Unveiling the aggregation and cytotoxicity of TDP-43 carboxy-terminal fragments
Published in Neuroscience and Cell & Molecular Biology
Protein aggregation is a hallmark of neurodegenerative diseases, such as Amyotrophic Lateral Sclerosis (ALS), Frontotemporal Dementia (FTD), Alzheimer’s disease (AD), and Parkinson’s disease (PD). Protein aggregates are cytotoxic and cause severe neuronal effects, resulting in neuronal cell death and neurodegeneration 1. In 2006, TDP-43 was first identified as a constituent protein of aggregates in motor neurons of patients with ALS and FTD 2,3. These aggregates have been reported to contain not only full-length TDP-43, but also various C-terminal fragments (CTFs) 3. In 2008, missense mutation-infected SNPs were identified in patients with ALS/FTD, increasing the suspicion of their involvement in the onset of these diseases 4. Since then, researchers worldwide have investigated the mechanisms responsible for the formation and cytotoxicity of TDP-43 aggregates 5.
Amyloids are well-known structures of protein aggregates, gaining popularity for being formed by amyloid-beta peptides associated with AD. However, not all protein aggregates are amyloids. Recently, liquid-like structures called droplets formed by liquid‒liquid phase separation, have begun to be understood. These structures, where proteins and biomolecules accumulate within cells, are diverse 6.
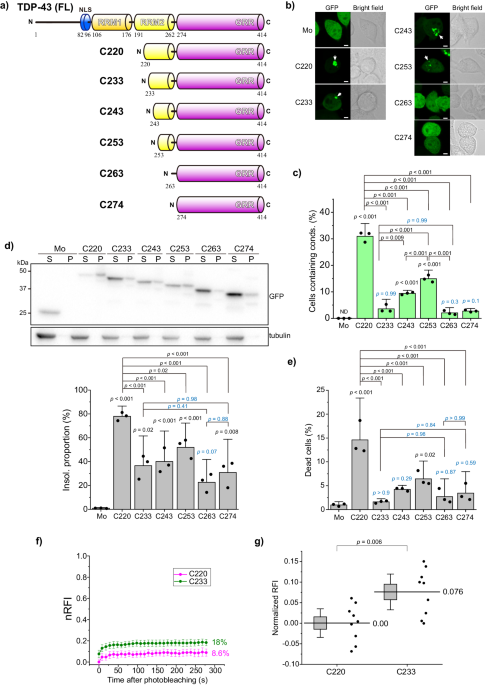
TDP-43 and its CTFs form aggregates, a concept that is rarely questioned. However, many questions remain regarding the limitations of these structures to amyloids and the aggregation processes they undergo. We addressed these questions by focusing on the aggregation process of a 25 kDa TDP-43 CTF (220-414 amino acids; TDP25/C220) in a recently published article 7. TDP-43 has an N-terminal multimerization domain (NTD), two RNA recognition motifs (RRM1/2), and an intrinsically disordered region (IDR) at its C-terminus (274-414 amino acids, C274). The IDR, which its diverse conformations and unstructured conformations, is believed to be prone to aggregation or condensation. However, TDP-43's IDR has been reported to rarely form condensates within cells 7,8. How much of the N-terminal amino acid sequence must be lost from C220 to approach the non-aggregating nature of IDR (such as C274)?
Classical genetic engineering methods often involve deletion mutants, a popular approach due to the advancements in molecular biology and genetic engineering techniques, allowing for the design of recombinant proteins expressed within cells. When amino acids are sequentially deleted from the N-terminus of TDP25/C220, they may be expected to gradually or abruptly approach the properties of IDR. However, this was not the case. Although C233, lacking 13 residues from C220, showed the lowest condensation population, C243, which lacks an additional 10 residues, showed a higher condensation population than C233. This may be due to the misfolding of the truncated RRM2 (tRRM2) corresponding to the deleted region, which dictates the condensation properties, likely leading to a conformational transition in the IDR. Furthermore, C220 and the tRRM2 region (220-262 amino acids; F220) formed cytoplasmic condensates, stained with amyloid-specific fluorescent probes. However, not all condensates contained amyloids, suggesting heterogeneous and diverse formations. This implies that condensates with different properties were formed within a single cell.
To evaluate the condensation and assembly properties of TDP-43 CTFs, a method that can control spatiotemporal intermolecular assembly within living cells is essential. We used an optogenetic tag (CRY2olig) derived from a modified plant cryptochrome that induces protein assembly upon blue light irradiation 9. C220 did not form condensates even when optogenetically induced to assemble from either the N- or C-terminus, whereas the IDR of the TDP-43 CTF was transiently assembled. Furthermore, forcing N-terminal assembly resulted in almost all cells transiently forming condensates, whereas forcing C-terminal aggregation made it difficult to form condensates. This suggests that following the N-terminal aggregation of the TDP-43 IDR, structural changes occur towards the C-terminus, resembling a zipper mechanism.
While investigating the impact of such aggregates and condensates on biological activities, an analysis of the frequency of cell death in cultured cells revealed a positive correlation between condensate formation and the proportion of cell death. In contrast, an analysis using the model organism Caenorhabditis elegans showed that C233 formed highly dynamic condensates, whereas C220 formed less dynamic condensates. Worms expressing C220 or C233 in their body wall muscles exhibited decreased motility; however, interestingly, C220 and C233 shortened the lifespan of the worms, suggesting that these aggregates may affect longevity non-autonomously.
Fluorescence correlation spectroscopy (FCS) analysis 10,11 of soluble oligomers of C220 and C233 has suggested that C233 exists as homo-oligomers, whereas C220 exists as hetero-oligomers, involving endogenous intracellular proteins. Oligomers that sequester essential amounts of cellular proteins can dysregulate protein homeostasis (proteostasis) and cause a loss of function, possibly leading to cytotoxicity.
The well-established concept that “aggregation = toxicity” still lacks a clear explanation of why it is toxic. Understanding the onset of ALS/FTD for TDP-43 and its CTFs is a critical biomedical issue. Although the day when a multifaceted approach involving molecular cell biology, biochemistry, and biophysics will unveil this mystery may be near, diligent research that steadily peels away the layers of the enigma is crucial.
References
1 Ross, C. A. & Poirier, M. A. Protein aggregation and neurodegenerative disease. Nature Medicine 10 Suppl, S10-17, doi:10.1038/nm1066IF: 82.9 Q1 (2004).
2 Arai, T. et al. TDP-43 is a component of ubiquitin-positive tau-negative inclusions in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Biochemical and Biophysical Research Communications 351, 602-611, doi:10.1016/j.bbrc.2006.10.093IF: 3.1 Q2 (2006).
3 Neumann, M. et al. Ubiquitinated TDP-43 in frontotemporal lobar degeneration and amyotrophic lateral sclerosis. Science 314, 130-133, doi:10.1126/science.1134108IF: 56.9 Q1 (2006).
4 Sreedharan, J. et al. TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis. Science 319, 1668-1672, doi:10.1126/science.1154584IF: 56.9 Q1 (2008).
5 Berning, B. A. & Walker, A. K. The Pathobiology of TDP-43 C-Terminal Fragments in ALS and FTLD. Frontiers in Neuroscience 13, 335, doi:10.3389/fnins.2019.00335IF: 4.3 Q2 (2019).
6 Shin, Y. & Brangwynne, C. P. Liquid phase condensation in cell physiology and disease. Science 357, doi:10.1126/science.aaf4382IF: 56.9 Q1 (2017).
7 Kitamura, A. et al. Hetero-oligomerization of TDP-43 carboxy-terminal fragments with cellular proteins contributes to proteotoxicity. Communications Biology, 7, 743, doi: 10.1038/s42003-024-06410-3IF: 5.9 Q1 (2024).
8 Kitamura, A. et al. Interaction of RNA with a C-terminal fragment of the amyotrophic lateral sclerosis-associated TDP43 reduces cytotoxicity. Scientific Reports 6, 19230, doi:10.1038/srep19230IF: 4.6 Q2 (2016).
9 Taslimi, A. et al. An optimized optogenetic clustering tool for probing protein interaction and function. Nature Communications 5, 4925, doi:10.1038/ncomms5925IF: 16.6 Q1 (2014).
10 Kitamura, A. et al. Cytosolic chaperonin prevents polyglutamine toxicity with altering the aggregation state. Nature Cell Biology 8, 1163-1170, doi:10.1038/ncb1478IF: 21.3 Q1 (2006).
11 Kitamura, A. & Kinjo, M. State-of-the-Art Fluorescence Fluctuation-Based Spectroscopic Techniques for the Study of Protein Aggregation. International Journal of Molecular Sciences 19, doi:10.3390/ijms19040964IF: 5.6 Q1 (2018).
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
DNA repair and human disease
Publishing Model: Hybrid
Deadline: Oct 31, 2026
Cell death and inflammatory signalling
Publishing Model: Hybrid
Deadline: Oct 28, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in