Host-microbiota interactions: from holobiont theory to analysis." Poster image from Fig. 1 of my paper, and the image within the text is from Fig. 2 of my paper used in accordance with the Creative Commons Attribution License 4.0.
References:
1. Sender R, Fuchs S, Milo R (2016) Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol 14(8): e1002533. https://doi.org/10.1371/journal.pbio.1002533
2. Margulis L. Symbiogenesis and symbionticism. In: Margulis L, Fester R, editors. Symbiosis as a source of evolutionary innovation: speciation and morphogenesis. Cambridge: MIT; 1991. p. 1-14.
Disclaimer: The opinions expressed herein do not represent the views of the TERI School of Advanced Studies or TERI.
Follow the Topic
-
Microbiome
This journal hopes to integrate researchers with common scientific objectives across a broad cross-section of sub-disciplines within microbial ecology. It covers studies of microbiomes colonizing humans, animals, plants or the environment, both built and natural or manipulated, as in agriculture.
Related Collections
With Collections, you can get published faster and increase your visibility.
Animal Gut Nutrition and Greenhouse Gas Mitigation
Animal Microbiome, Journal of Animal Science and Biotechnology and Microbiome call for submissions to the collection on Animal Gut Nutrition and Greenhouse Gas Mitigation.
Efforts to reduce greenhouse gas emissions from livestock systems increasingly hinge on innovations in animal gut nutrition. The dynamic relationship between the gut microbiome and nutrient utilization plays a pivotal role in shaping methane output, feed efficiency, and overall sustainability. Advances in microbial ecology—particularly in understanding the role of gut microbiome in nutrient metabolism—are opening new pathways for mitigating emissions while enhancing productivity. These developments support the implementation of climate-smart agricultural strategies to address climate change and its impacts.
Looking ahead, continued research in this field has the potential to yield innovative solutions such as targeted probiotic supplementation, which could further optimize gut function and enhance nutrient absorption. These advancements may lead to reduced greenhouse gas emissions while improving animal health and productivity. By deepening our understanding of the animal gut microbiome, we can contribute significantly to sustainable agricultural practices that benefit both the environment and food security.
We invite researchers to contribute to this special Collection on Animal Gut Nutrition and Greenhouse Gas Mitigation. Topics of interest include but are not limited to:
- Animal Gut Microbiome and Feed Efficiency
- Greenhouse Gas Mitigation Strategies
- Rumen Fermentation Dynamics
- Nutrient Utilization in Livestock
- Probiotic Supplementation Effects
- Sustainable Livestock Production Practices
- Climate-Smart Agriculture Innovations
This Collection supports and amplifies research related to SDG 13, Climate action.
All submissions in this collection undergo the relevant journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief of the relevant journal. As an open access publication, participating journals levy an article processing fee (Animal Microbiome fees, Journal of Animal Science and Biotechnology fees, Microbiome fees). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief of the journal where the article is being submitted.
Publishing Model: Open Access
Deadline: Sep 04, 2026
The Apple Microbiome
Microbiome and Environmental Microbiome are calling for submissions to our Collection on the Apple Microbiome.
With world apple production estimated at 84 million tons, the microbiome of the apple has significant implications for agriculture, food security, and human health. Understanding the complex interactions between apple plants and their associated microbial communities can lead to improved crop management strategies, enhanced fruit quality and longevity, and sustainable agricultural practices. Recent advances have highlighted the role of specific bacteria and fungi in promoting plant health and resilience against specific pathogens. Moreover, detailed profiling of these microbial communities, revealing their diversity and functional potential facilitate exciting future developments, such as the identification of beneficial microbial consortia for biocontrol and the formulation of tailored probiotic treatments for both plants and humans. By advancing our collective understanding in this area, we can work towards a more sustainable and resilient agricultural system.
Topics of interest include but are not limited to:
-Microbial diversity and function associated with apples
-Effects of soil health and rhizosphere interactions on apple production
-Impact of climate change on the apple microbiome
-Role of the apple microbiome in fruit quality
-Microbiome-driven strategies for disease resistance
This collection is open for submissions from all authors on the condition that the manuscript falls within both the scope of the collection and the journal it is submitted to.
All submissions in this collection undergo the relevant journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief of the relevant journal. As an open access publication, participating journals levy an article processing fee (Microbiome, Environmental Microbiome). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief of the journal where the article is being submitted.
Publishing Model: Open Access
Deadline: Aug 05, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in