The RNA-peptide world
Published in Chemistry
The RNA world hypothesis postulates that the first self-replicating systems were comprised only of RNA molecules1,2. Subsequently, the RNA world somehow evolved into the contemporary nucleic acid-protein world2-4. In the latter, RNAs carry genetic information that, for example, encodes the sequence of proteins, and proteins act as catalysts, for example, in RNA replication. This mutual dependence between RNAs and proteins creates a ‘chicken-or-egg’ (RNAs or peptides) conundrum for any theory related to the origin of life4. In this regard, the Carell group from the Ludwig-Maximilians-Universität (LMU) München became interested in addressing this issue.
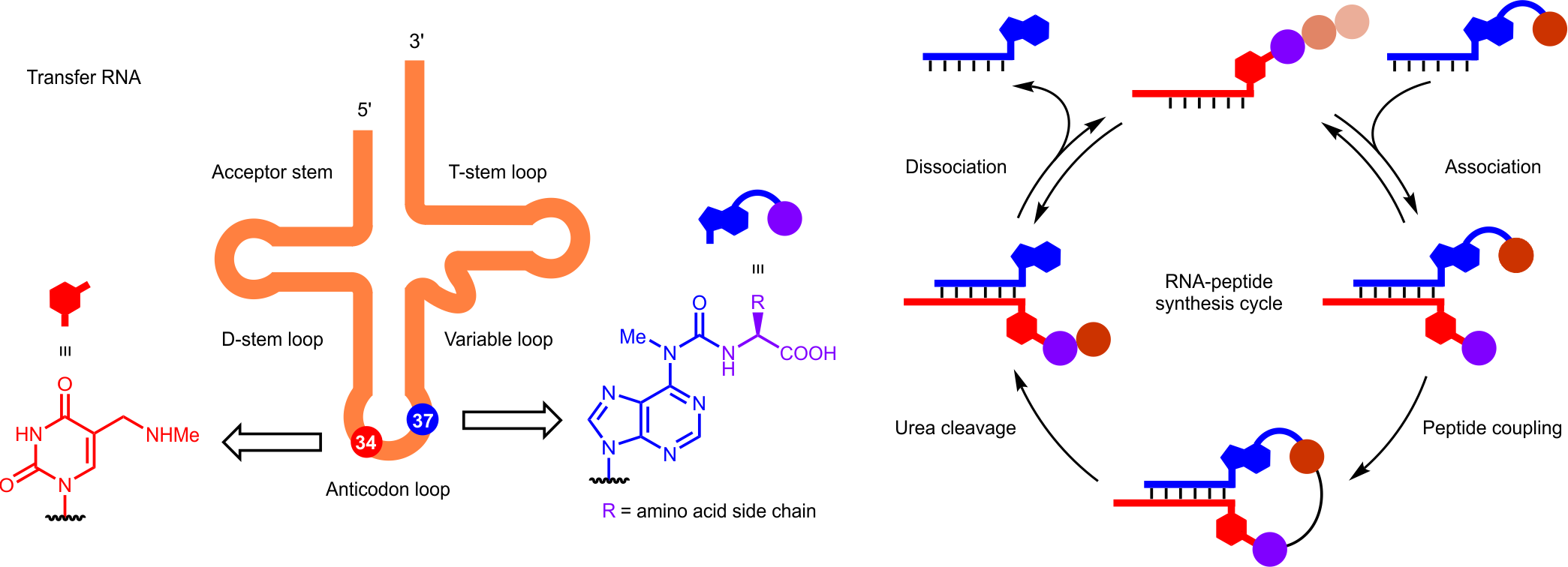
At the beginning of this study, we realized that contemporary transfer RNAs contain non-canonical nucleosides5 featuring carboxylic acid and amino groups (Fig. 1a). Specifically, adenosine is connected to an amino acid through a urea group and uridine bears a secondary amino group. In ribosomal translation, these non-canonical nucleosides affect the binding affinity and selectivity of codon-anticodon interactions6. In addition, both nucleosides are accessible through prebiotically plausible synthetic routes7,8 and, therefore, are considered as ‘fossils’ of an early RNA world6. With that in mind, we thought that the presence of these non-canonical nucleosides in complementary RNA strands could offer a prebiotic model for peptide synthesis on RNA.
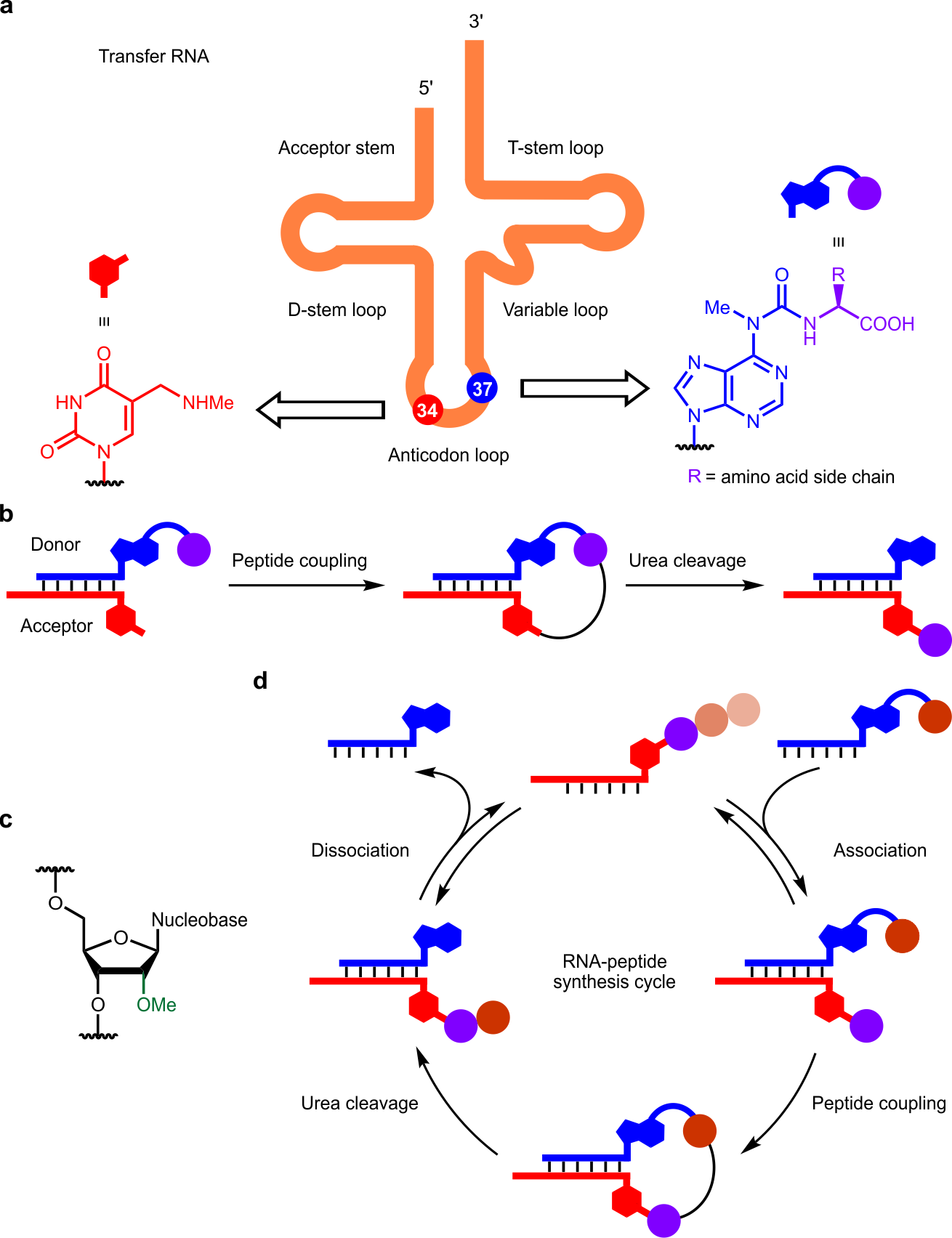
Fig. 1 | a, Transfer RNA and nucleobase modifications. b, Peptide coupling and urea cleavage reactions between RNA donor and acceptor strands. c, 2’-Methoxy modified ribose present in ribosomal RNA. d, Stepwise growth of peptides on RNA in an RNA-peptide synthesis cycle.
First, we prepared the phosphoramidite derivatives of the non-canonical nucleosides for their incorporation into two complementary RNA strands (Fig. 1b) using an automated solid-phase synthesizer. We designed an RNA strand (donor) containing an amino acid (or a peptide) modified adenosine at the 5’ end. The other RNA strand (acceptor) was equipped with a methylaminomethyl uridine at the 3’ end. Despite the apparent simplicity of the model system, the preparation of both the phosphoramidites and the oligonucleotides with a series of amino acids demanded a considerable synthetic effort from all co-authors of the article.
With the compounds in hand, we investigated the formation of a peptide bond between the terminal non-canonical nucleosides of the complementary RNA donor and acceptor strands (Fig. 1b) by means of high-performance liquid chromatography (HPLC) and matrix-assisted laser desorption/ionization coupled to time-of-flight (MALDI-TOF) mass spectrometry. Interestingly, we observed the formation of the desired product (that is a hairpin in which the terminal nucleobases are connected through an amino acid or a peptide) to a significant extent upon activation of the carboxylic acid in the RNA donor strand with both artificial9 and prebiotically plausible10 methods at slightly acidic pH conditions. Control experiments ruled out the formation of a putative ester bond between the carboxylic acid of the amino acid (or peptide) in the RNA donor strand with a ribose hydroxyl group in the RNA acceptor strand.
Next, we wondered whether the urea connection in the hairpin could be cleaved under prebiotically plausible conditions, leading to the transfer of the formed peptide to the RNA acceptor strand (Fig. 1b). Based on previous findings in the literature,11,12 we performed the urea cleavage reaction by heating the acidic aqueous solution of the hairpin at high temperature. Under these reaction conditions, we obtained a mixture of three products. One of them featured the sequence of the RNA donor strand but lacked the amino acid (or peptide). The two other products, derived from the RNA acceptor strand, contained the amino acid (or peptide) and its hydantoin derivative, respectively. The hydantoin is a side product in our prebiotic model, since it cannot be involved in a second amino acid/peptide transfer reaction (see below). Luckily, we found that, under specific reaction conditions, the amino acid/peptide-elongated RNA acceptor strand could be obtained as the major product.
We also noted that the urea cleavage reaction gave the amino acid/peptide-elongated RNA strand in low yield owing to the degradation of the RNA promoted by backbone hydrolysis. To overcome this issue, we included 2’-methoxy modified nucleosides (Fig. 1c) into our model. These non-canonical nucleosides are widespread in contemporary ribosomal RNAs13. In addition, their incorporation into the RNA acceptor strand minimized the backbone hydrolysis and, concomitantly, improved the efficiency of the urea cleavage reaction. Remarkably, both the described oligonucleotides and the applied experimental reaction conditions set a prebiotically plausible scenario for the stepwise growth of peptides covalently connected to RNA strands (Fig. 1d). In this scenario and in one-pot, we observed the growth of dipeptide- and tripeptide-elongated RNA strands in moderate yields.
In summary, we demonstrated the potential existence of an RNA-peptide world that is capable of growing peptides covalently attached to RNAs under prebiotically plausible conditions. The reported model enables the co-evolution of RNAs and peptides and, therefore, offers an answer to the ‘chicken-or-egg’ conundrum mentioned above. In this scenario, RNAs would be able to encode the sequence of the growing peptide, and peptides would display catalytic functions. We are excited to further explore the properties of the RNA-peptide world towards catalytic RNA-peptide chimeras using non-canonical nucleosides.
The Nature article can be found at: https://doi.org/10.1038/s41586-022-04676-3
References
- Gilbert, W. Origin of life: the RNA world. Nature 319, 618 (1986). https://doi.org/10.1038/319618a0
- Joyce, G. F. The antiquity of RNA-based evolution. Nature 418, 214-221 (2002). https://doi.org/10.1038/418214a
- Crick, F. H. C., Brenner, S., Klug, A. & Pieczenik, G. A speculation on the origin of protein synthesis. Orig. Life Evol. Biosph. 7, 389-397 (1976). https://doi.org/10.1007/BF00927934
- Bowman, J. C., Hud, N. V. & Williams, L. D. The ribosome challenge to the RNA world. J. Mol. Evol. 80, 143-161 (2015). https://doi.org/10.1007/s00239-015-9669-9
- Carell, T. et al. Structure and function of noncanonical nucleobases. Angew. Chem. Int. Ed. 51, 7110-7131 (2012). https://doi.org/10.1002/anie.201201193
- Grosjean, H. & Westhof, E. An integrated, structure- and energy-based view of the genetic code. Nucleic Acids Res. 44, 8020-8040 (2016). https://doi.org/10.1093/nar/gkw608
- Robertson, M. & Miller, S. Prebiotic synthesis of 5-substituted uracils: a bridge between the RNA world and the DNA-protein world. Science 268, 702-705 (1995). https://doi.org/10.1126/science.7732378
- Schneider, C. et al. Noncanonical RNA nucleosides as molecular fossils of an early Earth—generation by prebiotic methylations and carbamoylations. Angew. Chem. Int. Ed. 57, 5943-5946 (2018). https://doi.org/10.1002/anie.201801919
- Gartner, Z. J., Kanan, M. W. & Liu, D. R. Expanding the reaction scope of DNA-templated synthesis. Angew. Chem. Int. Ed. 41, 1796-1800 (2002). https://doi.org/10.1002/1521-3773(20020517)41:10<1796::AID-ANIE1796>3.0.CO;2-Z
- Liu, Z. et al. Harnessing chemical energy for the activation and joining of prebiotic building blocks. Nat. Chem. 12, 1023-1028 (2020). https://doi.org/10.1038/s41557-020-00564-3
- Hutchby, M. et al. Hindered ureas as masked isocyanates: facile carbamoylation of nucleophiles under neutral conditions. Angew. Chem. Int. Ed. 48, 8721-8724 (2009). https://doi.org/10.1002/anie.200904435
- Ohkubo, A. et al. New thermolytic carbamoyl groups for the protection of nucleobases. Org. Biomol. Chem. 7, 687-694 (2009). https://doi.org/10.1039/B816831H
- Decatur, W. A. & Fournier, M. J. rRNA modifications and ribosome function. Trends Biochem. Sci. 27, 344-351 (2002). https://doi.org/10.1016/S0968-0004(02)02109-6
Follow the Topic
-
Nature
A weekly international journal publishing the finest peer-reviewed research in all fields of science and technology on the basis of its originality, importance, interdisciplinary interest, timeliness, accessibility, elegance and surprising conclusions.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in