Is Aluminum going to revolutionize energy storage? Or are its properties just seducing us?
Published in Sustainability

Last October, a large battery company approached me about doing a project related to Al-based batteries. They were familiar with our work on alkaline Zn-MnO2 and Li-ion chemistries and thought that some of the approaches we were taking to improve energy density in those systems might be applied to Al, either primary (non-rechargeable) or secondary (rechargeable). Being good scientists and engineers my students (Ehsan Faegh, Benjamin Ng and Dillon Hayman) and I started working through the relevant literature with the thoughts of finding a right niche for us to start developing some new Al-related concepts. Although new ideas came to us, we never tried to get the company to fund them. In fact, when I visited with this company a couple of months later, I delivered a 2-hour seminar (plus Q&A) that argued for why they should not invest in Al at all. With additional work – constantly collaborating with Ehsan, Ben and Dillon over the next several months on the analysis and writing – that presentation became our paper in Nature Energy [1]. I felt that we had an obligation to reach out to the community and tell everyone what we had found.
I have always prided myself on the Engineer part of my title. In some circles, engineering has become ‘four letter word’, if you know what I mean. I chose engineering because I like to make real things that work. It’s why many of our best papers are so application oriented. It is why I love to work with industry – both on projects and consulting. It is also why I am always very curious about the end-game with any of the technologies that we are working on. What are we trying to do? What is even possible? Not just theoretically, but practically.
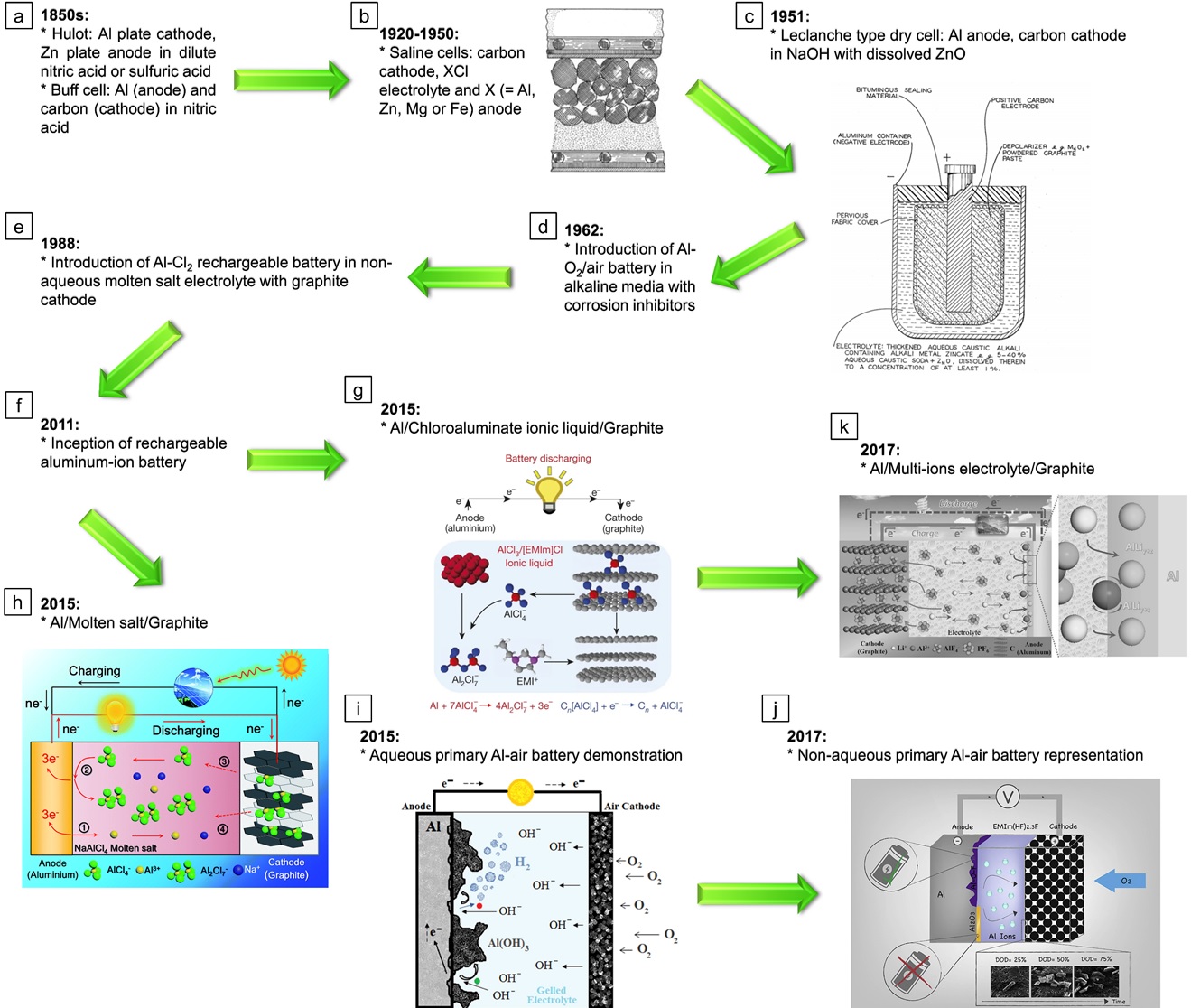
One of the things that was clear to me as we dug into the Al literature and recently funded projects is that nearly everyone (researchers, funding agencies, etc.) has been intoxicated by the promises of ultra-high capacity (2981 mAh/g) and energy density (4140 Wh/kg) for metallic Al. This is not new. Al has been explored as a battery electrode material for literally 170 years. A brief summary of that development is shown in Figure 1. However, no battery has come even close to meeting the capacity and energy density numbers above in a lab cell, let alone a practical one. And the pathway forward is quite unclear.
In aqueous batteries, Al is either rapidly corroding or catastrophically passivated. In non-aqueous cells, such as those with ionic liquid electrolytes used in aluminum ion batteries (AIBs), the Al complexes in the electrolyte and the overall reaction does not give 3 electrons per Al atom that is assumed in the high numbers above, but actually gets only 3 electrons for every 8 aluminum atoms. Most of the extra Al is not solid, but dissolved in the electrolyte – meaning a lot more weight and volume than “theoretical”. In addition, these AIBs typically use graphite cathodes that need many C atoms per Al, adding mass. The combination of electrolyte mass and cathode mass (in addition to other practical things like packaging, etc.) significantly drives down the achievable energy density to values closer to 50 Wh/kg. And the true limit when all practical components are taken into account is only around 80 Wh/kg. Though these values may be competitive with Pb-acid batteries, they are not competitive with Li-ion batteries at all. And primary chemistries that exist based on aluminum air batteries (AABs) have also only been able to achieve practical energy densities well below 100 Wh/kg, far below the alkaline and LiFeS2 primaries that we can already buy at the drug store. Another aspect where Al-based batteries have failed so far is in their lifetime. State-of-the-art chemistries have very poor long-term operational and shelf stability.
I hope that one day someone makes me eat these words, but from where I am standing Al-based batteries are just a dream. Their properties are quite tempting, but that only has made us try harder and harder not to let them go. That is why you will not see my group working on them anytime soon. It is hard for me to see a way forward for any of the existing approaches to get from our lab into your home. For me to be wrong, and it has happened before, it appears necessary for those who will press on in this area to completely rethink the chemistry and, hopefully, our paper also encourages them to critically think about the whole cell, not just the allure of one of the reactions.
References
1. Faegh, E; Ng, B.; Hayman D, Mustain WE. Practical Assessment of the Performance of Aluminum Battery Technologies. Nature Energy, DOI: 10.1038/s41560-020-00728-y.
2. Richards, J. W., Aluminium: its history, occurrence, properties, metallurgy and applications, including its alloys. HC Baird & Company: 1890.
3. Benjamin, P., The Voltaic Cell: Its Construction and Its Capacity. Wiley: 1893.
4. Heise, G. W.; Schumacher, E. A.; Cahoon, N., A Heavy Duty Chlorine‐Depolarized Cell. Journal of The Electrochemical Society 1948, 94 (3), 99-105
5. Sargent, D. E., Voltaic cell. Google Patents: 1951
6. Zaromb, S., The use and behavior of aluminum anodes in alkaline primary batteries. Journal of The Electrochemical Society 1962, 109 (12), 1125-1130.
7. Gifford, P.; Palmisano, J., An aluminum/chlorine rechargeable cell employing a room temperature molten salt electrolyte. Journal of The Electrochemical Society 1988, 135 (3), 650-654
8. Jayaprakash, N.; Das, S.; Archer, L., The rechargeable aluminum-ion battery. Chemical Communications 2011, 47 (47), 12610-12612
9. Lin, M.-C.; Gong, M.; Lu, B.; Wu, Y.; Wang, D.-Y.; Guan, M.; Angell, M.; Chen, C.; Yang, J.; Hwang, B.-J., An ultrafast rechargeable aluminium-ion battery. Nature 2015, 520 (7547), 324
10. Song, Y.; Jiao, S.; Tu, J.; Wang, J.; Liu, Y.; Jiao, H.; Mao, X.; Guo, Z.; Fray, D. J., A long-life rechargeable Al ion battery based on molten salts. Journal of Materials Chemistry A 2017, 5 (3), 1282-1291
11. Pino, M.; Chacón, J.; Fatás, E.; Ocón, P., Performance of commercial aluminium alloys as anodes in gelled electrolyte aluminium-air batteries. Journal of Power Sources 2015, 299, 195-201
12. Gelman, D.; Shvartsev, B.; Wallwater, I.; Kozokaro, S.; Fidelsky, V.; Sagy, A.; Oz, A.; Baltianski, S.; Tsur, Y.; Ein-Eli, Y., An aluminum–ionic liquid interface sustaining a durable Al-air battery. Journal of Power Sources 2017, 364, 110-120
13. Wang, S.; Jiao, S.; Tian, D.; Chen, H. S.; Jiao, H.; Tu, J.; Liu, Y.; Fang, D. N., A Novel Ultrafast Rechargeable Multi‐Ions Battery. Advanced Materials 2017, 29 (16), 1606349
Follow the Topic
-
Nature Energy
Publishing monthly, this journal is dedicated to exploring all aspects of this on-going discussion, from the generation and storage of energy, to its distribution and management, the needs and demands of the different actors, and the impacts that energy technologies and policies have on societies.
What are SDG Topics?
An introduction to Sustainable Development Goals (SDGs) Topics and their role in highlighting sustainable development research.
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Microgrids and Distributed Energy Systems
Publishing Model: Hybrid
Deadline: Jun 30, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in