Evaluation of the efficacy of cystinosin supplementation through CTNS mRNA delivery in experimental models for cystinosis
Published in Protocols & Methods, Biomedical Research, and General & Internal Medicine
Lysosomal storage disorders (LSD) are a group of over 70 monogenic diseases, affecting different lysosomal proteins and leading to lysosomal substrate accumulation. Cystinosis is a prototype lysosomal storage disorder caused by mutations in the CTNS gene, encoding the lysosomal cystine-H+ symporter cystinosin, and leading to cystine accumulation in all cells of the body. The kidneys are the first and the most severely affected organs, presenting glomerular and proximal tubular dysfunction.
The current therapeutic standard cysteamine, reduces cystine levels, but has many side effects and does not restore kidney function. Therefore, the search for novel treatment strategies is ongoing. Messenger RNA (mRNA) therapies are emerging in diferent disease areas, but they have not yet reached the kidney field.
Our aim
We aimed to study the feasibility to treat the genetic defect in cystinosis using synthetic mRNA in cystinotic cell models and ctns-/- zebrafish embryos.
What we did
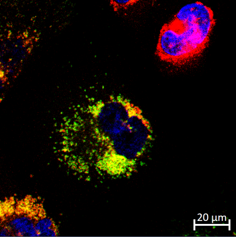
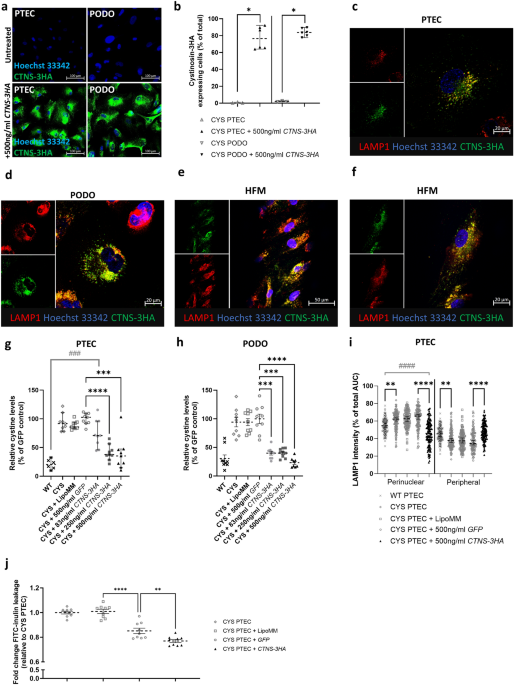
For this study, we used 2 in vitro models and 1 in vivo model to evaluate if mRNA-based protein replacement could result in the amelioration of the cystinosis phenotype. For the in vitro study, we used 2 kidney cell types that are affected by cystinosis: the proximal tubular epithelial cells (PTECs) and the podocytes (PODOs). In these cells, we evaluated the timeframe of cystinosin protein expression after transfection with a 3HA-tagged version of the CTNS mRNA, which was codon optimized and equiped with translation promoting UTRs. To assess efficiency, we measured intracellular cystine levels for up to 20 days. Next, we injected a mCherry-tagged version of the CTNS mRNA in a cystinotic zebrafish embryo at the one-cell stage. The protein expression was followed up during development and the cystine levels measured. Most importantly, we evaluated the effect of the mRNA-treament on the proximal tubular defect and proteinuria that has been described in these fish.
What we observed
In both the cell models and fish models, we were able to show the expression of the protein after the mRNA-treatment. This expresssion persisted for a few days, after which it became undetectable. However, the long term follow-up in the cells showed that the effect of a single dose remained for a minimum of 10 days, with a noticeable difference between the two cell types. In the zebrafish embryo, we could show that the kidney/pronephros function significantly improved, characterized by a reduced proteinuria, increased proximal tubular reabsorption rate, increased expression of multi-ligand receptors on the tubular cells and a reduced proximal tubular apoptosis.
What we concluded
This proof-of-principle study took the first steps in establishing an mRNA-based therapy to restore cystinosin expression and improve the cystinosis phenotype in vitro and in vivo. Thereby, we also paved the way for mRNA-based protein replacment to treat other genetic kidney diseases.
Follow the Topic
-
Scientific Reports
An open access journal publishing original research from across all areas of the natural sciences, psychology, medicine and engineering.
Related Collections
With Collections, you can get published faster and increase your visibility.
Phytochemicals and health
Publishing Model: Open Access
Deadline: Jul 28, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in