Red Edge Effect in the Service of Photocatalysis
Published in Chemistry

The project started when Dr. Sara Abednatanzi and Dr. Parviz Gohari Derakhshandeh affiliated to Ghent University, Belgium approached me to cooperate on photocatalysis with their covalent triazine frameworks (CTFs).
While conducting spectroscopic characterization, my team found that the CTFs exhibit strong red edge effect (REE) – the position of fluorescence maximum shifts gradually from shorter to longer wavelengths when excitation wavelength is changed from UV to vis.
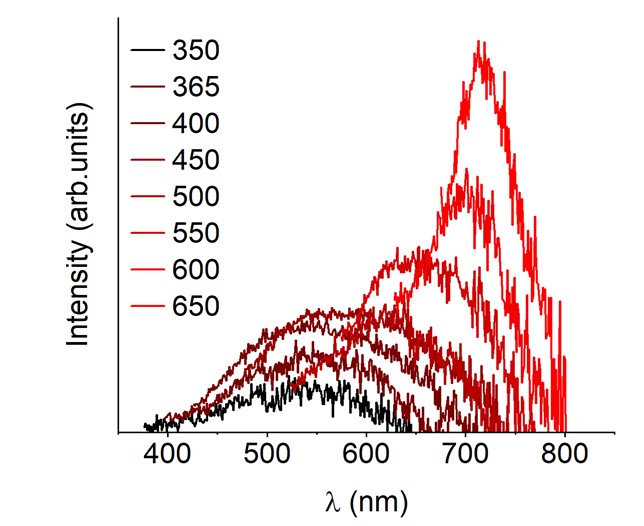
Therefore, the CTFs defy Kasha’s rule, according to which emission of a photon occurs only from the lowest excited state or vibrational sub-level. In other words, by selecting suitable photons we are able to generate CTF excited state of specific energy.
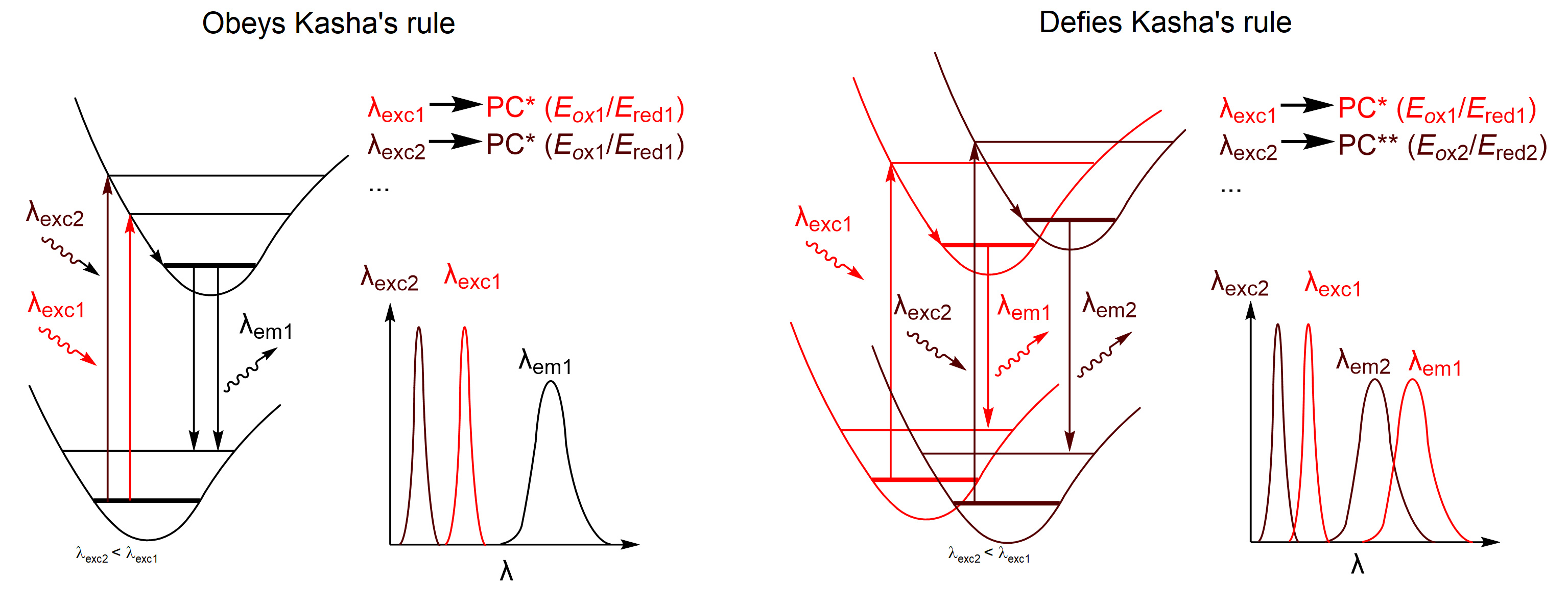
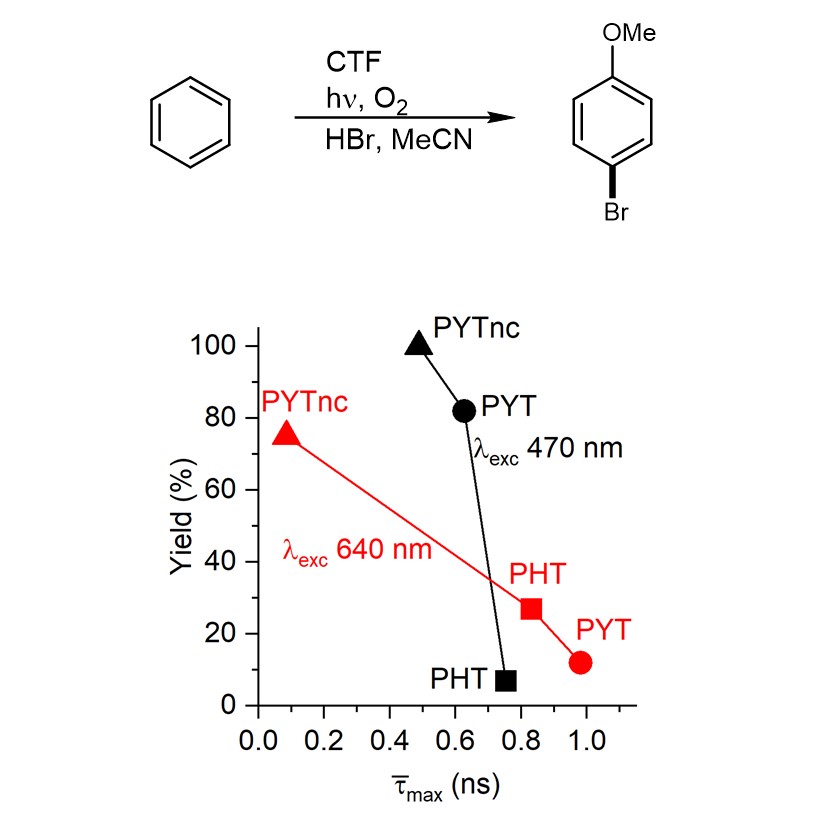
More important, the lifetime of such excited state is potentially long enough to engage in electron transfer, a redox process. Therefore, we concluded that REE may be applied in chromoselective photocatalysis. To verify this idea, we have chosen two reactions – oxidative bromination of aromatic compounds and dual Ni-photocatalytic C-N cross coupling. REE has important implications in these examples. In the first reaction, the yield of 4-bromoanisole obtained upon illumination at specific wavelength correlates with the fluorescence lifetime determined upon CTF excitation at the same wavelength.
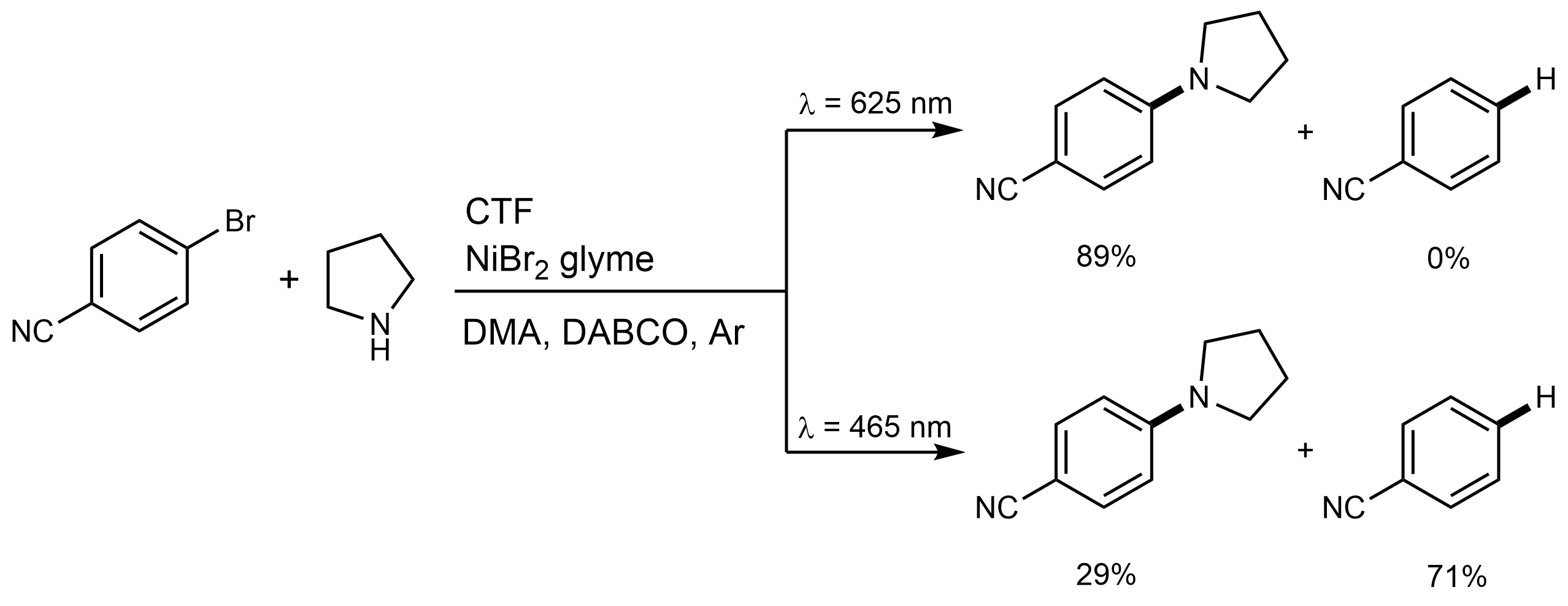
In the second reaction, we completely suppressed the undesirable dehalogenation of 4-cyanobromobenzene, which was the primary pathway under blue light, by employing photons on the edge of visible spectrum.
Indeed, earlier REE was observed in various carbon-based materials. Our work in Nature Communications proves that REE might serve as an express indicator to conclude whether or not a material is capable to mediate chromoselective photocatalysis.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in