
Understanding the three-dimensional shape, or conformation, of a molecule goes a long way in chemistry. This knowledge is vital for the accurate prediction of many properties – from chemical reactivity to biological activity. Minute alterations can render molecules unreactive or unable to perform their intended function. Conversely, alterations can also “turn on” the desired mode of action. Thus, understanding the rules that govern conformational interconversion is at the heart of chemistry.
When it comes to conformational analysis, organic molecules have a storied history. Starting from Derek Barton and Odd Hassel’s foundational work, chemists have grown to appreciate molecular conformations, which are now a standard fixture in the undergraduate chemistry curriculum. It is understood that high-energy conformations are transient and rarely considered for applications other than chemical reactivity. In theory, these conformations may have interesting properties other than the structural changes during bond-breaking and bond-making events. Unfortunately, there are no simple ways to observe and study these molecular shapes for an appreciable time. Cyclohexane, with its boat, twist-boat, half-chair, and chair forms, is a good illustration of many theoretically possible conformations.
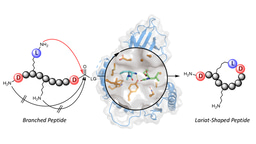
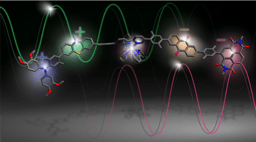
Apart from relatively small rings such as cyclohexane, there is currently no easy way to navigate the conformational space of more complex ring structures. In the area of polypeptides, it is known which amino acid sequences result in the formation of an ⍺- helix, a β-turn, and β-sheet structures. Other than the formation of canonical motifs, there is little understanding of how to induce unusual structural motifs in macrocycles. In this paper, we consider macrocycles composed mainly of amino acid residues and use the term ‘dark conformational space’ to describe metastable states in peptide structures. We show that high-energy conformations can be enforced using the constructs we term ‘dominant rotors’. These units remodel the conformational landscape of 16- to 22-membered rings and enable observation of peptide conformations that are uncommon because of their particular geometrical features. All the macrocycles in this study exhibit two-well behaviour: each atropdiastereomer adopts one preferred conformation in solution. However, macrocycles that harbour an N-methylated amino acid were found to have one of the two energy wells in a conformationally heterogeneous state.
This tool should now allow exploration of a wide range of structures, opening the tantalizing possibility that some molecular properties might have remained veiled because of the difficulties to generate and stabilize conformational ensembles that are energetically unfavourable. We also recognize that a drastic increase in the rate of synthesis is needed to facilitate exploration of the dark conformational space. Current efforts are directed toward addressing this challenge.
Written by Prof. Andrei K. Yudin and Dr. Diego B. Diaz
Link to Article: https://rdcu.be/cfeHT
Follow the Topic
-
Nature Chemistry

A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in