Identification of 27 allele-specific regulatory variants in Parkinson’s disease using a Massively Parallel Reporter Assay
Published in Neuroscience, Protocols & Methods, and Cell & Molecular Biology
Parkinson’s disease – a complex disease
Despite being described more than 200 years ago, the fundamental cause or causes of Parkinson’s disease (PD) are yet to be fully understood. As a result, there are no disease-modifying therapeutics for PD, and individuals remain reliant on symptomatic relief. While PD is predominantly considered as a sporadic disease, substantial evidence implicates the involvement of underlying genetic factors (i.e., variants [single nucleotide polymorphisms]) in determining an individual’s risk of developing PD. Understanding the biological mechanisms through which these genetic variants act to increase risk of PD is critical for advancing towards the development of disease-modifying therapeutics for PD.
Genome Wide Association Studies – how do we assign function?
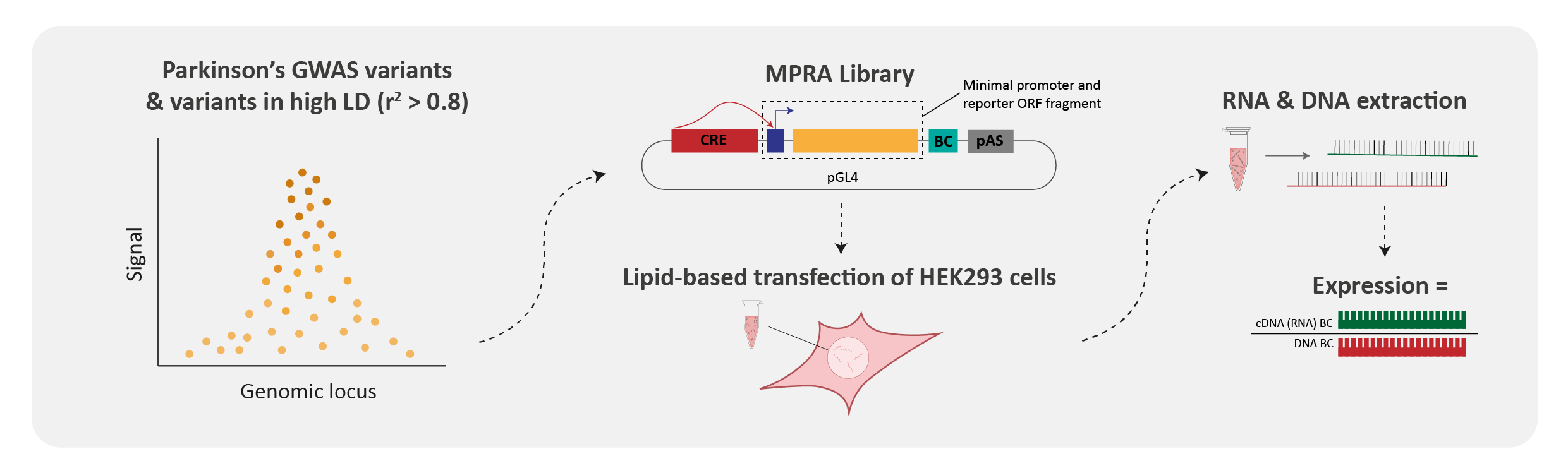
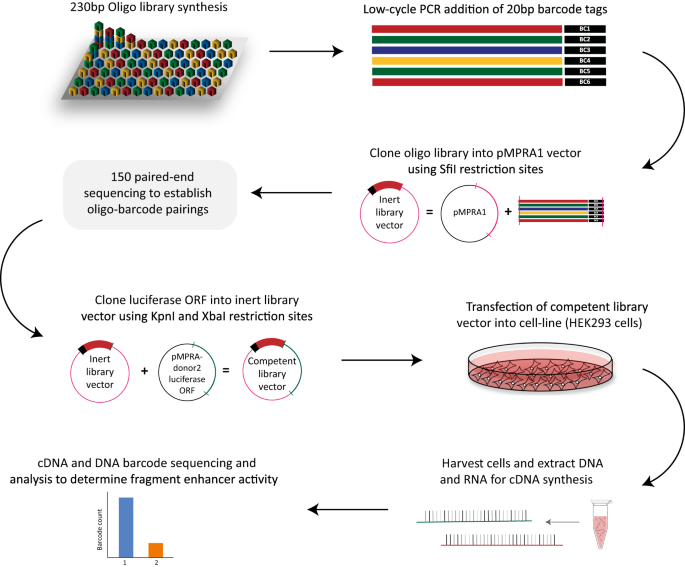
Genome wide association studies, or GWAS, essentially compare the DNA sequence of individuals with a particular disease to healthy matched controls. Although a simple concept, GWAS have been instrumental in identifying risk variants associated with almost every thinkable disease or trait, and at the time of concept were dubbed to revolutionise the field. However, knowing the disease risk variants is only the first piece of the puzzle. The next major challenge which largely still remains, is understanding how and why these variants lead to an increased risk of a particular disease, such as PD. This is particularly difficult given that over 95% of all variants identified by GWAS are located within non-coding regions of the genome, once referred to as ‘junk’ DNA. Massively parallel reporter assays (MPRA) offer one method by which we can start to understand how the disease variants are functioning. An MPRA is a high-throughput assay that allows one to screen thousands of different variants, and identify which variants are acting as regulators, and thus having a mechanistic impact, as opposed to being a bystander.
Key findings
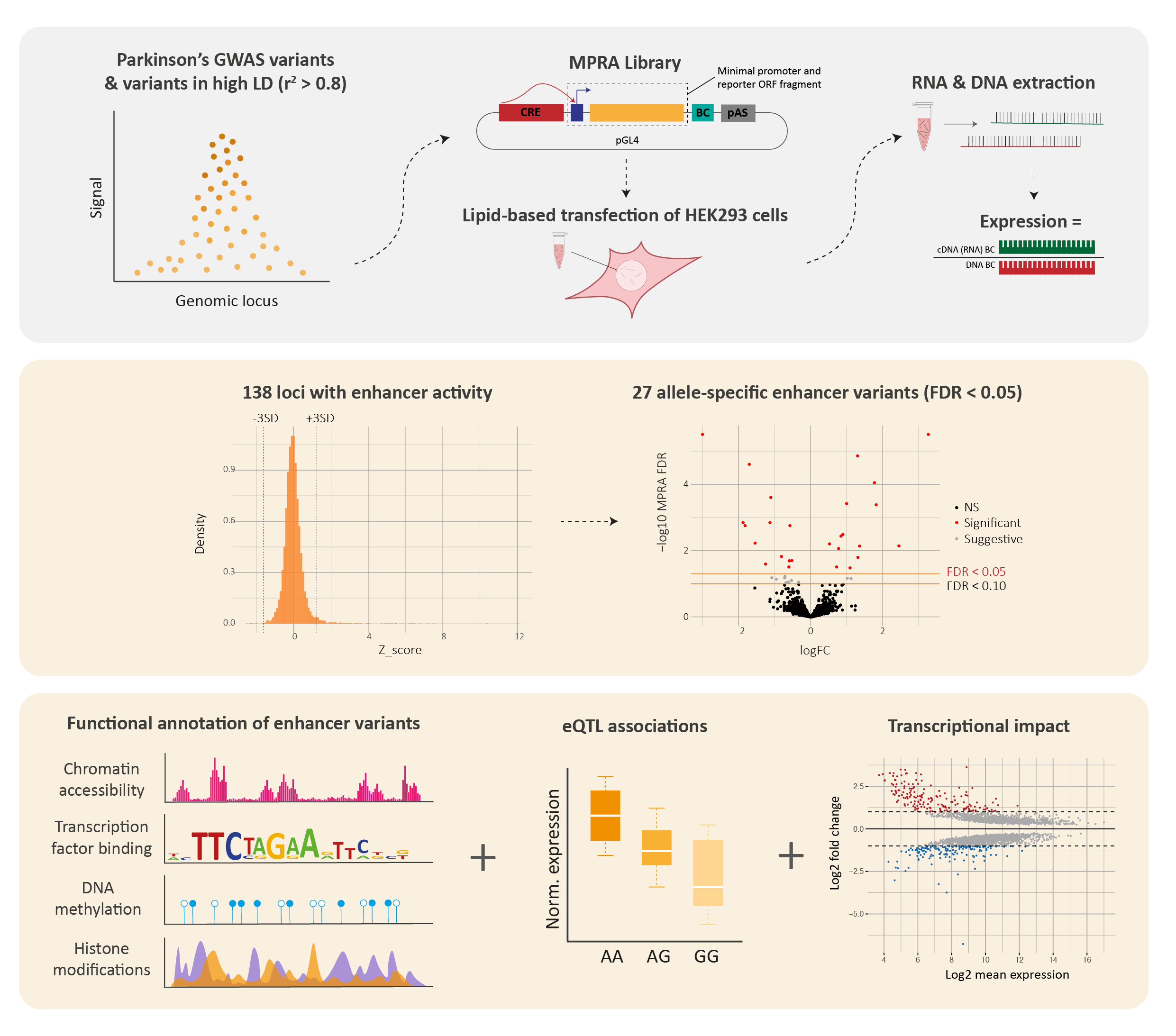
We tested the regulatory activity of over 5,000 variants and identified 138 variants with enhancer activity, of which 27 exhibited allele-specific regulatory activity in HEK293 cells. In agreement with previous studies, the variants that we identified to be acting as regulators (i.e., enhancers) were not typically the tag GWAS variant, indicating the need for deeper exploration of these loci.
Given our assay was conducted in HEK293 cells, we used several publicly available datasets to determine whether the enhancer variants that we identified within the MPRA were also likely to be active within brain cell-types or regions. ~60% of the MPRA enhancer variants overlapped with at least one epigenetic annotation (indicative of enhancer or promoter activity) in brain cell-types. Furthermore, analysis using the publicly available expression dataset GTEx showed that ~70% of the MPRA enhancer variants were associated with gene expression in brain tissues. Finally, we showed that the majority of MPRA allele-specific variants were predicted to disrupt at least one transcription factor binding site, indicating a potential mechanism through which these variants may be acting.
For a small subset of the identified allele-specific enhancer variants we used CRISPR-cas9 technology to generate isogenic cell-line pairs, in induced pluripotent stem cells [iPSCs], that differ at the base of interest. RNAseq analyses highlighted that the three interrogated variants affected the expression of multiple genes, thus confirming enhancer activity within their native chromatin context.
Implications and future directions
Our study provides a systematic characterisation of genetic risk loci associated with PD, and identified a subset of variants acting as allele-specific enhancers. Further interrogation of these variants through integration with functional assays, such as CRISPR interference assays, will strengthen our mechanistic understanding of the identified allele-specific enhancer variants within their native genomic context.
The main limitation of the study was the use of the HEK293 cell-line as opposed to a more PD-relevant cell-line (i.e., neuronal cell-line), which to some extent may limit the applicability of our findings when considering the function of these variants within the brain. Despite this, we and others have shown strong overlap between active regulatory regions in HEK293 cells and brain cell-types. There are also several well documented limitations associated with the MPRA method itself, such as sequence length of the regulatory element within the library; a lack of genome integration; and limited detection of weak regulatory activity. Nonetheless, as aforementioned, taking the identified enhancer variants forward for future functional work will help to alleviate some of these shortcomings.
Conclusions
This study provides insight into complex genetic loci that have previously been associated with PD, and provides a subset of validated regulatory variants that can be utilised in future studies exploring gene regulatory networks in PD. Moving forward, expanding our mechanistic understanding of how the identified regulatory variants exhibit risk will enable their utility in therapeutic target selection and for patient stratification, especially when considering genetically informed drug trials.
Follow the Topic
-
npj Parkinson's Disease
This journal publishes original basic science, translational and clinical research related to Parkinson's disease, including anatomy, etiology, genetics, cellular and molecular physiology, neurophysiology, epidemiology and therapeutic development and treatments.
Related Collections
With Collections, you can get published faster and increase your visibility.
The neuroimmune-axis and ageing in Parkinson’s Disease
Publishing Model: Open Access
Deadline: Jul 15, 2026
Cognition - preclinical models, and preclinical unmet need
Publishing Model: Open Access
Deadline: Jul 27, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in