Brainstem neuromelanin and iron MRI are novel imaging diagnostic markers for idiopathic and LRRK2 Parkinson’s disease.
Published in Neuroscience
Dopaminergic neuronal death in Parkinson’s disease (PD) is reflected by the loss of neuromelanin (NM) in the substantia nigra pars compacta and locus coeruleus, as well as iron accumulation in those brainstem structures. In our recent article published in npj Parkinson’s Disease, we accurately quantified and anatomically mapped brainstem NM and iron in genetically-determined PD patients carrying a mutation (G2019S) at the Leucine-rich repeat kinase 2 gene (LRRK2-PD), as well as in idiopathic Parkinson’s disease (iPD) and healthy controls (HC) to find accurate imaging biomarkers for the diagnosis of the disease.
First, we implemented a novel automated NM and iron MRI brainstem atlas to segment and quantify the substantia nigra pars compacta (SNc), locus coeruleus (LC), and red nucleus (RN), measuring their volume and signal contrast ratio (Figure 1). Then, the atlas was used to compare the NM and iron measurements in MRI scans from thirty-two HC, thirty-nine iPD, and twenty-four LRRK2-PD to assess its diagnostic potential.
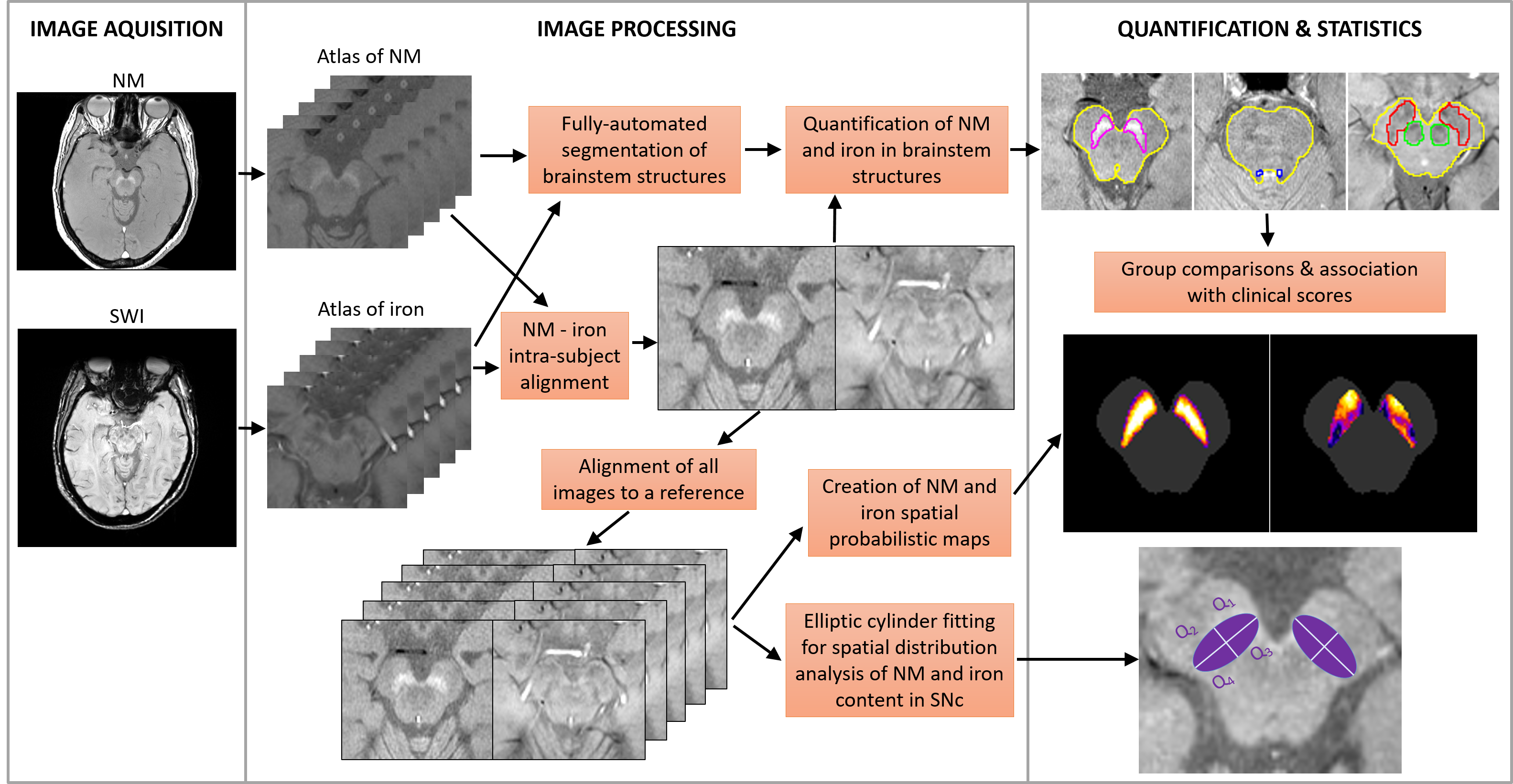
Figure 1 Automated atlas-based segmentation pipeline for NM and iron quantification in the brainstem.
Neuromelanin measurements
Both iPD and LRRK2-PD groups showed decreased NM volume and contrast ratio at SNc compared to HC (Figure 2a and 2b).These results reflect a spatial neurodegeneration with an impairment pattern involving areas responsible for the putaminal outputs (i.e., substantia nigra pars compacta lateral ventral to medial ventral and dorsal tiers), which are involved in the impaired motor function in PD.
Interestingly, the LC was preserved in LRRK2-PD compared with iPD (Figure 2c and 2d). This was strikingly opposed to the LC NM loss found in iPD compared with HCs. Very importantly, disease duration did not correlate either with the LC NM contrast ratio or volume in LRRK2-PD, thus reinforcing that NM preservation in LC preserved along the disease progression among LRRK2-PD. Thus, LC preservation is a main imaging signature for LRRK2-PD.
Iron measurements
SNc iron volume was larger in iPD and LRRK2-PD than in controls. A similar trend was found for the contrast ratio (Figure 2e and 2f). In contrast to controls, iron deposition in the SNc was similar to that of the substantia nigra pars reticulata (SNr) in PD patients. In addition, LRRK2-PD presented a higher contrast ratio signal in the RN than iPD (Figure 2g).
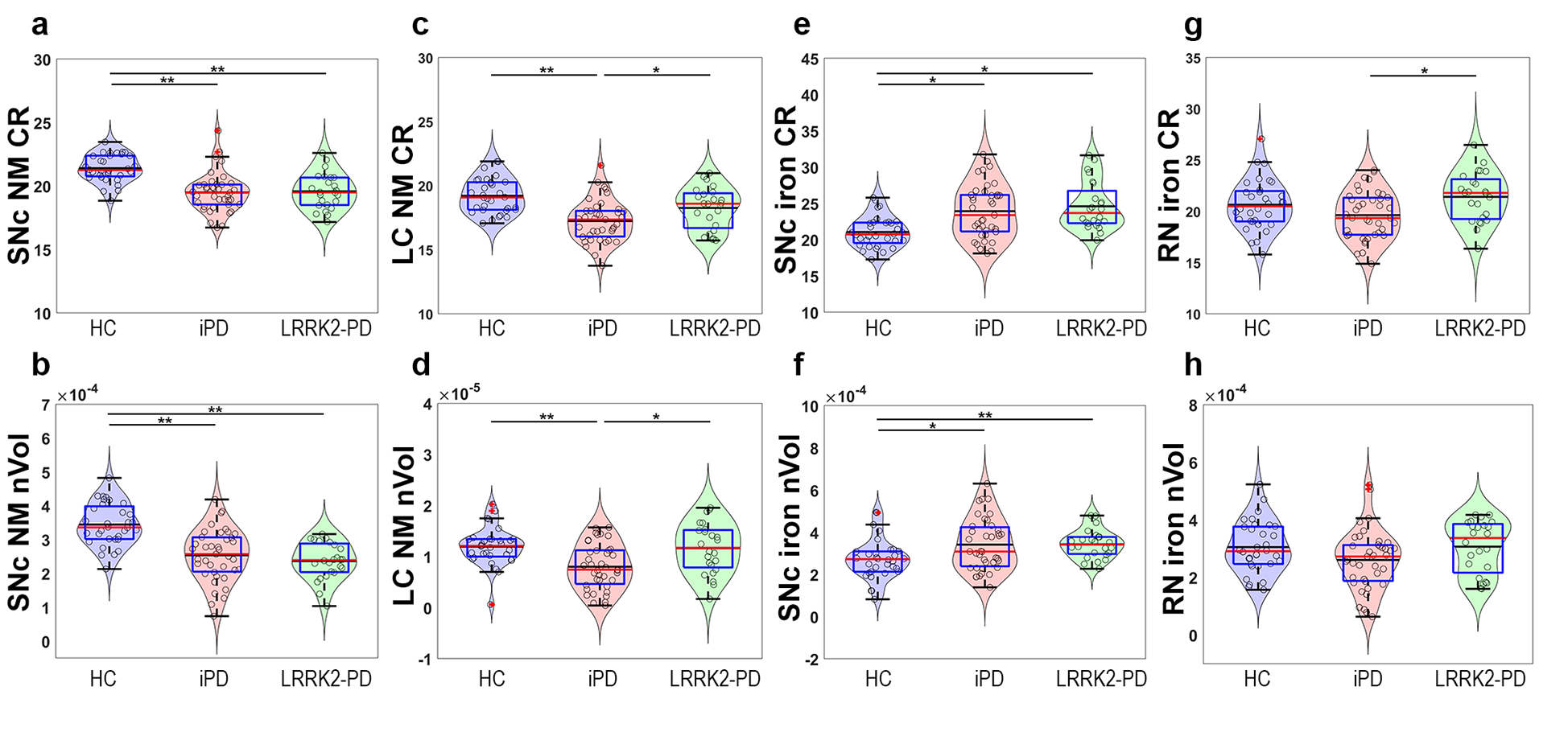
Figure 2 Box and Violin plots of the quantitative MRI measures. a SNc NM contrast ratio (CR), b SNc NM normalized volume (nVol), c LC NM CR, d LC NM nVol, e SNc iron CR, f SNc iron nVol, g RN iron CR, and h RN iron nVol. Red lines represent the median and black lines represent the mean, whereas grey horizontal lines indicate statistically significant differences between groups (* p < 0.05, ** p < 0.001, corrected for multiple testing).
Asymmetry in brainstem measurements
In all subjects, controls and patients, a volume asymmetry with left brain predominance was consistently found for both NM and iron in the SNc and also in NM content in the LC, which could be related to handedness.
Diagnostic potential
To assess the diagnostic potential of our atlas-based quantification, we analyzed the performance of the MRI measurements of NM and iron in the SNc, NM of the LC, and iron of the RN individually and combined using binary logistic regression models (see Figure 3). SNc NM was the best individual discriminator between HC and PD (AUC=0.89), followed by LC NM (AUC=0.77) and SNc iron (AUC=0.77). When NM and iron in the SNc were combined, the AUC raised to 0.93, and when the LC NM and RN iron parameters were added to the model, the highest performance (AUC=0.94) was achieved for the HC vs. PD comparison.
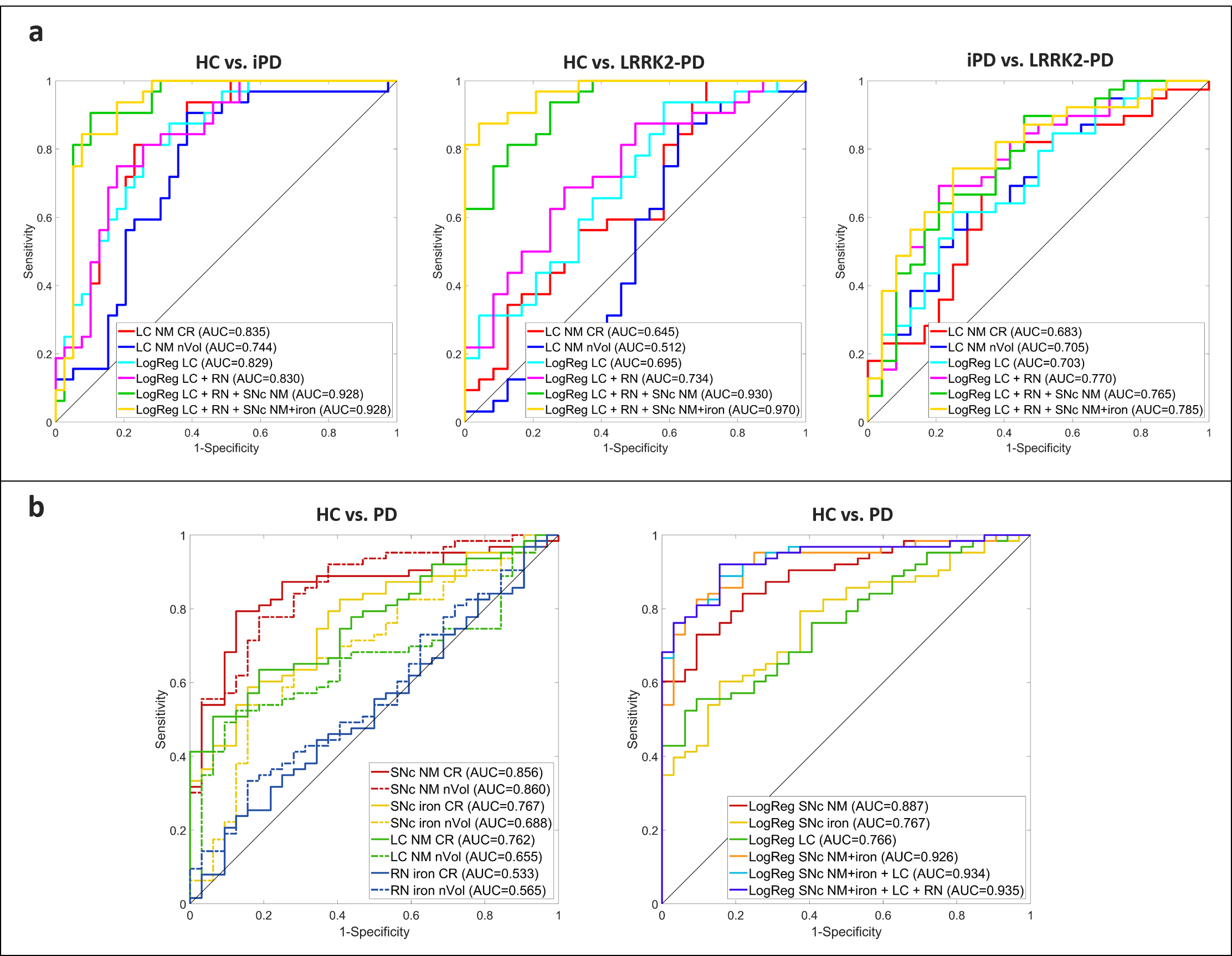
Figure 3 Diagnostic performance of brainstem measurements. a Pairwise comparison of HC, iPD and LRRK2-PD groups. b HC vs. PD group using individual and combined parameters through logistic regression models.
Associations between brainstem measurements and clinical outcomes in PD
We employed robust regression analyses to test for potential associations between MRI brainstem measurements and clinical and sociodemographic variables in the entire cohort of PD patients. Brainstem measurements of the SNc were predicted by disease duration, the presence of psychiatric symptoms, and age. Specifically, longer disease duration was related to a higher contrast ratio and greater volume of iron in the SNc and the presence of psychiatric symptoms was related to a decrease of NM contrast ratio in the SNc. Older age was related to a decrease of iron volume in the SNc. Finally, being male was related to a lower NM volume in the locus coeruleus.
Therefore, the use of an automated atlas for the analysis of neuromelanin and iron burden in the substantia nigra pars compacta and locus coeruleus in PD allowed us to find novel hallmarks for PD subtypes. The preservation of the NM content in the locus coeruleus is a valid imaging signature for G2019S LRRK2 PD. In terms of clinical applicability, the specific imaging patterns found could be used as diagnostic tools for PD and PD-LRRK2 and can help to quantify the level of brainstem neurodegeneration as disease progresses.
Follow the Topic
-
npj Parkinson's Disease
This journal publishes original basic science, translational and clinical research related to Parkinson's disease, including anatomy, etiology, genetics, cellular and molecular physiology, neurophysiology, epidemiology and therapeutic development and treatments.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
The neuroimmune-axis and ageing in Parkinson’s Disease
Publishing Model: Open Access
Deadline: Jul 15, 2026
Cognition - preclinical models, and preclinical unmet need
Publishing Model: Open Access
Deadline: Jul 27, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in