New tricks from old drugs: repurposing the 80s
Published in Bioengineering & Biotechnology, Neuroscience, and General & Internal Medicine
Since their advent in the 1980s, the antidepressants of the selective serotonin reuptake inhibitor (SSRI) class have had a profound global impact, making billions for pharmaceutical corporations, providing inspiration for some memorable TV shows, and of course improving mental health for millions of people worldwide. Today, SSRIs remain by far the most prevalent psychotropic drugs, with over 80 million annual prescriptions in the UK alone. Despite this, their mode of action remains a bit of a mystery.
SSRIs are thought to work by preventing the brain cells from hoarding the neurotransmitter serotonin, thus increasing the amount of serotonin available to sustain the signalling pathways that prevent us from being depressed. There isn't, however, much evidence to show that these drugs do much to serotonin levels in the brain, and anyhow it's still not certain to what extent depression is actually caused by disbalance of serotonin (1). Besides, these drugs don't work for everyone - and when (or rather if) they do, the desired effect may take weeks to appear, often accompanied by numerous and sometimes unpleasant physical and mental side effects. More generally, how SSRIs affect the cells around the body is unknown, in part due to most cell-based studies to date using much higher drug concentrations than those observed in the bodies of the patients (2), and the resulting lack of insight has been nothing but... well, depressing.

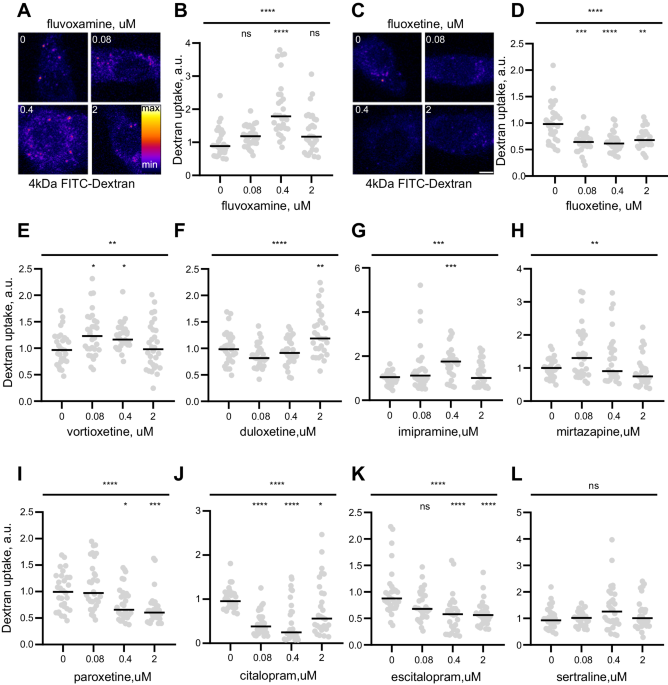
So, what do clinically relevant concentrations of SSRIs actually do outside the brain? To address this question, we took all the current SSRIs prescribed in the UK for depression, and considered their effects on cell biology of a kidney-derived cell line. Following from our earlier work (3,4), we focused on membrane trafficking, which encompasses a network of vesicular pathways for transporting and sorting of material in and out of cells. To our surprise, we found that (almost) all SSRIs affected membrane trafficking: some enhanced it, while others had the opposite effect. To make things even more interesting, in some cases, the same SSRI could either increase or decrease membrane trafficking depending on the concentration, suggesting that the effects of antidepressants are likely to be far more complex than just regulating serotonin levels in the brain.
To explore our observations in greater detail, we focused on one of the elder SSRIs: a venerable 40-year old drug called fluvoxamine, that has recently enjoyed something of a revival in the drug repurposing craze brought upon by the COVID-19 pandemic (5). In line with our earlier findings, we showed that fluvoxamine reliably increased membrane trafficking in several cell types, most likely by increasing the capacity of endosomes - the main interchange stations of the membrane trafficking network. We also confirmed that both inbound and outbound vesicular traffic was increased, and identified one of the fluvoxamine-boosted pathways as the classic textbook example of endocytosis, involving clathrin-coated pits. RNA sequencing and bioinformatic analysis showed hardly any effect of fluvoxamine, suggesting that the drug directly affected membrane trafficking rather regulated gene expression.
Our findings suggested that fluvoxamine could control traffic though the cells, known as transcytosis. One place where this effect could come handy is the blood-brain barrier, which protects the brain from the rest of the body. Compared to "normal" cells, endothelial cells comprising this barrier generally have very little transcytosis, making drug delivery into the brain very tricky. Could fluvoxamine induce enough transcytosis to ferry anything across the blood-brain barrier?
To answer this question, we first looked at endothelial cell cultures. Much like in kidney-derived cells, we saw that fluovoxamine enlarged the endosomes and boosted molecular transport in and out of the cells. Then, to establish whether this effect happens in vivo, we injected mice with fluvoxamine mixed with a fluorescent tracer dye that usually does not reach the brain, and looked at levels of fluorescence in their brains. Remarkably, a single shot of this cocktail was enough to induce a build-up of brain fluorescence, showing that fluvoxamine does indeed open up the blood-brain barrier to allow molecules that ordinarily wouldn't pass through.
These findings open many questions. How do different SSRIs regulate membrane trafficking on the molecular level? Can the effects of SSRIs be leveraged to control infection? Could other psychotropic drugs regulate membrane trafficking? How do SSRIs affect membrane trafficking in other organs? Is SSRI-regulated membrane trafficking involved in transport of serotonin? Are membrane trafficking effects linked to long-term complications of SSRI use, such as post-SSRI sexual dysfunction? While addressing these questions will require a lot of further work, detailed investigation of how SSRIs regulate membrane trafficking will provide much-needed insight into their desirable - and undesirable - effects in the brain and across the body.
In the meantime, the findings of this paper suggest that SSRIs could be used to help brain delivery of large therapeutic molecules, in particular monoclonal antibodies designed to treat neurodegenerative disorders such as Alzheimer's disease (6). Increased brain levels of these molecules will allow to decrease the required dosages, cutting down the currently prohibitive per capita treatment costs. Although the ability of SSRIs to aid delivery of other drugs still needs to be assessed in the clinic, the potential advantages of bringing new dementia treatments closer to millions of people worldwide should make it a worthwhile endeavour.
https://www.nature.com/articles/s41380-024-02626-1
References
(1) Moncrieff J &al. Mol Psychiatry. 2023 Aug;28(8):3243-3256. https://www.nature.com/articles/s41380-022-01661-0
(2) Casarotto PC &al. Cell. 2021 Mar 4;184(5):1299-1313.e19. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7938888/
(3) Glebov OO. FEBS J. 2020 Sep;287(17):3664-3671. https://febs.onlinelibrary.wiley.com/doi/10.1111/febs.15369
(4) Glebov OO. Front Pharmacol. 2021 Dec 16;12:787261. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8716620/
(5) Glebov OO &al. BMC Med. 2023 Jun 21;21(1):209. https://bmcmedicine.biomedcentral.com/articles/10.1186/s12916-023-02877-9
(6) van Dyck CH &al. N Engl J Med. 2023 Jan 5;388(1):9-21. https://www.nejm.org/doi/full/10.1056/NEJMoa2212948
Follow the Topic
-
Molecular Psychiatry
This journal publishes work aimed at elucidating biological mechanisms underlying psychiatric disorders and their treatment, with emphasis on studies at the interface of pre-clinical and clinical research.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in