iPSC-derived hippocampal neurons from discordant monozygotic twins to understand the pathophysiology of Schizophrenia
Published in Neuroscience
Schizophrenia and its elusive genetic component
Schizophrenia (SCZ) is characterized mainly by hallucinations, delusions, and mental confusion and occurs at the rate of approximately 1% in the general population1. Dr. Emile Kraepelin, a German psychiatrist, first described SCZ as a psychological disorder in 1887, under the name "dementia praecox" or "early dementia"2. Later, in 1911, Dr. Eugen Bleuler (a Swiss psychiatrist) renamed it "Schizophrenia" due to its distinct manifestation from dementia, combining the Greek words Schizo meaning "split "and, phrene meaning "mind"2. Dr. Kraeplin and Dr. Bleuer's subsequent work on subtyping SCZ symptoms along "positive" and "negative" dimensions serves as the foundation for the modern classification system provided by the DSM-5 (Diagnostic and Statistical Manual of Mental Disorders- fifth edition)2,3.
Genome-wide association studies (GWAS) primarily spearheaded by the Psychiatric Genomic Consortium (PGC) have found 170 genetic loci associated with SCZ patients4.
Several studies have investigated environmental factors like parental age, prenatal stress due to malnutrition or infection, substance abuse, socioeconomic status, etc. as potential risks for developing SCZ 4. However, studies of heritability estimates establish SCZ as one of the highly heritable neuropsychiatric illnesses (~80%), and hence the contribution of the genetic component appears to be dominant5. Animal models of SCZ are not appropriate due to its polygenic nature and in such a scenario SCZ patient-specific induced pluripotent stem cells (iPSCs) offer a unique opportunity to study the cellular and molecular pathways affected in patient-derived neurons.
Previously, Prof. Fred H. Gage's lab was among the first few labs to perform a detailed study of the iPSC-derived forebrain cortical neurons from patients diagnosed with familial SCZ in 20116. Decreased neuronal connectivity, reduced neurites, and loss in expression of PSD-95 (a postsynaptic protein) were found as a characteristic of SCZ neurons compared to control neurons6. This correlated with previous reports of thinner cortical areas and reduced hippocampal volumes in SCZ patients as documented by neuroimaging studies7.
Moving forward, in this publication we have investigated hippocampal neurons from monozygotic twins discordant for SCZ 8. Discordant monozygotic twins share the risk genes for SCZ yet the SCZ is not manifested in the unaffected co-twin, perhaps due to the presence of protective factors5. Hence, we hypothesized that investigating hippocampal neurons from discordant monozygotic twin pairs would help to understand the intricacies of molecular pathways in SCZ pathophysiology. At this point, it is important to mention that when the study began the co-twins were considered unaffected, but over the time period of the study, both co-twins were later diagnosed with Major depressive disorder. This work was initiated at Prof. Fred H. Gage's laboratory at the Salk Institute and was further completed and compiled in the laboratory of Prof. Shani Stern at the University of Haifa, Israel.
Findings from the SCZ iPSC-derived hippocampal neurons from discordant monozygotic twins
We analyzed two pairs of Monozygotic (MZ) twins discordant for SCZ. The unaffected co-twin siblings discordant for SCZ were diagnosed with Major depressive disorder. Also, we compared them to three pairs of healthy (control and unaffected) monozygotic twins. Hence, we had three groups:
- MZ co-twins with SCZ
- MZ co-twins with Major depressive disorder
- MZ twins who were healthy
iPSCs were reprogrammed from fibroblasts from these individuals and then further the hippocampal dentate gyrus (DG) granule neurons were derived from iPSCs using our previously published protocols9,10. The neurons were analyzed and compared for their morphology, histology for the expression of protein markers, transcriptomic markers using RNA-sequencing, and neurophysiology using whole cell patch clamp technique and multielectrode arrays (MEA).
We found that morphologically the iPSC-derived hippocampal DG neurons from SCZ patients were less arborized and displayed delayed maturation, while neurons derived from the co-twin displayed a mid-state between the SCZ and the control groups. Proper arborizations and timely maturation of neurons are important for forming neuronal connections for the appropriate functioning of neuronal networks.
Further, the RNA-seq analysis of the DG neurons derived from the SCZ patients compared to their unaffected co-twin and the control twins revealed dysregulation of synaptic pathways as well as the Wnt signaling pathway. Wnt signaling pathway dysregulation has been reported previously in both 2D and organoid-based 3D iPSC models by Prof Gage's group as well as other groups4 and in joint work of the two labs in bipolar disorder neurons.
The electrophysiological parameters of the SCZ group neurons were found to be altered compared to both co-twin and control groups. The SCZ patient-derived neurons were hypoexcitable and exhibited lower excitatory post-synaptic current rates than the other two groups. Interestingly, the co-twins were also less excitable when compared to the control group, but more excitable than their affected twin. Also, we found that the neurons formed three distinct groups based on their morphology and their electrophysiological parameters.
Interpretation and future outlook
The fact that both unaffected and affected twin pairs differ from control twins in terms of morphology and electrophysiology suggests an underlying germline genetic defect, which is corroborated by transcriptional profile differences. The role of epigenetic factors in disease onset in twin groups cannot be dismissed. However, the ability to detect a phenotype using iPSC-derived neurons suggests that genetic differences between patients are significant. Previous studies on postmortem brains found more severe changes in the morphology of SCZ neurons than in mood disorders or major depressive disorders in subiculum and cortical neurons, respectively11,12. This means that the phenotypic changes we observed in the patient's iPSC-derived neurons could point to relevant mechanisms underlying their symptoms. Finally, this strengthens the validity of iPSC-based models to study the underlying neuropathology of psychiatric disorders and investigate therapeutic approaches.
References:
- Tandon, R. et al. Definition and description of schizophrenia in the DSM-5. Schizophr Res 150, 3–10 (2013).
- Adityanjee, Aderibigbe, Y. A., Theodoridis, D. & Vieweg, W. V. R. Dementia praecox to schizophrenia: The first 100 years. Psychiatry and Clinical Neurosciences 53, 437–448 (1999).
- Nayak, R., Rosh, I., Kustanovich, I. & Stern, S. Mood Stabilizers in Psychiatric Disorders and Mechanisms Learnt from In Vitro Model Systems. Int J Mol Sci 22, (2021).
- Choudhary, A., Peles, D., Nayak, R., Mizrahi, L. & Stern, S. Current progress in understanding schizophrenia using genomics and pluripotent stem cells: A meta-analytical overview. Schizophr Res (2022) doi:10.1016/j.schres.2022.11.001.
- Cardno, A. G. & Gottesman II. Twin studies of schizophrenia: from bow-and-arrow concordances to star wars Mx and functional genomics. Am J Med Genet 97, 12–17 (2000).
- Brennand, K. J. et al. Modelling schizophrenia using human induced pluripotent stem cells. Nature 473, 221–225 (2011).
- Haijma, S. V. et al. Brain volumes in schizophrenia: a meta-analysis in over 18 000 subjects. Schizophr Bull 39, 1129–1138 (2013).
- Stern, S. et al. Monozygotic twins discordant for schizophrenia differ in maturation and synaptic transmission. Molecular Psychiatry (2024) doi:10.1038/s41380-024-02561-1.
- Sarkar, A. et al. Efficient Generation of CA3 Neurons from Human Pluripotent Stem Cells Enables Modeling of Hippocampal Connectivity In Vitro. Cell Stem Cell 22, 684-697.e9 (2018).
- Stern, S. et al. Mechanisms Underlying the Hyperexcitability of CA3 and Dentate Gyrus Hippocampal Neurons Derived From Patients With Bipolar Disorder. Biol Psychiatry 88, 139–149 (2020).
- Rosoklija, G. et al. Structural Abnormalities of Subicular Dendrites in Subjects With Schizophrenia and Mood Disorders: Preliminary Findings. Archives of General Psychiatry 57, 349–356 (2000).
- Larsen, N. Y. et al. Layer III pyramidal cells in the prefrontal cortex reveal morphological changes in subjects with depression, schizophrenia, and suicide. Translational Psychiatry 12, 363 (2022).
Follow the Topic
-
Molecular Psychiatry
This journal publishes work aimed at elucidating biological mechanisms underlying psychiatric disorders and their treatment, with emphasis on studies at the interface of pre-clinical and clinical research.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcement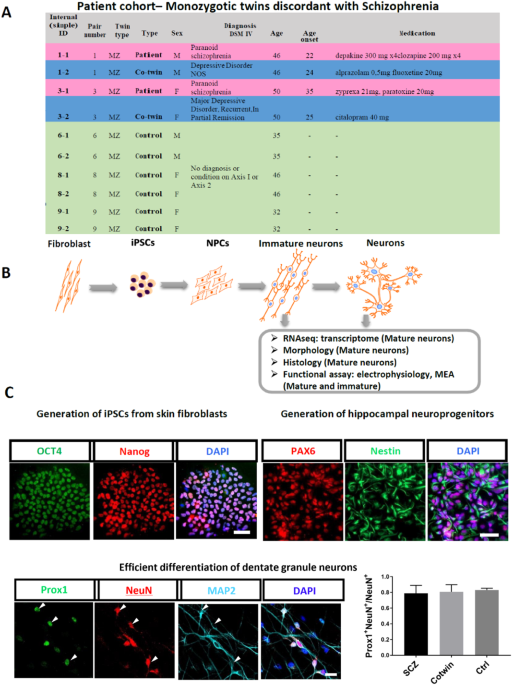






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in