Vanadium Vanguard: Navigating Extraction and Utilization for a Sustainable Future
Vanadium (V), a critical metal with diverse applications across industries, presents both opportunities and challenges in its extraction and utilization. Its significance in strengthening steel for construction, enabling energy storage in batteries, and driving innovations in renewable energy technologies underscores its critical role in modern society. However, the increasing demand for V and the limitations of traditional extraction methods have led to concerns regarding sustainable supply and environmental impact.
Conventional V extraction primarily relies on terrestrial mining operations, which are resource-intensive and environmentally disruptive. The scarcity of high-grade V ores exacerbates these challenges, resulting in price volatility and supply uncertainties. Moreover, the extraction process generates significant waste streams and environmental pollution, further underscoring the urgent need for sustainable alternatives.
In response to these challenges, unconventional approaches, such as harnessing waste streams and innovative extraction techniques, are an increasing focus. For example, the excess of 1 trillion litres of discharged oil sands tailings in Alberta, Canada, represents a valuable source rich in V (190 ppm) that can be recovered using advanced technologies. This approach creates value for an otherwise environmental waste and offers a sustainable solution for industries reliant on this critical metal.
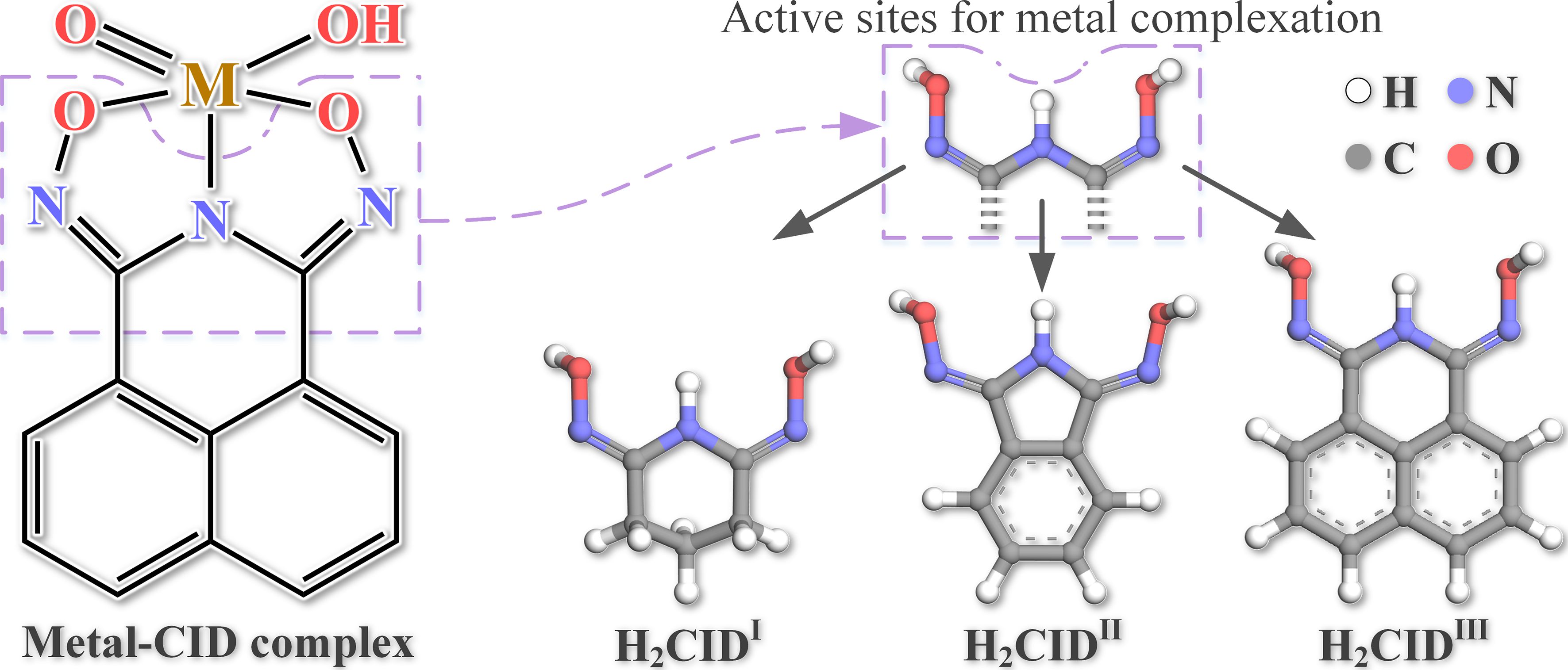
Chelating agents hold promise for recovering V from waste sources, yet achieving requisite selectivity poses a significant challenge. Waste sources typically harbor a blend of metal ions, organics, and other inorganic compounds, complicating V recovery. Additionally, these waste environments, often acidic, can challenge the structural stability of these extraction materials. Our research in Nature Communications presents the use of an acid-stable chelate, a naphthalimidedioxime (L), engineered specifically for efficient and selective extraction of V. Coupling complexation and precipitation steps, the technique (refer to Figure 1) allows for selective V recovery, even from an industrial waste stream, real tailings from oil sands.

Through a simple addition of L to the oil sands tailings leachate, which contains 19 competing metal ions ranging in concentration from 0.04 to 12465 mg/L, along with a total organic carbon (TOC) content of 695 mg/L and pH mediation, 95% of the V can be precipitated (Figure 2). Unlike traditional methods, the chelate demonstrates remarkable resistance to acids even in harsh industrial conditions. A key advantage of the chelate is its ability to facilitate V extraction without requiring attachment to a solid support, a common necessity in other extractants that often results in a heterogeneous extraction process limited by mass transfer. The scalability of L synthesis further enhances its practical applicability.
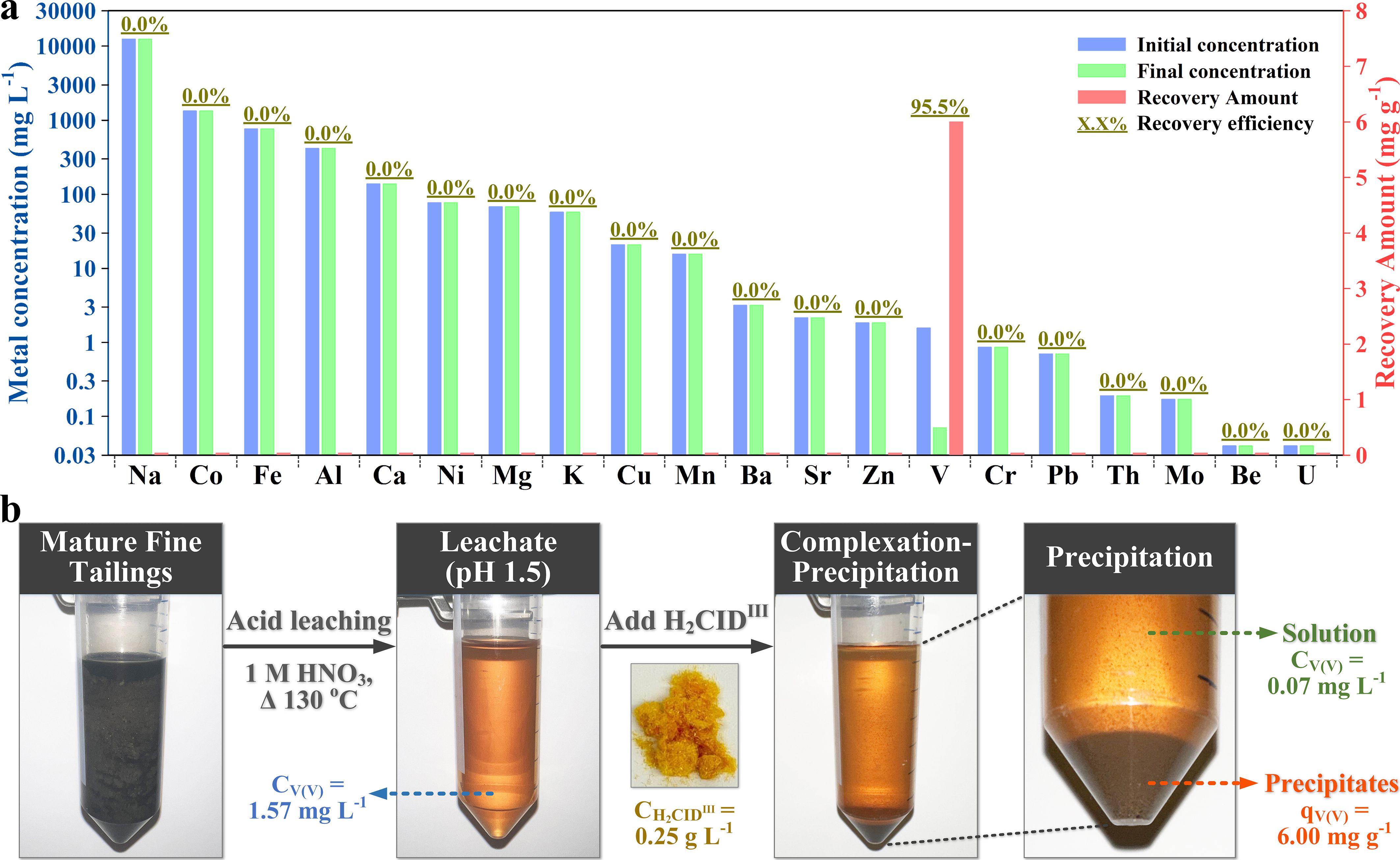
At the molecular scale, we have studied the nature of the complexation phenomenon. A key point is that, while a few metals are chelated, only complexes with V are anionic. This allows precipitation of exclusively the V complexes simply by pH variation. The V complexation characteristics encompass excellent acid resistance, swift adsorption kinetics, high adsorption capacities across a wide pH range, and straightforward desorption and reusability. Its high selectivity enables V isolation, even in complex wastewater streams containing multiple metals in higher concentrations. By minimizing undesired metal extraction and waste generation, this specific extraction method promotes resource efficiency and environmental stewardship, aligning with global sustainability objectives.
The implication of this innovation is potentially far-reaching. By leveraging waste streams as a source of V, we address environmental concerns and provide a sustainable solution for industries reliant on this critical, and increasingly used, metal. Ongoing work encompasses translating the molecular extraction features to an optimal process separation and the exploration of other industrial wastes as V sources.
Follow the Topic
-
Nature Communications
An open access, multidisciplinary journal dedicated to publishing high-quality research in all areas of the biological, health, physical, chemical and Earth sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Women's Health
Publishing Model: Hybrid
Deadline: Ongoing
Biosensing
Publishing Model: Hybrid
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in