Tumor-Osteoclast Spatiotemporal Coupling: A Cell Behavior Target for Bone Metastasis Prevention
Published in Cancer, Materials, and Anatomy & Physiology
Why is osteoclast important?
Although the importance of bone metastasis has been widely recognized, the detailed process and mechanism still need to be explored. Unlike primary tumors, which are usually treated with local surgery or radiotherapy, bone metastasis is a systemic disease. During the process, the detection of metastases changes from difficult to simple, but the difficulty of treatment also increases with the development of the metastases. Meanwhile, the low proliferation and immune editing of early metastases further increase the complexity of immune recognition. Therefore, achieving effective early intervention is a challenge in preventing bone metastasis.
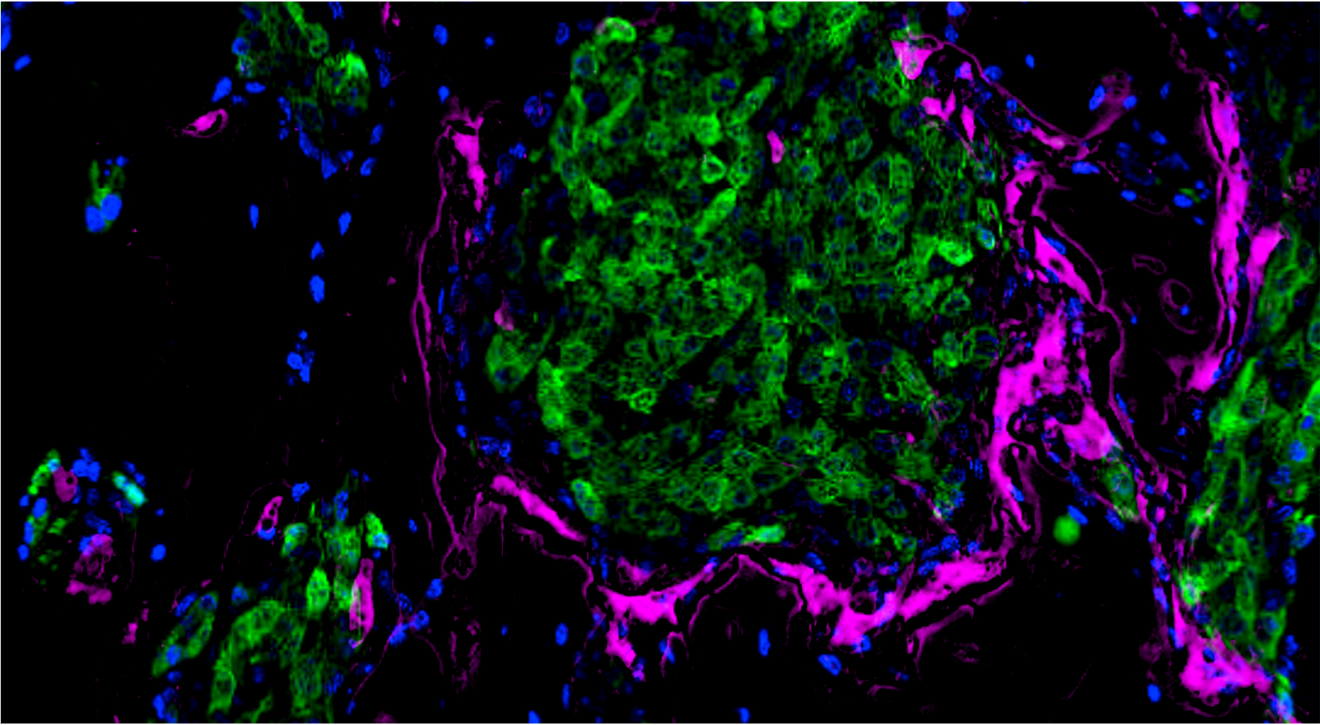
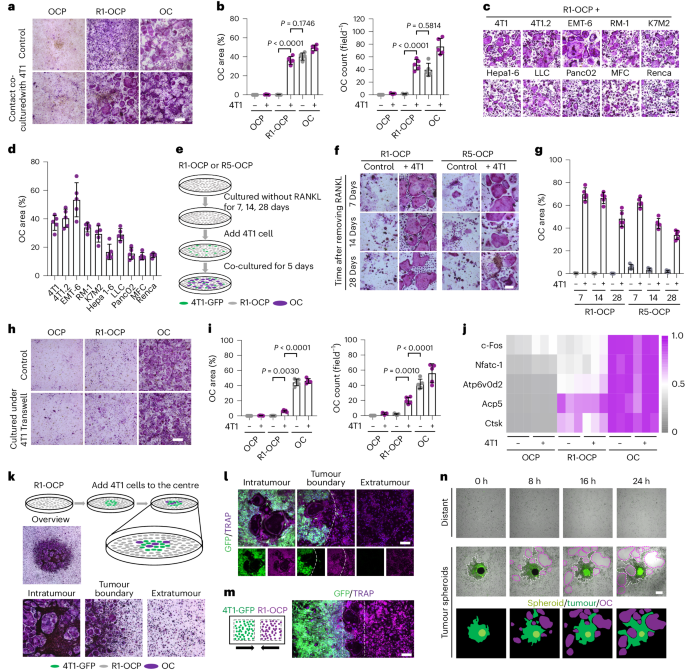
Due to the "hard base" of bone tissue, osteoclast, the only cell with bone resorption ability, are considered as the necessary "pioneer" for the development of bone metastases. However, in previous studies, the formation of osteoclasts in tumors has been reported to be related to the secretion of exosomes, cytokines and other factors by tumor cells, but this induction effect is not very strong. In fact, in our long-term experiments, we have found that osteoclasts often surround the metastatic lesion, as shown in poster image. This phenomenon has led us to speculate whether there is an in situ spatial interaction between tumor cells and osteoclasts, in addition to the effects of exosomes and cytokines?
How to explore the spatiotemporal relationship?
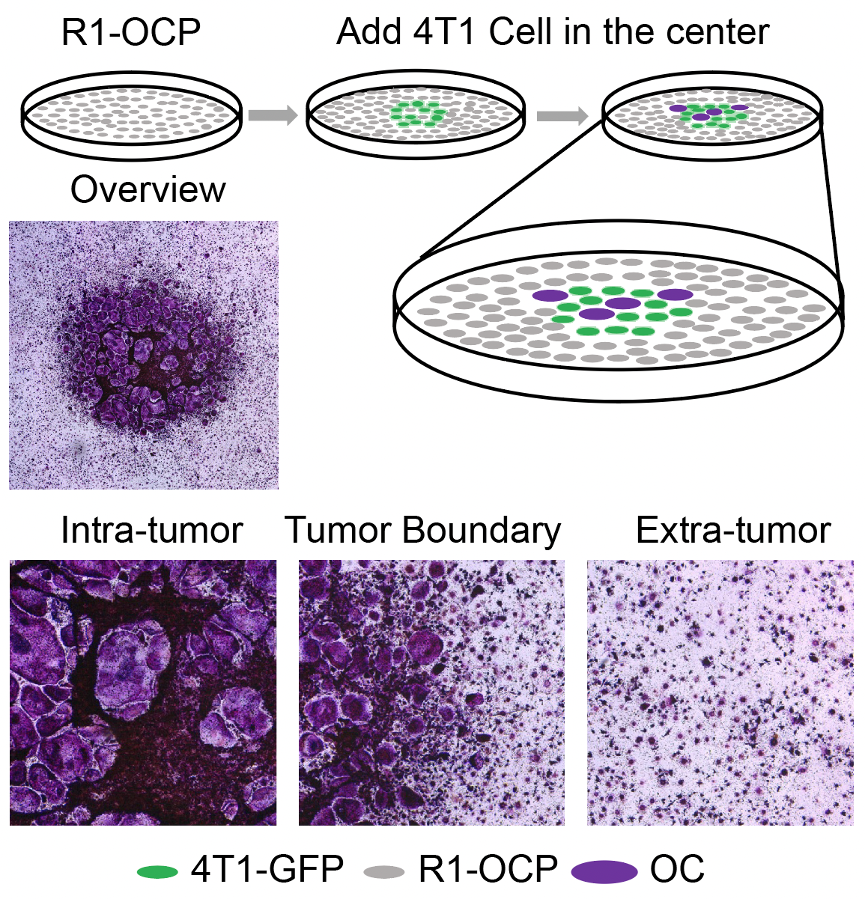
Figure 1. Schematic representation of culturing 4T1-GFP in the middle of R1-OCP for osteoclast induction with TRAP staining image of intra-tumor, tumor-boundary and extra-tumor regions.
Is it a special type of osteoclast?
We found that this type of osteoclast is induced by tumor cell cytoplasmic transfer mediated by migrasome. The currently known cytoplasmic contents that undergo transfer include proteins and mRNA. In particular, we found that this osteoclast is RANKL-independent and activated by non-classical AP-1 transcriptional complex. Therefore, it is special in terms of induction mode (contact required), induction mediator (migrasome) and transcription mode (alternative transcription factor pathway). Therefore, in order to distinguish it from previously reported osteoclasts, we named it “Tumasteoclast” (TAOC). The definition of this new subtype of osteoclasts may bring new content to the study of bone metastasis.
What is behavior targeting?
Due to the cell diversity in metastases, single molecule or single pathway targeted therapies may have off target risks. However, in the process of bone metastasis, the cellular behavior of constructing the tumor microenvironment itself is conservative, especially the formation of necessary TAOC. In tumor-TAOC coupling, TAOC is enormous and has strong destructive power, with tumor cells adjacent to TAOC. This relationship is very similar to "infantry-armor cooperation". To target this TAOC coupling behavior, we draw inspiration from anti-tank landmines and aim to construct a cell landmine that is specifically triggered by TAOC to simultaneously kill tumor cells and TAOC.
In the bone targeted liposomes we constructed, sodium bicarbonate ruptures the liposomes triggered by hydrogen ions secreted by osteoclasts, releasing high concentrations of sodium hydrogen phosphate for in-situ production of calcium phosphorus crystals and physical killing of tumor cells. This physical disruption of membrane integrity effectively avoids the biochemical tolerance of tumor cells, and further activate the immune response through immunogenic cell death.
Future directions
In our study, we reported the spatiotemporal coupling relationship between tumor cells and TAOC. This spatiotemporal coupling relationship may exist widely among various cells. Detailed exploration of these cellular behaviors can deepen our understanding of cellular interactions. Meanwhile, the behavior-targeting strategy may provide inspiration for more research on avoiding pharmacological and biochemical resistance in tumors. - Chenhui Gu and Xianfeng Lin are at Sir Run Run Shaw Hospital, Zhejiang University School of Medicine, Hangzhou, China.
Follow the Topic
-
Nature Nanotechnology
An interdisciplinary journal that publishes papers of the highest quality and significance in all areas of nanoscience and nanotechnology.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in