Quantifying bias introduced by sample collection in human gut microbiome measurements
Published in Microbiology
Accurate microbial measurements are crucial for understanding associations between the human gut microbiome and human health. Traditionally, studies use DNA or RNA sequencing to measure and report the proportions, or “relative abundance”, of microorganisms, genes, or RNAs in a stool sample. Prior studies have demonstrated that experimental choices, from stool collection to bioinformatic analysis, can affect the measured relative abundances of microbes and microbial genes within a sample1. Additionally, most microbiome studies are limited to measuring which microbes are in the gut, but not how many are in the gut (the “absolute abundance”). It is becoming increasingly evident that absolute abundance provides a richer source of information and has implications for understanding interactions between microbes and the host that are concentration-dependent2–4. It remains unknown how experimental methods, such as stool preservation and storage conditions, impact measured microbial composition and concentration.
To shed light on this issue, we carried out a prospective study to assess the effect of preservative and storage choices on relative and absolute microbiome measurements. We collected stool samples from ten adult donors in California, USA, in each of two commercially available stool collection kits and also without preservatives, and stored them in a range of “real-world” temperature conditions. Additionally, to increase study transparency, we pre-registered our study objectives and statistical analysis plan with the Open Science Foundation5.
We found that the choice of preservative and storage temperature had a significant impact on the relative and absolute abundances of common gut microbes among donors, thus confounding microbial community measurements. Taxonomic diversity of samples was significantly different across preservation methods, with lower alpha diversity in preserved samples compared to those that were immediately frozen without a preservative. Use of preservatives introduced variation in observed relative abundances at both the metagenomic and metatranscriptomic level. Samples stored in Zymo DNA/RNA Shield buffer showed additional biases in metagenomic composition at 23°C and 40°C.
Further, we measured the absolute abundance of microbes in collected samples by using a simple and robust method that targets the bacterial/archaeal 16S ribosomal RNA gene by qPCR6. We found that samples that were immediately frozen without a preservative had an average of 1.33 x 1012 prokaryotes per gram of dry stool (Fig. 1a). Using an estimated value of colon volume, we calculated an ~100 trillion (1.2 x 1014) total prokaryotes in the human gut, approximately 3.2x higher than a previous widely cited estimate7. Surprisingly, we found that measurements of absolute microbial count were substantially impacted by storage conditions. Samples stored in OMNIgene GUT OMR-200 buffer had higher measured prokaryotic concentration while Zymo-preserved stool had lower measured prokaryotic concentrations compared to unpreserved samples (Fig. 1a). We used these concentration measurements to identify which taxa contributed to changes found in relative abundance profiles. Samples stored in OMNIgene preservative had higher concentrations of both major bacterial phyla (Bacteroidetes and Firmicutes) relative to no preservative controls, with greater enrichment of Bacteroidetes relative to Firmicutes. Zymo preserved samples showed absolute depletion of both phyla, with greater depletion of Firmicutes relative to Bacteroidetes (Fig. 1b). We demonstrate that absolute quantification can identify changes in total prokaryotic load and the specific taxa that contribute to changing relative proportions.
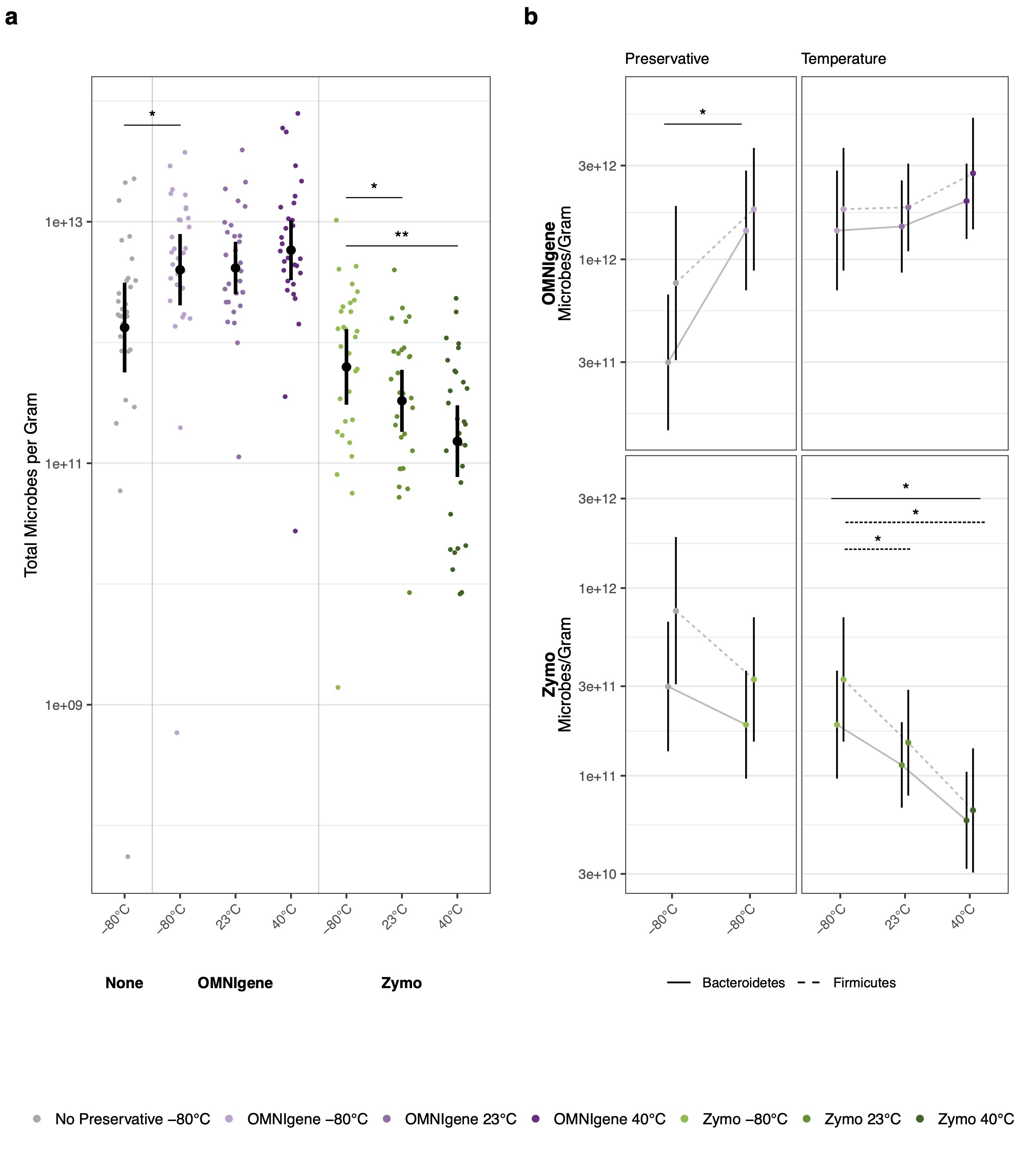
Our study highlights the importance and ease of measuring absolute abundance and emphasizes that sample storage practices can significantly affect observed microbial measurements. We recommend using the OMNIgene collection kits for large cohort microbiome studies, where cold-chain transport may not be feasible. For metatranscriptomic applications, we recommend using the Zymo collection kits, although samples preserved in the OMNIgene OMR-200 kit can be used in limited metatranscriptomic analyses. Our study has implications for current meta-analyses, for which samples may have been collected and stored differently, and also may explain the inconsistencies between studies that identify associations between microbiome composition and clinical outcomes.
In conclusion, it is essential to acknowledge that sample storage practices can significantly affect observed microbial measurements. We hope these findings will inform best practices for large cohort study design and enable researchers to optimize sample collection methods for their specific research questions. Additionally, we highlight the relative ease of measuring absolute concentration in microbiome studies, and we strongly recommend incorporating these measurements into common practice. We believe that total microbial load could potentially serve as an important biomarker for tracking disease progression and assessing treatment outcomes, and hope that future research will leverage this method to better correlate the microbiome and health outcomes.
References
- Sinha, R., Abnet, C.C., White, O., Knight, R., and Huttenhower, C. (2015). The microbiome quality control project: baseline study design and future directions. Genome Biology 16, 276. 10.1186/s13059-015-0841-8.
- Hagan, T., Cortese, M., Rouphael, N., Boudreau, C., Linde, C., Maddur, M.S., Das, J., Wang, H., Guthmiller, J., Zheng, N.-Y., et al. (2019). Antibiotics-Driven Gut Microbiome Perturbation Alters Immunity to Vaccines in Humans. Cell 178, 1313-1328.e13. 10.1016/j.cell.2019.08.010.
- Vandeputte, D., Kathagen, G., D’hoe, K., Vieira-Silva, S., Valles-Colomer, M., Sabino, J., Wang, J., Tito, R.Y., De Commer, L., Darzi, Y., et al. (2017). Quantitative microbiome profiling links gut community variation to microbial load. Nature 551, 507–511. 10.1038/nature24460.
- Rao, C., Coyte, K.Z., Bainter, W., Geha, R.S., Martin, C.R., and Rakoff-Nahoum, S. (2021). Multi-kingdom ecological drivers of microbiota assembly in preterm infants. Nature 591, 633–638. 10.1038/s41586-021-03241-8.
- Dahlen, A. (2022). Microbiome Benchmarking: Stability and Variability with Cold-Chain Break. 10.17605/OSF.IO/RF2JK.
- Jian, C., Luukkonen, P., Yki-Järvinen, H., Salonen, A., and Korpela, K. (2020). Quantitative PCR provides a simple and accessible method for quantitative microbiota profiling. PLOS ONE 15, e0227285. 10.1371/journal.pone.0227285.
- Sender, R., Fuchs, S., and Milo, R. (2016). Revised Estimates for the Number of Human and Bacteria Cells in the Body. PLoS Biol 14, e1002533. 10.1371/journal.pbio.1002533.
Follow the Topic
-
Nature Biotechnology
A monthly journal covering the science and business of biotechnology, with new concepts in technology/methodology of relevance to the biological, biomedical, agricultural and environmental sciences.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in