Assembly drives regioselective azide-alkyne cycloaddition reaction
Published in Chemistry
The research group of Prof. Gaolin Liang at Southeast University focuses on the nanochemistry based on click reaction and supramolecular self-assembly (Angew. Chem., Int. Ed. 2023, e202306427; J. Am. Chem. Soc. 2023, 145, 7918; Sci. Adv. 2020, 6, eaba3190; Nat. Commun. 2018, 9, 2550; J. Am. Chem. Soc. 2017, 139, 1041; J. Am. Chem. Soc. 2016, 138, 11128; Angew. Chem., Int. Ed. 2015, 54, 3639-3642), as well as biomedical analysis (Angew. Chem., Int. Ed. 2022, 61, e202114766; J. Am. Chem. Soc. 2022, 144, 11897). We have developed a novel CBT-Cys click reaction and successfully combined with supramolecular self-assembly strategy to develop molecular probes for diseases diagnosis (Nat. Chem. 2010, 2, 54; Nat. Commun. 2019, 10, 1192; Angew. Chem., Int. Ed. 2015, 54, 9700). Based on our combined background, we developed an assembly-driven azide-alkyne cycloaddition reaction for metal-free biomedical implication. This work has been published on Nature Communications very recently.
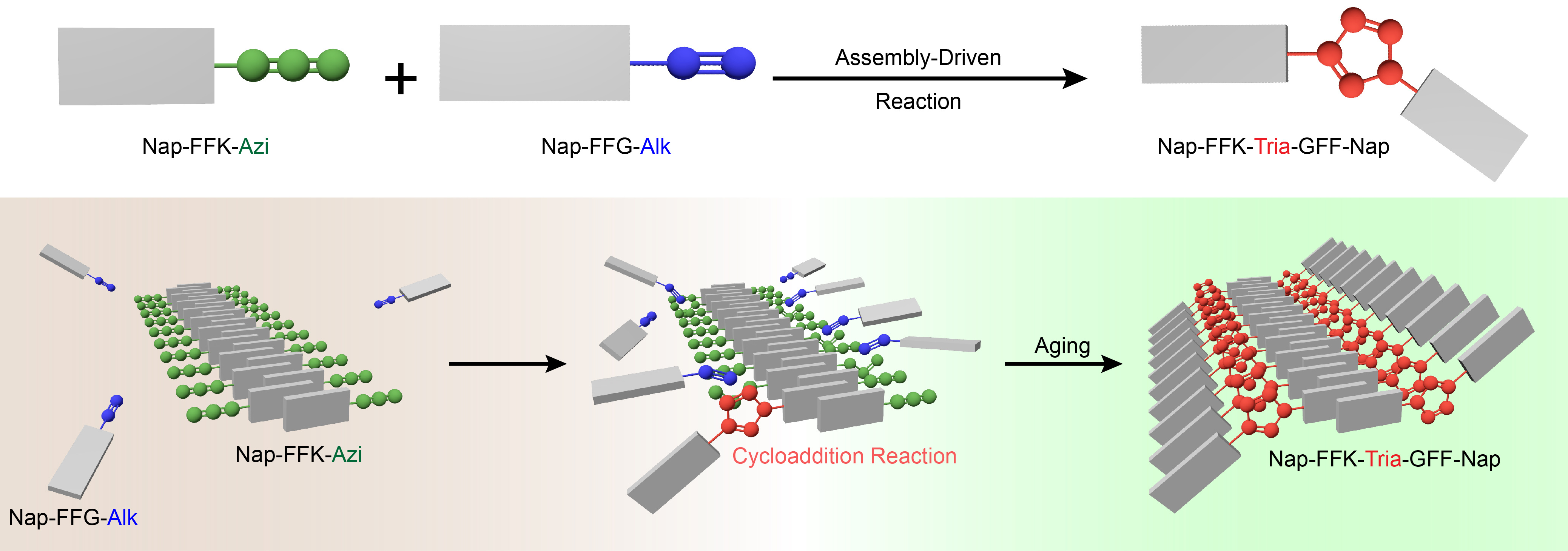
Azide-alkyne cycloaddition reaction is a very common organic reaction to synthesize nitrogen-containing heterocycles. Once catalyzed by Cu(I) or Ru(II), it turns out to be a click reaction and thus is widely applied in chemical biology for labeling. However, besides their poor regioselectivity towards this reaction, these metal ions are not biologically friendly. Hence, it is an urgent need to develop a metal-free azide-alkyne cycloaddition reaction for biomedical applications. In this work, we found that, in the absence of metal ions, supramolecular self-assembly in an aqueous solution could realize this reaction with excellent regioselectivity (Fig. 1). Nap-Phe-Phe-Lys(azido)-OH firstly self-assembled into nanofibers. Then, Nap-Phe-Phe-Gly(alkynyl)-OH at equivalent concentration approached to react with the assembly to yield the cycloaddition product Nap-Phe-Phe-Lys(triazole)-Gly-Phe-Phe-Nap to form nanoribbons. Due to space confinement effect, the product was obtained with excellent regioselectivity.
-01.jpg)
Traditional copper-catalyzed azide-alkyne cycloaddition reaction (CuAAC)
We first synthesized the three compounds Nap-FFK-Azi, Nap-FFG-Alk, and Nap-FFK-Tria-GFF-Nap. Besides from the desired product of Nap-FFK-Tria-GFF-Nap (retention time 20.3 min), the by-product 1,5-isomer of Nap-FFK-Tria-GFF-Nap (retention time 16.1 min) also appeared on the HPLC trace (Fig. 2), suggesting poor regioselectivity of CuAAC.
-01.jpg)
Monitoring of assembly-driven azide-alkyne cycloaddition reaction
As shown in Fig. 3a, after 7 d aging, desired 1,4-disubstituted cycloaddition product Nap-FFK-Tria-GFF-Nap (HPLC retention time 20.3 min) was yielded in the hydrogel. Interestingly, the 1,5-isomer of Nap-FFK-Tria-GFF-Nap was not found in the hydrogel (Fig. 3a), suggesting excellent regioselectivity of this assembly-driven cycloaddition reaction. The reaction yield increased gradually and reached its plateau of 19.1% at 14 d.
Necessary conditions for supramolecular self-assembly in aqueous solution (e.g., hydrophobic interaction, π-π stacking, hydrogen bonding, etc) do not exist in organic solvent such as N,N-dimethylformamide (DMF). When Nap-FFK-Azi and Nap-FFG-Alk remained monomeric state in DMF, no cycloaddition product of Nap-FFK-Tria-GFF-Nap was found in the incubation (Fig. 3a), indicating that the product in above hydrogel was indeed yielded under assembly effect.
In TEM images, dense and slim nanofibers were observed in Nap-FFK-Azi hydrogel, helical ribbons were found in Nap-FFG-Alk hydrogel, and broader helical ribbons presented in Nap-FFK-Tria-GFF-Nap solution. After 7 d aging, besides slim nanofibers, helical ribbons in Nap-FFK-Azi + Nap-FFG-Alk hydrogel was clearly observed (Fig. 3b). Above observations indicated that cycloaddition reaction between Nap-FFK-Azi and Nap-FFG-Alk indeed occurred in the hydrogel after excluding the interference of co-assembly.
CD results confirmed the inference further (Fig. 3c). CD spectrum indicated the β-turn-like structures in Nap-FFK-Azi nanofibers, β-sheet-like structures in helical nanoribbons of Nap-FFG-Alk, and β-sheet-like structures in Nap-FFK-Tria-GFF-Nap nanoribbons. Interestingly, peaks close to those in Nap-FFK-Tria-GFF-Nap solution appeared in the CD spectrum of Nap-FFK-Azi + Nap-FFG-Alk hydrogel, suggesting the nanoribbons in the hydrogel are made of Nap-FFK-Tria-GFF-Nap.
.jpg)
Mechanism of assembly-driven azide-alkyne cycloaddition reaction
To validate the mechanism of assembly-driven regionally selective azide-alkyne cycloaddition reaction, we used TEM observations to real time monitor the formation of above nanostructures. TEM images in Fig. 4a showed that, at 0.5 h, those raw materials at nanoscale for self-assembling into bigger nanostructures (e.g., nanoparticles for nanofiber formation, nanofibers for nanoribbon formation) started to appear in hydrogels of Nap-FFK-Azi and Nap-FFG-Alk, or solution of Nap-FFK-Tria-GFF-Nap. The final nanostructures (i.e., nanofibers in hydrogel Nap-FFK-Azi, nanoribbons in hydrogel Nap-FFG-Alk and solution Nap-FFK-Tria-GFF-Nap) started to form at 3 h and stabilize at 6 h. In contrast, TEM images of Nap-FFK-Azi + Nap-FFG-Alk hydrogel showed that dense and slim nanofibers (i.e., Nap-FFK-Azi nanofibers) formed at 3 h (Fig. 4b). After that, from 24 h to 4 d, we found that nanoparticles (i.e., generation of the cycloaddition product Nap-FFK-Tria-GFF-Nap) were continuously generated along the nanofibers. Finally, the nanoparticles connected with each other to form helical nanoribbons at day 7. This result also indicates that occurrence of the cycloaddition reaction in Nap-FFK-Azi + Nap-FFG-Alk group improve the stability of the intermediate nanoribbon structure of the two precursor peptides to the final nanoribbons. Above TEM observations directly validate our hypothesis that, along with the self-assembly of nanofiber, Nap-FFK-Azi or Nap-FFG-Alk is drawn near to the nanofiber with space-confined effect, undergoing cycloaddition reaction to yield regionally selective product Nap-FFK-Tria-GFF-Nap.
.jpg)
In summary, we rationally designed three compound and validated that assembly could drive metal-free azide-alkyne cycloaddition reaction with excellent regioselectivity. Previous studies have revealed that hydrophobic interactions and hydrogen-bonding could accelerate the Diers-Alder reaction. This work demonstrated another example of using assembly to realize metal-free azide-alkyne cycloaddition with excellent regioselectivity. Currently, metal-free bioorthogonal click reactions show promising potential in a variety of biological applications including biomolecule labelling, prodrug activation, and cancer theranostics. Encouraged by this work, we are applying supramolecular self-assembly to realize more reactions without metal ion catalysis, and those works are on the way.
Reference:
- Wenjun Zhan,# Lingling Xu,# Zhiyu Liu,# Xiaoyang Liu, Ge Gao, Tiantian Xia, Xiaotong Cheng, Xianbao Sun, Fu-Gen Wu, Qian Yu, and Gaolin Liang*. Tandem Guest-Host-Receptor Recognitions Precisely Guide Ciprofloxacin to Eliminate Intracellular Staphylococcus aureus. Angew. Chem., Int. Ed. 2023, e202306427 (DOI: 10.1002/anie.202306427).
- Xiaoyang Liu, Wenjun Zhan, Ge Gao, Qiaochu Jiang, Xinping Zhang, Hongbo Zhang, Xianbao Sun, Wei Han, Fu-Gen Wu, and Gaolin Liang*. Apoptosis-Amplified Assembly of Porphyrin Nanofiber Enhances Photodynamic Therapy of Oral Tumor. J. Am. Chem. Soc. 2023, 145(14), 7918–7930 (DOI: 10.1021/jacs.2c13189).
- Miaomiao Zhang, Yong Guan, Zheng Dang, Pinggen Zhang, Zhen Zheng, Liang Chen, Wen Kuang, Chenchen Wang, and Gaolin Liang*. Directly observing intracellular nanoparticle formation with nanocomputed tomography. Sci. Adv. 2020, 6(43), eaba3190 (DOI: 10.1126/sciadv.aba3190).
- Hongbin He, Hua Jiang, Yun Chen, Jin Ye, Aoli Wang, Chao Wang, Qingsong Liu, Gaolin Liang, Xianming Deng,* Wei Jiang,* and Rongbin Zhou*. Oridonin is a covalent NLRP3 inhibitor with strong anti-inflammasome activity. Nat. Commun. 2018, 9, 2550 (DOI: 10.1038/s41467-018-04947-6).
- Zijuan Hai, Jindan Li, Jingjing Wu, Jiacheng Xu, and Gaolin Liang*. Alkaline Phosphatase-Triggered Simultaneous Hydrogelation and Chemiluminescence. J. Am. Chem. Soc. 2017, 139(3), 1041–1044 (DOI: 10.1021/jacs.6b11041).
- Zhen Zheng, Peiyao Chen, Maolin Xie, Chengfan Wu, Yufeng Luo, Wentao Wang, Jun Jiang, and Gaolin Liang*. Cell Environment-Differentiated Self-Assembly of Nanofibers. J. Am. Chem. Soc. 2016, 138(35), 11128–11131 (DOI: 10.1021/jacs.6b06903).
- Chenchen Wang, Wei Du, Chengfan Wu, Shan Dan, Miao Sun, Tong Zhang, Bin Wang, Yue Yuan,* and Gaolin Liang*. Cathespin B-Initiated Cypate Nanoparticle Formation for Tumor Photoacoustic Imaging. Angew. Chem., Int. Ed. 2022, 61, e202114766 (DOI: 10.1002/anie.202114766).
- Ge Gao,# Yao-Wen Jiang,# Wenjun Zhan, Xiaoyang Liu, Runqun Tang, Xianbao Sun, Yu Deng, Lingling Xu, and Gaolin Liang*. Trident Molecule with Nanobrush-Nanoparticle-Nanofiber Transition Property Spatially Suppresses Tumor Metastasis. J. Am. Chem. Soc. 2022, 144(26), 11897–11910 (DOI: 10.1021/jacs.2c05743).
- Yue Yuan, Lin Wang, Wei Du, Zhanling Ding, Jia Zhang, Tao Han, Linna An, Huafeng Zhang*, and Gaolin Liang*. Intracellular Self-Assembly of Taxol Nanoparticles for Overcoming Multi-Drug Resistance. Angew. Chem., Int. Ed. 2015, 54(33), 9700–9704 (DOI: 10.1002/anie.201504329).
- Gaolin Liang, Hongjun Ren, and Jianghong Rao*. A biocompatible condensation reaction for controlled assembly of nanostructures in living cells. Nat. Chem. 2010, 2, 54–60 (DOI: 10.1038/nchem.480).
- Peiyao Chen,# Yinchu Ma,# Zhen Zheng,# Chengfan Wu, Yucai Wang,* and Gaolin Liang*. Facile syntheses of conjugated polymers for photothermal tumour therapy. Nat. Commun. 2019, 10, 1192 (DOI: 10.1038/s41467-019-09226-6).
- Shuang Liu, Anming Tang, Maolin Xie, Yundan Zhao, Jun Jiang, and Gaolin Liang*. Oligomeric Hydrogels Self-Assembled from Reduction-Controlled Condensation. Angew. Chem., Int. Ed. 2015, 54 (12), 3639–3642 (DOI: 10.1002/anie.201409952).
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in